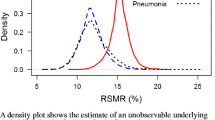
Abstract
Black patients are less likely to receive certain surgical interventions. To test whether a health risk disparity and thus differential appropriateness for surgery explains a treatment disparity, researchers must adjust observed rates for patient-level health differences using valid contextual regression controls to increase patient comparability. As an alternative to the standard health adjustment with predetermined diagnosis groups, I propose a machine learning-based method that better captures clinical practices to adjust for the important predictors of invasive surgery applied to the context of acute myocardial infarction (AMI). With data from the Nationwide Inpatient Sample, this method decreases the standard adjusted AMI surgery disparity by 45–55%. Nonetheless, a significant surgery disparity of 5.9 percentage points with hospital fixed effects and 4.5 percentage points with physician fixed effects remains after adjusting for predictive controls. The smaller yet persistent disparity provides evidence of differential AMI treatment beyond that explained by health risk differences.


Similar content being viewed by others
Notes
I follow the Office of Management and Budget and the Institute of Medicine and define Black race as“a person having origins in any of the Black racial groups of Africa” but also recognize the considerable criticism of racial classifications and the arguable lack of biological and genetic bases beyond their social construction (Jones 2001; Kaufman and Cooper 2001; Williams and Sternthal 2010; The Office of Management and Budget 2016; Sen and Wasow 2016; Yudell et al. 2016). I also follow the Institute of Medicine and the literature and use the term “Black” and “African American” interchangeably to refer to patient race as reported in the Nationwide Inpatient Sample while also noting the difficulty of recording information on race in the clinical record discussed further in Sect. 3 (Nelson and Smedley 2003; The Office of Management and Budget 2016).
In this paper, I use “risk” in terms of the underlying chance of a given treatment and not risk of mortality or complication.
Community hospitals, as defined by American Hospital Association, include non-federal, short-term, general and other specialty hospitals and exclude federal hospitals, long-term hospitals, psychiatric hospitals, alcohol/chemical dependency treatment facilities, and hospitals units within institutions such as prisons (Health Forum LLC 2018).
394,857 observations dropped due to missing data with the race variable as an important component assumed to be missing at random for purposes of this study. See Houchens (2015) for a more thorough discussion of race and missingness in the NIS.
States that do not supply physician identifiers are AK, CA, CT, HI, IL, IN, LA, MA, MS, NC, OK, UT, VT, WI.
NIS contains up to 25 diagnoses codes per patient (15 prior to the 2009 NIS) but the maximum number of diagnoses varies by state. The mean number of diagnoses per patient in the AMI sample is 10.
A base surgery rate of 36%, near the middle of the distribution, suggests that a linear approximation yields the same conclusions as a conditional logistic regression. I ran logistic regressions on subsets of the data which confirmed this.
HCUP excludes cardiac arrhythmia from the comorbidity software following concerns about its reliability as a comorbidity (Thompson et al. 2015).
Full code list available in Appendix.
References
Agarwal, S., Tuzcu, E.M., Kapadia, S.R.: Choice and selection of treatment modalities for cardiac patients: an interventional cardiology perspective. J. Am. Heart Assoc. (2015). https://doi.org/10.1161/JAHA.115.002353
Alsan, M., Garrick, O., Graziani, G.: Does diversity matter for health? Experimental evidence from Oakland. Am. Econ. Rev. 109(12), 4071–4111 (2019). https://doi.org/10.1257/aer.20181446
Angrist, J.D., Pischke, J.-S.: Mostly Harmless Econometrics: An Empiricist’s Companion, p. 373. Princeton University Press, Princeton (2009). isbn: 9781400829828
Apostolakis, E.E., et al.: Left ventricular diastolic dysfunction of the cardiac surgery patient; a point of view for the cardiac surgeon and cardio-anesthesiologist. J. Cardiothor. Surg. 4, 67 (2009). https://doi.org/10.1186/1749-8090-4-67
Arrow, K.J., Arrow, K.J.: The theory of discrimination. In: In Discrimination in Labor Markets, pp. 3–33 (1973). https://doi.org/10.1.1.657.5158. http://citeseerx.ist.psu.edu/viewdoc/summary?
Association of American Medical Colleges Diversity in Medicine: Facts and Figures 2019. Tech. rep. Association of American Medical Colleges (2019) https://www.aamc.org/data-reports/workforce/interactive-data/figure-18-percentage-all-active-physicians-race/ethnicity-2018
Balsa, A.I., McGuire, T.G.: Statistical discrimination in health care. J. Health Econ. 20(6), 881–907 (2001)
Balsa, A.I., McGuire, T.G., Meredith, L.S.: Testing for statistical discrimination in health care. Health Serv. Res. 40(1), 227–252 (2005). https://doi.org/10.1111/j.1475-6773.2005.00351.x
Balsa, A.I., et al.: Clinical uncertainty and healthcare disparities. Am. J. Law Med. 29, 203 (2003)
Barnato, A.E., et al.: Hospital-level racial disparities in acute myocardial infarction treatment and outcomes. Med. Care 43(4), 308–19 (2005)
Becker, G.S.: The Economics of Discrimination. University of Chicago press, Chicago (1957)
Belloni, A., Chernozhukov, V., Hansen, C.: Inference on treatment effects after selection among high-dimensional controls. Rev. Econ. Stud. 81(2), 608–650 (2013). https://doi.org/10.1093/restud/rdt044. arXiv:1201.0224v3
Bertrand, M., Mullainathan, S.: Are Emily and Greg more employable than Lakisha and Jamal? A field experiment on labor market discrimination. Am. Econ. Rev. 94(4), 991–1013 (2004). https://doi.org/10.1257/0002828042002561
Bilheimer, L.T., Klein, R.J.: Data and measurement issues in the analysis of health disparities (2010). https://doi.org/10.1111/j.1475-6773.2010.01143.x. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2965888/pmc/articles/PMC2965888/?report=abstract
Bradley, E.H., et al.: Racial and ethnic differences in time to acute reperfusion therapy for patients hospitalized with myocardial infarction. JAMA 292(13), 1563 (2004). https://doi.org/10.1001/jama.292.13.1563
Bravata, D.M., et al.: Systematic review: the comparative effectiveness of percutaneous coronary interventions and coronary artery bypass graft surgery. Ann. Intern. Med. 147(10), 703 (2007). https://doi.org/10.7326/0003-4819-147-10-200711200-00185
Breiman, L.: Classification and Regression Trees, p. 358. Chapman & Hall, London (1993). isbn: 0412048418
Buchmueller, T.C., et al.: Effect of the affordable care act on racial and ethnic disparities in health insurance coverage. Am. J. Public Health 106(8), 1416–1421 (2016). https://doi.org/10.2105/AJPH.2016.303155
Card, D., Dobkin, C., Maestas, N.: Does medicare save lives? Q. J. Econ. 124(2), 597–636 (2009). https://doi.org/10.1162/qjec.2009.124.2.597
Chandra, A., Khullar, D., Lee, T.H.: Addressing the challenge of gray-zone medicine. N. Engl. J. Med. 372(3), 203–205 (2015). https://doi.org/10.1056/NEJMp1409696
Chapman, E.N., Kaatz, A., Carnes, M.: Physicians and implicit bias: how doctors may unwittingly perpetuate health care disparities. J. Gen. Intern. Med. 28(11), 1504–1510 (2013). https://doi.org/10.1007/s11606-013-2441-1
Charles, K.K., Hurst, E., Roussanov, N.: Conspicuous consumption and race. Q. J. Econ. 124(2), 425–467 (2009). https://doi.org/10.1162/qjec.2009.124.2.425
Charlson, M.E., et al.: A new method of classifying prognostic comorbidity in longitudinal studies: development and validation. J. Chronic Dis. 40(5), 373–83 (1987)
Chen, T., Guestrin, C.: XGBoost: a scalable tree boosting system. In: Proceedings of the ACM SIGKDD International Conference on Knowledge Discovery and Data Mining, Vol. 13-17-Augu. Association for Computing Machinery, pp. 785–794. isbn: 9781450342322. (2016). https://doi.org/10.1145/2939672.2939785. arXiv: 1603.02754
Cooper, L.T.: Myocarditis. N. Engl. J. Med. 360(15), 1526–1538 (2009). https://doi.org/10.1056/NEJMra0800028
Cooper-Patrick, L.: Race, gender, and partnership in the patient–physician relationship. JAMA 282(6), 583 (1999). https://doi.org/10.1001/jama.282.6.583
Cram, P., et al.: Racial disparities in revascularization rates among patients with similar insurance coverage. J. Natl. Med. Assoc. 101(11), 1132–1139 (2009)
Currie, J., MacLeod, W.B., Van Parys, J.: Provider practice style and patient health outcomes: the case of heart attacks. J. Health Econ. 47, 64–80 (2016). https://doi.org/10.1016/j.jhealeco.2016.01.013
Dracup, K., et al.: The physician’s role in minimizing prehospital delay in patients at high risk for acute myocardial infarction: recommendations from the national heart attack alert program. Ann. Intern. Med. 126(8), 645 (1997). https://doi.org/10.7326/0003-4819-126-8-199704150-00010
Dyke, C.M., et al.: Systolic and diastolic blood pressure variability as risk factors for adverse events after coronary artery bypass grafting (2019). https://doi.org/10.1001/jamasurg.2018.3233
Elixhauser, A., et al.: Comorbidity measures for use with administrative data. Med. Care 36(1), 8–27 (1998)
Ellis, S.G., et al.: In-hospital cardiac mortality after acute closure after coronary angioplasty: analysis of risk factors from 8,207 procedures. J. Am. Coll. Cardiol. 11(2), 211–216 (1988). https://doi.org/10.1016/0735-1097(88)90082-4
Elwert, F.: Graphical Causal Models, pp. 245–273. Springer, Dordrecht (2013). https://doi.org/10.1007/978-94-007-6094-3_13
Ewens, M., Tomlin, B., Wang, L.C.: Statistical discrimination or prejudice? A large sample field experiment. Rev. Econ. Stat. 96(1), 119–134 (2014). https://doi.org/10.1162/REST_a_00365
Fischman, D.L., et al.: A randomized comparison of Coronary–Stent placement and balloon angioplasty in the treatment of coronary artery disease. N. Engl. J. Med. 331(8), 496–501 (1994). https://doi.org/10.1056/NEJM199408253310802
Ford, E., Newman, J., Deosaransingh, K.: Racial and ethnic differences in the use of cardiovascular procedures: findings from the California Cooperative Cardiovascular project. Am. J. Public Health 90(7), 1128–34 (2000)
Frank, I.E., Friedman, J.H.: A statistical view of some chemometrics regression tools. Technometrics 35(2), 109 (1993). https://doi.org/10.2307/1269656
Friedman, J., Hastie, T., Tibshirani, R.: Regularization paths for generalized linear models via coordinate descent. J. Stat. Softw. 33(1), 1–22 (2010). https://doi.org/10.18637/jss.v033.i01
Friedman, J.H.: Greedy function approximation: a gradient boosting machine. Ann. Stat. 29(5), 1189–1232 (2001). https://doi.org/10.1214/aos/1013203451
Gaskin, D.J., et al.: Do hospitals provide lower-quality care to minorities than to whites? Health Affairs (Project Hope) 27(2), 518–27 (2008). https://doi.org/10.1377/hlthaff.27.2.518
Gelbach, J.B.: When do covariates matter? and which ones, and how much? J. Labor Econ. 34(2), 509–543 (2016). https://doi.org/10.1086/683668
Geronimus, A.T.: The weathering hypothesis and the health of African-American women and infants: evidence and speculations (1992)
Goff, D.C., et al.: Knowledge of heart attack symptoms in a population survey in the United States: the react trial. Arch. Intern. Med. 158(21), 2329–2338 (1998)
Gordon, H.S., Paterniti, D.A., Wray, N.P.: Race and patient refusal of invasive cardiac procedures (2004). https://doi.org/10.1111/j.1525-1497.2004.30131.x
Graham, G.: Racial and ethnic differences in acute coronary syndrome and myocardial infarction within the United States: from demographics to outcomes. Clin. Cardiol. 39(5), 299–306 (2016). https://doi.org/10.1002/clc.22524
Green, A.R., et al.: Implicit bias among physicians and its prediction of thrombolysis decisions for black and white patients. J. Gen. Intern. Med. 22(9), 1231–8 (2007). https://doi.org/10.1007/s11606-007-0258-5
Greene, W.: The behaviour of the maximum likelihood estimator of limited dependent variable models in the presence of fixed effects. Econom. J. 7(1), 98–119 (2004). https://doi.org/10.1111/j.1368-423x.2004.00123.x
Groeneveld, P.W., Heidenreich, P.A., Garber, A.M.: Racial disparity in cardiac procedures and mortality among long-term survivors of cardiac arrest. Circulation 108(3), 286–291 (2003)
Hanlon, C., et al.: Maintain and expand the Healthcare Cost and Utilization Project (HCUP) state documentation of racial and ethnic health disparities to inform strategic action. Technical report (2011). http://www.hcup-us.ahrq.gov/reports.jsp
Hansson, G.K.: Inflammation, atherosclerosis, and coronary artery disease. N. Engl. J. Med. 352(16), 1685–1695 (2005). https://doi.org/10.1056/NEJMra043430
Health Forum LLC: Fast Facts on U.S. Hospitals. (2018). https://www.aha.org/statistics/fast-facts-us-hospitals. Accessed 22 May 2018
Healthcare Cost and Utilization Project (HCUP): HCUP Central Distributor SID File Composition (2017). https://www.hcup-us.ahrq.gov/db/state/siddist/siddist_hospital.jsp. Accessed 22 May 2018
Hernán, M.A., et al.: Causal knowledge as a prerequisite for confounding evaluation: an application to birth defects epidemiology. Am. J. Epidemiol. 155(2), 176–184 (2002). https://doi.org/10.1093/aje/155.2.176
Houchens, R.: Missing data methods for the NIS and the SID. HCUP Methods Series Report (2015)
Hougland, P., et al.: Using ICD-9-CM codes in hospital claims data to detect adverse events in patient safety surveillance. Agency for Healthcare Research and Quality (2008). http://www.ncbi.nlm.nih.gov/pubmed/21249878
Jones, C.P.: Invited Commentary: “Race”, racism, and the practice of epidemiology. Am. J. Epidemiol. 154(4), 299–304 (2001). https://doi.org/10.1093/aje/154.4.299
Kaufman, J.S., Cooper, R.S.: Commentary: Considerations for use of racial/ethnic classification in etiologic research. Am. J. Epidemiol. 154(4), 291–298 (2001). https://doi.org/10.1093/aje/154.4.291
Keller, K.B., Lemberg, L.: Prinzmetal’s angina. Am. J. Crit. Care 13(4), 350–4 (2004)
Koulayev, S., Simeonova, E., Skipper, N.: Can physicians affect patient adherence with medication? Health Econ. 26(6), 779–794 (2017). https://doi.org/10.1002/hec.3357
Kressin, N.R., Petersen, L.A.: Racial differences in the use of invasive cardiovascular procedures: review of the literature and prescription for future research. Ann. Intern. Med. 135(5), 352–66 (2001)
Krstajic, D., et al.: Cross-validation pitfalls when selecting and assessing regression and classification models. J. Cheminform. 6(1), 10 (2014). https://doi.org/10.1186/1758-2946-6-10
LaPar, D.J., et al.: Primary payer status affects outcomes for cardiac valve operations. J. Am. Coll. Surg. 212(5), 759–767 (2011). https://doi.org/10.1016/j.jamcollsurg.2010.12.050
Lipton, B.J., Decker, S.L., Sommers, B.D.: The affordable care act appears to have narrowed racial and ethnic disparities in insurance coverage and access to care among young adults. Med. Care Res. Rev. 76(1), 32–55 (2019). https://doi.org/10.1177/1077558717706575
McGuire, T.G., et al.: Testing for statistical discrimination by race/ethnicity in panel data for depression treatment in primary care. Health Serv. Res. 43(2), 531–551 (2008). https://doi.org/10.1111/j.1475-6773.2007.00770.x
Morrison, D.A., et al.: Percutaneous coronary intervention versus coronary artery bypass graft surgery for patients with medically refractory myocardial ischemia and risk factors for adverse outcomes with bypass: a multicenter, randomized trial. J. Am. Coll. Cardiol. 38(1), 143–149 (2001). https://doi.org/10.1016/S0735-1097(01)01366-3
Nelson, A., Smedley, B. (eds.): Unequal Treatment. National Academies Press, Washington, D.C. (2003). isbn: 978-0-309-08532-8. https://doi.org/10.17226/10260
Oakley, C.M.: Myocarditis, pericarditis and other pericardial diseases. Heart 84(4), 449–454 (2000). https://doi.org/10.1136/heart.84.4.449
Okelo, S., et al.: Race and the decision to refer for coronary revascularization: the effect of physician awareness of patient ethnicity. J. Am. Coll. Cardiol. 38(3), 698–704 (2001). https://doi.org/10.1016/S0735-1097(01)01418-8
Ornato, J.P., Hand, M.M.: Warning signs of a heart attack. Circulation 104(11), 1212–3 (2001). https://doi.org/10.1161/HC2501.093258
Pearl, J.: Causal inference in statistics: an overview. Stat. Surv. 3, 96–146 (2009). https://doi.org/10.1214/09-SS057
Phelps, E.S.: The statistical theory of racism and sexism. Am. Econ. Rev. 62(4), 659–661 (1972). https://doi.org/10.2307/1806107
Popescu, I., Cram, P., Vaughan-Sarrazin, M.S.: Differences in admitting hospital characteristics for black and white medicare beneficiaries with acute myocardial infarction. Circulation 123(23), 2710–2716 (2011). https://doi.org/10.1161/CIRCULATIONAHA.110.973628
Prasad, A., Lerman, A., Rihal, C.S.: Apical ballooning syndrome (Tako-Tsubo or stress cardiomyopathy): a mimic of acute myocardial infarction. Am. Heart J. 155(3), 408–417 (2008). https://doi.org/10.1016/j.ahj.2007.11.008
Prins, C., et al.: Cardiac surgery risk-stratification models. Cardiovasc. J. Afr. 23(3), 160–4 (2012). https://doi.org/10.5830/CVJA-2011-047
Quan, H., et al.: Coding algorithms for defining comorbidities in ICD-9-CM and ICD-10 administrative data. Med. Care 43(11), 1130–9 (2005)
Rivera, C., María, A., Ruiz-Bailén, M., Aguilar, L.R.: Takotsubo cardiomyopathy—a clinical review. Med. Sci. Moni. Int. Med. J. Exp. Clin. Res. 17(6), RA135–RA147 (2011). https://doi.org/10.12659/msm.881800
Radovanovic, D., et al.: Validity of Charlson Comorbidity Index in patients hospitalised with acute coronary syndrome. Insights from the nationwide AMIS Plus registry 2002–2012. Heart 100(4), 288–294 (2014). https://doi.org/10.1136/heartjnl-2013-304588
Rodriguez, F., et al.: Young hispanic women experience higher in-hospital mortality following an acute myocardial infarction. J. Am. Heart Assoc. 4(9), e002089 (2015). https://doi.org/10.1161/JAHA.115.002089
Romano, P., Hussey, P., Ritley, D.: Selecting quality and resource use measures: a decision guide for community quality collaboratives. Technical report (2010). www.ahrq.gov
Romano, P.S., et al.: Validity of selected AHRQ patient safety indicators based on VA national surgical quality improvement program data. Health Serv. Res. 44(1), 182–204 (2009). https://doi.org/10.1111/j.1475-6773.2008.00905.x
Ruppert, D.: The elements of statistical learning: data mining, inference, and prediction. J. Am. Stat. Assoc. 99(466), 567–567 (2004). https://doi.org/10.1198/jasa.2004.s339
Sen, M., Wasow, O.: Race as a bundle of sticks: designs that estimate effects of seemingly immutable characteristics. Annu. Rev. Polit. Sci. 19(1), 499–522 (2016). https://doi.org/10.1146/annurev-polisci-032015-010015
Shahian, D.M., et al.: The Society of Thoracic Surgeons 2008 cardiac surgery risk models: Part 1|Coronary artery bypass grafting surgery. Ann. Thorac. Surg. 88(1), S2–S22 (2009). https://doi.org/10.1016/j.athoracsur.2009.05.053
Simeonova, E.: Race, quality of care and patient outcomes: what can we learn from the department of veterans affairs? Atlant. Econ. J. 37(3), 279–298 (2009). https://doi.org/10.1007/s11293-009-9184-8
Simeonova, E.: Doctors, patients and the racial mortality gap. J. Health Econ. 32(5), 895–908 (2013). https://doi.org/10.1016/j.jhealeco.2013.07.002
Singh, J.A., et al.: Trends in and disparities for acute myocardial infarction: an analysis of Medicare claims data from 1992 to 2010. BMC Med. 12, 190 (2014). https://doi.org/10.1186/s12916-014-0190-6
Skinner, J., et al.: Mortality after acute myocardial infarction in hospitals that disproportionately treat black patients. Circulation 112(17), 2634–2641 (2005). https://doi.org/10.1161/CIRCULATIONAHA.105.543231
Stammann, A., Heiss, F., Mcfadden, D.: Fixed effects logit models with large panel data estimation of fixed EECTS logit models with large panel data. Econstor (2016). http://hdl.handle.net/10419/145837www.econstor.eu
Tariq, A.R., Mantha, A.: P492Impact of BMI on clinical outcomes and readmissions after cardiac catheterization in the USA. Eur. Heart J. (2017). https://doi.org/10.1093/eurheartj/ehx501.P492
The Office of Management and Budget: Standards for the classification of federal data on race and ethnicity. Technical report (2016). https://www.federalregister.gov/documents/2016/09/30/2016-23672/standards-for-maintaining-collecting-and-presenting-federal-data-on-race-and-ethnicity
Thompson, N.R., et al.: A new Elixhauser-based comorbidity summary measure to predict in-hospital mortality. Med. Care 53(4), 374–9 (2015). https://doi.org/10.1097/MLR.0000000000000326
Tibshirani, R.: Regression shrinkage and selection via the Lasso (1996). https://doi.org/10.2307/2346178
Tversky, A., Kahneman, D.: Judgment under uncertainty: heuristics and biases. Science (New York, N.Y.) 185(4157), 1124–31 (1974). https://doi.org/10.1126/science.185.4157.1124
Ugolini, C., Nobilio, L.: Risk adjustment for coronary artery bypass graft surgery: an administrative approach versus EuroSCORE. Int. J. Qual. Health Care 16(2), 157–164 (2004). https://doi.org/10.1093/intqhc/mzh016
United States Census Bureau: QuickFacts: United States. Technical report (2019). https://www.census.gov/quickfacts/fact/table/US/PST045219. https://www.census.gov/quickfacts/fact/table/US/IPE120218%0A. https://www.census.gov/quickfacts/fact/table/US/PST045216
Utah Department of Health: Adverse Events Related to Medical Care (2001). http://stats.health.utah.gov/wp-content/uploads/2016/11/adverse_events.pdf. Accessed 09 Aug 2017
Uva, M.S., et al.: Cardiac surgery and morbid obesity. Portuguese J. Cardiol. 21(3), 255–64 (2002). discussion 267–9
VanderWeele, T.J., Hernán, M.A.: Causal effects and natural laws: towards a conceptualization of causal counterfactuals for nonmanipulable exposures, with application to the effects of race and sex. In: Causality: Wiley Series in Probability and Statistics. Wiley Blackwell, pp. 101–113. (2012). https://doi.org/10.1002/9781119945710.ch9
Weinstein, J.N., et al.: Communities in action: pathways to health equity, CH. 2. National Academies Press, Washington, DC (2017). https://doi.org/10.17226/24624. isbn: 9780309452991
Williams, D.R., Mohammed, S.A.: Discrimination and racial disparities in health: evidence and needed research. J. Behav. Med. 32(1), 20–47 (2009). https://doi.org/10.1007/s10865-008-9185-0
Williams, D.R., Sternthal, M.: Understanding racial-ethnic disparities in health: sociological contributions. J. Health Soc. Behav. 51(1 suppl), S15–S27 (2010). https://doi.org/10.1177/0022146510383838
Williams, D.R., et al.: Moving upstream: how interventions that address the social determinants of health can improve health and reduce disparities. J. Public Health Manag. Pract. (2008). https://doi.org/10.1097/01.PHH.0000338382.36695.42
Xu, Z., et al.: Gradient boosted feature selection. In: Proceedings of the 20th ACM SIGKDD International Conference on Knowledge Discovery and Data Mining, pp. 522–531 (2014). https://doi.org/10.1145/2623330.2623635
Yancy, C.W., et al.: 2017 ACC/AHA/HFSA focused update of the 2013 ACCF/AHA guideline for the management of heart failure: a report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines and the Heart Failure Society of Amer. J. Cardiac Fail. 23(8), 628–651 (2017). https://doi.org/10.1016/j.cardfail.2017.04.014
Yudell, M., et al.: Taking race out of human genetics. Science 351(6273), 564–565 (2016). https://doi.org/10.1126/science.aac4951
Zestcott, C.A., Blair, I.V., Stone, J.: Examining the presence, consequences, and reduction of implicit bias in health care: a narrative review. Group Process. Intergroup Relat. 19(4), 528–542 (2016a). https://doi.org/10.1177/1368430216642029
Zestcott, C.A., Blair, I.V., Stone, J.: Examining the presence, consequences, and reduction of implicit bias in health care: a narrative review. Group Process. Intergroup Relat. (2016b). https://doi.org/10.1177/1368430216642029
Acknowledgements
I would like to thank Indiana University professors Seth Freedman, Kosali Simon, Coady Wing, Predrag Radivojac, Brad Heim, Sergio Fernandez, Alex Hollingsworth, Justin Ross, Evan Ringquist, Dan Sacks, participants at the APPAM Fall Research Conference, Midwestern Economics Association Annual Meeting, ASHEcon Biennial Conference. A special thank you to Sean Mooney and the Mooney Lab, Anirban Basu, and participants of the PHEnOM series at the University of Washington. Thank you to the National Library of Medicine research training grant (T15-LM007442-19) and PI Peter Tarczy-Hornoch. I also thank the Agency for Healthcare Research and Quality and its HCUP data partners (https://www.hcup-us.ahrq.gov/db/hcupdatapartners.jsp). All Errors are my own.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Appendices
Appendix 1: Cross-validation
Cross-validation repeatedly builds and evaluates a given model on different exclusive samples of the data, called folds, to estimate the model’s out-of-sample predictive accuracy (Ruppert 2004). K-folds cross-validation denotes the number of folds and repetitions of this cross-validation process as K. Figure 3 illustrates a threefold cross-validation process, where the entire dataset is cut into three folds. In round 1, folds 2 and 3 build the prediction model. The predictions are then tested for their accuracy on fold 1. 3-fold cross validation repeats the process for 3 total rounds until each fold is the hold-out testing sample. The average predictive accuracy for all rounds is an estimate of an overall accuracy. To evaluate my predictions, I use the standard 10-fold cross-validation, which is ten folds of the data and ten total rounds of prediction accuracy evaluation.
Appendix 2: Post-double selection
As before, to find important predictors of surgery, I first regress invasive surgery on I, a vector of all potential candidate variables to be selected for risk-adjustment.
This step models important confounding variables in my model of interest (Eq. 1), to keep the residual variance small.
I next regress my independent variable of interest, an indicator for Black race of the patient, on the same vector of potential candidate variables, I. This step models variables that are strongly related to the Black race and thus potentially important confounding factors. Finding potential confounding variables for both the dependent and independent variables of interest lowers the chances of omitting important explanatory variables.
I then estimate my model of interest (Eq. A1) as a linear probability model including variables important in either the invasive surgery (\(R_{S}\)) or the Black indicator (\(R_{B}\)) prediction steps, which when combined becomes (\(R_S \cup R_B \)), as equation 16.
The inclusion of predictors of both race and invasive treatment in the linear regression model is an application of the post-double-selection method. The method allows for valid inference from my standard model of interest by controlling for additional potential confounders of race (Belloni et al. 2013). Table 9 presents variables chosen for the prediction of Black race.
Appendix 3: Important predictors of black race
Table 9 presents variables chosen for the prediction of Black race. Black patients are more likely to be diagnosed with Sarcoidosis, cocaine abusers, and, Sickle-cell. Some of these variables may be mechanisms of discrimination rather than surgery confounders. For example, are Black patients much more likely to be cocaine abusers or simply more likely to be coded as such? Similar concerns about potential bias in the recording of these variables make including the predictors of Black race as controls in the main specification problematic. The lasso model, though, has a high AUC of .98 on an independent holdout sample selecting 98 variables.
Appendix 4: Boosted trees
Boosted versions of tree-based models build many trees with a small number of selected variables in a stage-wise fashion and aggregate the many models into a final “boosted” model that performs more accurate predictions than any of its parts (Friedman 2001). Boosted trees minimize a prediction error function called a loss function. For this paper, I implement boosted trees using the XGBoost gradient boosted tree algorithm from the XGBoost package in R (Chen and Guestrin 2016). XGBoost has a few main benefits for dealing with clinical data. For missing clinical data, XGBoost will automatically learn the best surgery/non-surgery split based on the outcomes of this loss function. In other words, XGBoost finds the best imputed response for missing values based on reduced predictive accuracy loss during the model building phase. Therefore, if there is a prediction signal in the distribution of missing data, it is learned and fit by the model. The algorithm also averages the predictive gain from each variable in each iteration to give an overall predictive importance of each variable in the final model. These feature rankings can be used to select features but does not explicitly exclude variables like lasso (Ruppert 2004). Gradient Boosted Feature Selection changes the penalty function to explicitly drop unimportant variables, like lasso does, to more explicitly meet the sparsity assumption required by the post-double-selection method (Belloni et al. 2013; Xu et al. 2014). However, variable selection through an importance threshold leads to sparsity and very similar chosen variables in this case.
Boosted tree variable selection gives consistent results in terms of variables selected when compared to the main method of lasso. All selection algorithms, lasso and the tree based methods, choose the many of the same important predictors with the tree based models again choosing a subset of the logistic lasso model selected variables, showing the consistency of machine learning variable selection. The order of the variable importance changes, but 95 of the top 100 variables remain the same.
Appendix 5: Disparity variation through time
Table 10 presents the main hospital fixed effects specification with lasso chosen adjusters and either a linear time variable interacted with the Black race indicator variable (column 1) or the Black race indicator interacted with a indicator for each year of the data (column 2). For column 2, the base year is 2003. The results suggest that the disparity decreases through time since the positive coefficient on Black\(*\)Linear moves the disparity estimate closer to zero. If the analysis is broken down to each year interacted with the Black race indicator, the findings in column 2 indicate the only individually significantly different year is 2010. This finding suggests that, in 2010, the disparity was significantly decreased as the positive coefficient moves the disparity estimate towards zero.
Appendix 6: All chosen predictors of surgery


Rights and permissions
About this article
Cite this article
Hammarlund, N. Racial treatment disparities after machine learning surgical risk-adjustment. Health Serv Outcomes Res Method 21, 248–286 (2021). https://doi.org/10.1007/s10742-020-00231-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10742-020-00231-7