Abstract
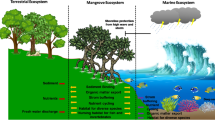
The Sundarbans is the largest continuous block of mangrove distributed over both India and Bangladesh and is threatened with pronounced erosion and mudflat degradation in its western reaches. Since 2013, considerable progress has been achieved through the restoration of highly degraded mudflats by the extensive utilization of four native grass species plantings, Porteresia coarctata, Myriostachya wightiana, Paspalum vaginatum, and Sporobolus virginicus. These grasses proved to exhibit the highest survival (80–90%) in the lower and middle intertidal zone and during the last three years (2013–2016) almost 6000 m2 area have been restored by the growth of these grasses. Increased osmolyte synthesis under degraded conditions in these native grasses offered osmotic protection and helped to expand and restore the mudflat to an elevation suitable for mangrove establishment. The rhizosphere of the four grasses was engaged in the decomposition cycle, releasing more nutrients, more nutrient cycling soil microbial enzymes, and encouraged high densities of nutrient cycling bacteria. The study demonstrates that stabilization and reclamation of the denuded mudflat by halotolerant grasses is an essential prerequisite for any successful mangrove restoration program. The facilitative role of the salt marsh is crucial to aid colonization by mangrove propagules.







Similar content being viewed by others
References
Atwell, B., P. Kriedemann & C. Turnbull, 1999. Plants in Action: Adaptation in nature, Performance in Cultivation. Macmillan Education Australia, South Yarra, VIC.
Bakken, L. R., 1997. Culturable and non-culturable bacteria in soil. In van Elsas, J. D., J. T. Trevor & E. M. H. Wellington (eds), Modern Soil Microbiology. Marcel Dekker, New York: 47–61.
Bates, L., R. Waldren & I. Teare, 1973. Rapid determination of free proline for water-stress studies. Plant and Soil 39: 205–207.
Belliard, J., S. Temmerman & M. Toffolon, 2016. Ecogeomorphic relations between marsh surface elevation and vegetation properties in a temperate multi-species salt marsh. Earth Surface Processes and Landforms 42: 855–865.
Bharathkumar, S., D. Paul & S. Nair, 2008. Microbial diversity of culturable heterotrophs in the rhizosphere of salt marsh grass, Porteresia coarctata (Tateoka) in a mangrove ecosystem. Journal of Basic Microbiology 48: 10–15.
Brosnan, J. T. & J. Deputy, 2008. Seashore Paspalum. Turf Management. Cooperative Extension Service, University of Hawaii at Manoa, Manoa.
Carter, M. R. & E. G. Gregorich, 2008. Soil Sampling and Methods of Analysis. Canadian Society of Soil Science, CRC Press. Taylor & Francis Group, Oxford.
Darbre, A. & F. Norris, 1956. Vitamins in germination: determination of free and combined inositol in germinating oats. Biochemical Journal 64: 441–446.
Datta, N. P., M. S. Khera & T. R. Saini, 1962. A rapid colorimeteric procedure for the determination of the organic carbon in the soil. Journal of the Indian Society of Soil Science 10: 67–74.
Deng, S. P. & M. A. Tabatabai, 1994. Cellulase activity of soils. Soil Biology & Biochemistry 26: 1347–1354.
Donnelly, M. & L. Walters, 2014. Trapping of Rhizophora mangle propagules by coexisting early successional species. Estuaries and Coasts 37: 1562–1571.
Dorich, R. A. & D. W. Nelson, 1983. Direct colorimetric measurement of ammonium in potassium chloride extracts of soils. Soil Science Society of America Journal 47: 833–836.
DuBois, M., K. Gilles, J. Hamilton, P. Rebers & F. Smith, 1956. Colorimetric method for determination of sugars and related substances. Analytical Chemistry 28: 350–356.
Fresquez, P. & B. Sabey, 1989. Microbial community in the rhizosphere of native plant species growing on reclaimed coal mine soils varying in age. Arid Soil Research and Rehabilitation 3: 369–384.
Fresquez, P., B. Sabey & D. Klein, 1988. Soil fungal community-plant rhizosphere interactions during the early stages of ecosystem development on reclaimed coal mine soils. Journal of American Society of Mining and Reclamation 1988: 9–14.
Friess, D. A., K. W. Krauss, E. M. Horstman, T. Balke, T. J. Bouma, D. Galli & E. L. Webb, 2011. Are all intertidal wetlands naturally created equal? Bottlenecks, thresholds and knowledge gaps to mangrove and saltmarsh ecosystems. Biological Reviews 87: 346–366.
Gaitonde, M. & M. Griffiths, 1966. A spectrophotometric method for the determination of microquantities of free inositol in biological material. Analytical Biochemistry 15: 532–535.
Grieve, C. & S. Grattan, 1983. Rapid assay for determination of water soluble quaternary ammonium compounds. Plant and Soil 70: 303–307.
Huang, L., T. Baumgartl & D. Mulligan, 2012. Is rhizosphere remediation sufficient for sustainable revegetation of mine tailings? Annals of Botany 110: 223–238.
Jagtap, T., S. Bhosale & S. Charulata, 2006. Characterization of Porteresia coarctata beds along the Goa coast, India. Aquatic Botany 84: 37–44.
Kirwan, M. & J. Megonigal, 2013. Tidal wetland stability in the face of human impacts and sea-level rise. Nature 504: 53–60.
Krauss, K., K. McKee, C. Lovelock, D. Cahoon, N. Saintilan, R. Reef & L. Chen, 2013. How mangrove forests adjust to rising sea level. New Phytologist 202: 19–34.
Krishnaswamy, U., M. Muthusamy & L. Perumalsamy, 2009. Studies on the efficiency of the removal of phosphate using bacterial consortium for the biotreatment of phosphate wastewater. European Journal of Applied Sciences 1: 6–15.
Lewis, R. R. & B. Brown, 2014. Ecological Mangrove Rehabilitation Manual.
Lovelock, C. E., D. R. Cahoon, D. A. Friess, G. R. Guntenspergen, K. W. Krauss, R. Reef, K. Rogers, M. L. Saunders, F. Sidik, A. Swales, N. Saintilan, L. X. Thuyen & T. Triet, 2015. The vulnerability of Indo-Pacific mangrove forests to sea-level rise. Nature 526: 559–563.
McIntosh, J. L., 1969. Bray and Morgan soil extractants modified for testing acid soils from different parent materials. Agronomy Journal 61: 259–265.
McKee, K., J. Rooth & I. Feller, 2007. Mangrove recruitment after forest disturbance is facilitated by herbaceous species in the Caribbean. Ecological Applications 17: 1678–1693.
Mishra, R. R., M. R. Swain, T. K. Dangar & H. Thatoi, 2012. Diversity and seasonal fluctuation of predominant microbial communities in Bhitarkanika, a tropical mangrove ecosystem in India. International Journal of Tropical Biology and Conservation 60: 909–924.
Möller, I., 2006. Quantifying saltmarsh vegetation and its effect on wave height dissipation: results from a UK East coast saltmarsh. Estuarine, Coastal and Shelf Science 69: 337–351.
Moore, S. & W. H. Stein, 1948. Photometric ninhydrin method for use in the chromatography of amino acids. Biological Chemistry 176: 367–388.
Park, G., H. Oh & S. Ahn, 2009. Improvement of the ammonia analysis by the phenate method in water and wastewater. Bulletin of the Korean Chemical Society 30: 2032–2038.
Payo, A., A. Mukhopadhyay, S. Hazra, T. Ghosh, S. Ghosh, S. Brown, R. Nicholls, L. Bricheno, J. Wolf, S. Kay, A. Lázár & A. Haque, 2016. Projected changes in area of the Sundarban mangrove forest in Bangladesh due to SLR by 2100. Climatic Change 139: 279–291.
Raha, A. K., A. Mishra, S. Bhattacharya, S. Ghatak, P. Pramanick, S. Dey, I. Sarkar & C. Jha, 2014. Sea level rise and submergence of Sundarban Islands: a time series study of estuarine dynamics. Journal of Ecology and Environmental Sciences 5: 114–123.
Rashid, P. & A. Ahmed, 2011. Anatomical adaptations of Myriostachya wightiana Hook. F. to salt stress. Dhaka University Journal of Biological Sciences 20: 205–208.
Sánchez, J., 1998. Colorimetric assay of alditols in complex biological samples. Journal of Agricultural & Food Chemistry 46: 157–160.
Sharma, S., K. Nadaoka, M. Nakaoka, W. Uy, R. MacKenzie, D. Friess & M. Fortes, 2017. Growth performance and structure of a mangrove afforestation project on a former seagrass bed, Mindanao Island, Philippines. Hydrobiologia. doi:10.1007/s10750-017-3252-x.
Singh, K., V. Pandey & R. Singh, 2013. Cynodon dactylon: an efficient perennial grass to revegetate sodic lands. Ecological Engineering 54: 32–38.
Solórzano, L., 1969. Determination of ammonia in natural waters by the phenolhypochlorite method. Limnology and Oceanography 14: 799–801.
Verchot, L. V. & T. Borelli, 2005. Application of para-nitrophenol (pNP) enzyme assays in degraded tropical soils. Soil Biology & Biochemistry 37: 625–633.
Ward, R., D. Friess, R. Day & R. MacKenzie, 2016. Impacts of climate change on mangrove ecosystems: a region by region overview. Ecosystem Health and Sustainability 2(4): e01211.
Whalen, J. K. & P. R. Warman, 1996. Arylsulfatase activity in soil and soil extracts using natural and artificial substrates. Biology and Fertility of Soils 22: 373–378.
Williams, M. J., 2007. Native Plants for Coastal Restoration: What, When, and How for Florida. USDA, NRCS, Brooksville Plant Materials Center, Brooksville, FL.
Acknowledgements
We gratefully acknowledge the financial support by Department of Biotechnology, Government of India for carrying out the research work (File No.BT/PR7501/BCE/8/982/2013).The authors would like to express their sincere thanks to the anonymous reviewers and Dr. Daniel A. Friess, Editor, MMM4 Special Issue, for valuable comments made on earlier versions of the manuscript which helped us to improve it.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no competing financial interest.
Additional information
Guest editors: K. W. Krauss, I. C. Feller, D. A. Friess, R. R. Lewis III / Causes and Consequences of Mangrove Ecosystem Responses to an Ever-Changing Climate
Rights and permissions
About this article
Cite this article
Begam, M.M., Sutradhar, T., Chowdhury, R. et al. Native salt-tolerant grass species for habitat restoration, their acclimation and contribution to improving edaphic conditions: a study from a degraded mangrove in the Indian Sundarbans. Hydrobiologia 803, 373–387 (2017). https://doi.org/10.1007/s10750-017-3320-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10750-017-3320-2