Abstract
Non-native aquatic macrophytes have invaded different types of ecosystems all over the world. The exotic submersed macrophyte Hydrilla verticillata recently invaded the Paraná basin, Brazil, being recorded by the first time in the natural habitats of this river in 2005. We investigated the effects of this species on ostracod assemblages and compared the abundance, richness, and Shannon–Wiener diversity of ostracod assemblages that colonize the invading species with those that colonize Egeria najas, a native submersed species with similar architecture and physical complexity. Fragments of these two species were left for 28 days in tanks to root and grow and then they were transferred to a floodplain lake where they remained in pairs (one plant of each species; N = 7) during 30 days for colonization by ostracods. A detrented correspondence analysis was used to summarize ostracod assemblage composition. Although there were no significant differences in ostracod abundance, richness and Shannon diversity when analyzed separately, cumulative curves, which permit to eliminate effects of abundance on richness, indicated a significantly higher number of ostracod species on H. verticillata. Assemblage composition was significantly different between both plant species, as shown by the first DCA axis. Our results show that H. verticillata might provide favorable habitats for native ostracod assemblages.




Similar content being viewed by others
References
Agostinho, A. A. & M. Zalewski, 1995. The dependence of fish community structure and dynamics on floodplain and riparian ecotone zone in Paraná River, Brazil. Hydrobiologia 303: 141–148.
Alix, M. S., R. W. Scribailo & J. D. Price, 2009. Hydrilla verticillata (Hydrocharitaceae): an undesirable addition to Indiana’s aquatic flora. Rhodora 111: 131–136.
Arrington, D. A., K. O. Winemiller & C. A. Layman, 2005. Community assembly at the patch scale in a species rich tropical river. Oecologia 144: 157–167.
Barrat-Segretain, M. H., 2001. Invasive species in the Rhône River floodplain (France): replacement of Elodea canadensis Michaux by Elodea nutallii St John in two former river channels. Archiv für Hydrobiolie 152: 237–251.
Barrientos, C. A. & M. S. Allen, 2008. Fish abundance and community composition in native and non-native plants following hydrilla colonisation at Lake Izabal, Guatemala. Fisheries Management and Ecology 15: 99–106.
Colon-Gaud, J. C., W. E. Kelso & D. A. Rutherford, 2004. Spatial distribution of macroinvertebrates inhabiting hydrilla and coontais beds in the Atchafalaya Basin, Louisiana. Journal of Aquatic Plant Management 42: 85–91.
Colwell, R. K., 1997. Estimates 5: statistical estimation of species richness and shared species from samples. User Guide; 22 p, http://viceroy.eeb.uconn.edu/estima-tes.
Cook, C. D. K. & R. Lüönd, 1982. A revision of the genus Hydrilla (Hydrocharitaceae). Aquatic Botany 13: 485–504.
Cook, C. D. K. & K. Urmi-König, 1984. A revision of the genus Egeria (Hydrocharitaceae). Aquatic Botany 19: 73–96.
Creed, R. P., R. P. Cherry, J. R. Pflaum & C. J. Wood, 2009. Dominant species can produce a negative relationship between species diversity and ecosystem function. Oikos 118: 723–732.
Da Rocha, C. M. C., V. Venekey, T. N. C. Bezerra & J. R. B. Souza, 2006. Phytal marine nematode assemblages and their relation with the macrophytes structural complexity in a Brazilian tropical rocky beach. Hydrobiologia 553: 219–230.
Dibble, E. D., S. M. Thomaz & A. A. Padial, 2006. Spatial-complexity measured at a multi-scale among three aquatic plant species. Journal of Freshwater Ecology 21: 239–247.
Douglas, M. M. & R. A. O’Connor, 2003. Effects of the exotic macrophyte, para grass (Urochloa mutica), on benthic and epiphytic macroinvertebrates of a tropical floodplain. Freshwater Biology 48: 962–971.
Gotelli, N. J. & R. K. Colwell, 2001. Quantifying biodiversity: procedure and pitfalls in the measurement and comparison of species richness. Ecology Letters 4: 379–391.
Grant, I. F., E. A. Egan & M. Alexander, 1983. Measurements of rates of grazing of the ostracod Cyprinotus carolinensis on blue green algae. Hydrobiologia 106: 199–208.
Hershner, C. & K. J. Havens, 2008. Managing invasive aquatic plants in a changing system: strategic consideration of ecosystem services. Conservation Biology 22: 544–550.
Higuti, J., L. F. M. Velho, F. A. Lansac-Toha & K. Martens, 2007. Pleuston communities are buffered from regional flood pulses: the example of ostracods in the Paraná River floodplain, Brazil. Freshwater Biology 52: 1930–1943.
Higuti, J., F. A. Lansac-Tôha, L. F. M. Velho, R. L. Pinto, L. C. G. Vieira & K. Martens, 2009a. Composition and distribution of Darwinulidae (Crustacea, Ostracoda) in the alluvial valley of the upper Paraná River, Brazil. Brazilian Journal of Biology 69(2): 253–262.
Higuti, J., F. A. Lansac-Tôha, L. F. M. Velho & K. Martens, 2009b. Biodiversity of non-marine ostracods (Crustacea, Ostracoda) in the alluvial valley of the upper Paraná River, Brazil. Brazilian Journal of Biology 69 (2 Suppl.): 661–668.
Hill, M. O. & H. G. Gauch, 1980. Detrended correspondence analysis: an improved ordination technique. Vegetatio 42: 47–58.
Johnson, F. A. & F. Montalbano III, 1984. Selection of plant communities by wintering waterfowl on Lake Okeechobee, Florida. The Journal of Wildlife Management 48: 174–178.
Langeland, K. A., 1996. Hydrilla verticillata (L.f.) Royle (Hydrocharitaceae), “The Perfect Aquatic Weed”. Castanea 61: 293–304.
Levine, J. M., 2008. Biological invasions. Current Biology 18: R57–R60.
Lodge, D. M., 1986. Selective grazing on periphyton – a determinant of fresh-water gastropod microdistributions. Freshwater Biology 16: 831–841.
MacArthur, R. H. & J. W. MacArthur, 1961. On bird species diversity. Ecology 42: 594–598.
Madeira, P. T., J. A. Coetzee, T. D. Center, E. E. White & P. W. Tipping, 2007. The origin of Hydrilla verticillata recently discovered at a South African dam. Aquatic Botany 87: 176–180.
Martens, K. & F. Behen, 1994. A checklist of the non-marine ostracods (Crustacea, Ostracoda) from the inland waters of South America and adjacent islands. Travaux Scientifiques du Musée d’ Histoire Naturelle de Luxembourg 22: 1–81.
Martens, K., I. Schön, C. Meisch & D. J. Horne, 2008. Global diversity of ostracods (Ostracoda, Crustacea) in freshwater. Hydrobiologia 595: 185–193.
Mazzeo, N., L. Rodriguez-Gallego, C. Kruk, M. Meerhoff, J. Gorga, G. Lacerot, F. Guintans, M. Loureiro, D. Larrea & F. Garcia-Rodriguez, 2003. Effects of Egeria densa Planch. beds on a shallow lake without piscivores fish. Hydrobiologia 506(509): 591–602.
McCune, B. & M. J. Mefford, 1999. PC-ORD. Multivariate analysis of ecological data. Version 5.0. MjM Software, Gleneden Beach, Oregon, USA.
Mony, C., T. J. Koschnick, W. T. Haller & S. Muller, 2007. Competition between two invasive Hydrocharitaceae (Hydrilla verticillata (L.f.) Royle and Egeria densa Planch.) as influenced by sediment fertility and season. Aquatic Botany 86: 236–242.
Mormul, R. P., S. M. Thomaz, M. J. Silveira & L. Rodrigues, 2010. Epiphyton or macrophyte: which primary producer attracts the snail Hebetancylus moricandi? American Malacological Bulletin 28.
Morse, D. R., J. H. Lawton, M. M. Dodson & M. H. Williamson, 1985. Fractal dimension of vegetation and the distribution of arthropod body lengths. Nature 314: 731–733.
Oertli, B. & J. B. Lachavanne, 1995. The effects of shoot age on colonization of an emergent macrophyte (Typha latifolia) by macroinvertebrates. Freshwater Biology 34: 421–431.
Pelicice, F. M. & A. A. Agostinho, 2006. Feeding ecology of fishes associated with Egeria spp. patches in a tropical reservoir, Brazil. Ecology of Freshwater Fish 15: 10–19.
Pelicice, F. M., A. A. Agostinho & S. M. Thomaz, 2005. Fish assemblages associated with Egeria in a tropical reservoir: investigating the effects of plant biomass and diel period. Acta Oecologica 27: 9–16.
Pieterse, A. H. & K. J. Murphy, 1990. Aquatic Weeds. The Ecology and Management of Nuisance Aquatic Vegetation. Oxford Science Publications, Oxford.
Raizer, J. & M. E. C. Amaral, 2001. Does the structural complexity of aquatic macrophytes explain the diversity of associated spider assemblages. Journal of Arachnology 29: 227–237.
Roca, J. R. & D. L. Danielopol, 1991. Exploration of interstitial habitats by the phytophylous ostracods Cypridopsis vidua (O.F. Muller): experimental evidence. Annales de Limnologie 27: 243–252.
Rommens, W., J. Maes, N. Dekeza, P. Inghelbrecht, T. Nhiwatiwa, E. Holsters, F. Ollevier, B. Marshall & L. Brendonck, 2003. The impact of water hyacinth (Eichhornia crassipes) in a eutrophic subtropical (Lake Chivero, Zimbabwe). I. Water quality. Archiv für Hydrobiologie 158: 373–388.
Schmit, O., G. Rossetti, J. Vandekerkhove & F. Mezquita, 2007. Food selection in Eucypris virens (Crustacea, Ostracoda) under experimental conditions. Hydrobiologia 585: 135–140.
Sousa, W. T. Z., S. M. Thomaz, K. J. Murphy, M. J. Silveira & R. P. Mormul, 2009. Environmental predictors of the occurrence of exotic Hydrilla verticillata (L.f.) Royle and native Egeria najas Planch. in a sub-tropical river floodplain: the Upper River Paraná, Brazil. Hydrobiologia 632: 65–78.
StatSoft, Inc., 2005. STATISTICA (data analysis software system), version 7.
Taniguchi, H. & M. Tokeshi, 2004. Effects of habitat complexity on benthic assemblages in a variable environment. Freshwater Biology 49: 1164–1178.
Taniguchi, H., S. Takano & M. Tokeshi, 2003. Influences of habitat complexity on the diversity and abundance of epiphytic invertebrates on plants. Freshwater Biology 48: 718–728.
Theel, H. J., E. D. Dibble & J. D. Madsen, 2008. Differential influence of a monotypic and diverse native aquatic plant bed on a macroinvertebrate assemblage; an experimental implication of exotic plant induced habitat. Hydrobiologia 600: 77–87.
Thomaz, S. M., E. D. Dibble, L. R. Evangelista, J. Higuti & L. M. Bini, 2008. Influence of aquatic macrophyte habitat complexity on invertebrate abundance and richness in tropical lagoons. Freshwater Biology 53: 358–367.
Thomaz, S. M., P. Carvalho, A. A. Padial & J. T. Kobayashi, 2009. Temporal and spatial patterns of aquatic macrophyte diversity in the Upper Paraná River floodplain. Brazilian Journal of Biology 69: 617–625.
Vieira, L. C. G., L. M. Bini, L. F. M. Velho & G. R. Mazão, 2007. Influence of spatial complexity on the density of periphytic rotifers, microcrustaceans and testate amoebae. Fundamental and Applied Limnology 170: 77–85.
Wang, J. W., D. Yu, W. Xiong & Y. Q. Han, 2008. Above- and belowground competition between two submersed macrophytes. Hydrobiologia 607: 113–122.
Wilson, J. W. & A. Ricciardi, 2009. Epiphytic macroinvertebrate asssemblages on Eurasian watermilfoid (Myriophyllum spicatum) and native milfoils Myriophyllum sibericum and Myriophyllum alterniflorum in eastern North America. Canadian Journal of Fisheries and Aquatic Sciences 66: 18–30.
Acknowledgments
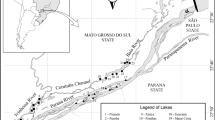
We acknowledge the helpful comments of two reviewers on earlier versions of the manuscript. Figure 1 was kindly furnished by Eduardo Ribeiro da Cunha (Universidade Estadual de Maringá, Brazil). This study was funded by Brazilian Council of Research (CNPq), through a Long Term Ecological Program (site number 6). S. M. Thomaz received a CNPq Productivity Researcher Grant and acknowledges this agency for long-term funding. R. P. Mormul acknowledges the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES) for furnishing a PhD fellowship. We also thank CAPES for funding K. Martens during his trip to Brazil.
Author information
Authors and Affiliations
Corresponding author
Additional information
Handling editor: B. Oertli
Rights and permissions
About this article
Cite this article
Mormul, R.P., Thomaz, S.M., Higuti, J. et al. Ostracod (Crustacea) colonization of a native and a non-native macrophyte species of Hydrocharitaceae in the Upper Paraná floodplain (Brazil): an experimental evaluation. Hydrobiologia 644, 185–193 (2010). https://doi.org/10.1007/s10750-010-0112-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10750-010-0112-3