Abstract
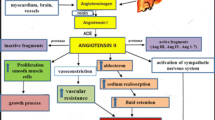
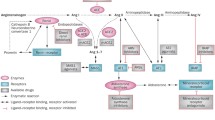
Blockade of the renin–angiotensin–aldosterone system (RAAS) with ACE inhibitors has been a cornerstone of heart failure therapy for over 15 years. More recently, further blockade of RAAS with aldosterone antagonists and angiotensin receptor blockers (ARBs) has been studied. While these therapies have certainly improved outcomes in the treatment of heart failure, morbidity and mortality remain extremely high. Furthermore, polypharmacy and complex regimens of seven medications on average is the norm for management of heart failure. This results in increased costs, patient burden, and uncertainty as to the best course of therapy. The ability to personalize patients’ therapeutic regimens using pharmacogenomics has the potential of providing more effective and efficient use of RAAS-modulating medications. This review highlights the implications of major RAAS pharmacogenetic studies, while outlining future directions for translation to practice.



Similar content being viewed by others
References
Hospitalization in the United States (1997) HCUP Fact Book No 1. AHRQ Publication No. 00-0031, May 2000. Agency for Healthcare Research and Quality, Rockville, MD. http://www.ahrq.gov/data/hcup/factbk1
Cohn JN, Tognoni G (2001) A randomized trial of the angiotensin-receptor blocker valsartan in chronic heart failure. N Engl J Med 345(23):1667–1675
McMurray JJ, Ostergren J, Swedberg K, Granger CB, Held P, Michelson EL et al (2003) Effects of candesartan in patients with chronic heart failure and reduced left-ventricular systolic function taking angiotensin-converting-enzyme inhibitors: the CHARM-Added trial. Lancet 362(9386):767–771
Pfeffer MA, McMurray JJ, Velazquez EJ, Rouleau JL, Kober L, Maggioni AP et al (2003) Valsartan, captopril, or both in myocardial infarction complicated by heart failure, left ventricular dysfunction, or both. N Engl J Med 349(20):1893–1906
Zineh I, Johnson JA (2006) Pharmacogenetics of chronic cardiovascular drugs: applications and implications. Expert Opin Pharmacother 7(11):1417–1427
Rigat B, Hubert C, Alhenc-Gelas F, Cambien F, Corvol P, Soubrier F (1990) An insertion/deletion polymorphism in the angiotensin I-converting enzyme gene accounting for half the variance of serum enzyme levels. J Clin Invest 86(4):1343–1346
Costerousse O, Allegrini J, Lopez M, Alhenc-Gelas F (1993) Angiotensin I-converting enzyme in human circulating mononuclear cells: genetic polymorphism of expression in T-lymphocytes. Biochem J 290(Pt 1):33–40
Cambien F, Poirier O, Lecerf L, Evans A, Cambou JP, Arveiler D et al (1992) Deletion polymorphism in the gene for angiotensin-converting enzyme is a potent risk factor for myocardial infarction. Nature 359(6396):641–644
Sayed-Tabatabaei FA, Oostra BA, Isaacs A, van Duijn CM, Witteman JC (2006) ACE polymorphisms. Circ Res 98(9):1123–1133
Kammerer CM, Gouin N, Samollow PB, VandeBerg JF, Hixson JE, Cole SA et al (2004) Two quantitative trait loci affect ACE activities in Mexican–Americans. Hypertension 43(2):466–470
Inoue I, Nakajima T, Williams CS, Quackenbush J, Puryear R, Powers M et al (1997) A nucleotide substitution in the promoter of human angiotensinogen is associated with essential hypertension and affects basal transcription in vitro. J Clin Invest 99(7):1786–1797
Martin MM, Willardson BM, Burton GF, White CR, McLaughlin JN, Bray SM et al (2001) Human angiotensin II type 1 receptor isoforms encoded by messenger RNA splice variants are functionally distinct. Mol Endocrinol 15(2):281–293
Warnecke C, Surder D, Curth R, Fleck E, Regitz-Zagrosek V (1999) Analysis and functional characterization of alternatively spliced angiotensin II type 1 and 2 receptor transcripts in the human heart. J Mol Med 77(10):718–727
Abdollahi MR, Lewis RM, Gaunt TR, Cumming DV, Rodriguez S, Rose-Zerilli M et al (2007) Quantitated transcript haplotypes (QTH) of AGTR1, reduced abundance of mRNA haplotypes containing 1166C (rs5186:A>C), and relevance to metabolic syndrome traits. Hum Mutat 28(4):365–373
Lung CC, Chan EK, Zuraw BL (1997) Analysis of an exon 1 polymorphism of the B2 bradykinin receptor gene and its transcript in normal subjects and patients with C1 inhibitor deficiency. J Allergy Clin Immunol 99(1 Pt 1):134–146
Bassett MH, Zhang Y, Clyne C, White PC, Rainey WE (2002) Differential regulation of aldosterone synthase and 11beta-hydroxylase transcription by steroidogenic factor-1. J Mol Endocrinol 28(2):125–135
Lee-Kirsch MA, Gaudet F, Cardoso MC, Lindpaintner K (1999) Distinct renin isoforms generated by tissue-specific transcription initiation and alternative splicing. Circ Res 84(2):240–246
McNamara DM, Holubkov R, Postava L, Janosko K, MacGowan GA, Mathier M et al (2004) Pharmacogenetic interactions between angiotensin-converting enzyme inhibitor therapy and the angiotensin-converting enzyme deletion polymorphism in patients with congestive heart failure. J Am Coll Cardiol 44(10):2019–2026
Shin J, Lobmeyer MT, Gong Y, Zineh I, Langaee TY, Yarandi H et al (2007) Relation of beta(2)-adrenoceptor haplotype to risk of death and heart transplantation in patients with heart failure. Am J Cardiol 99(2):250–255
Spiering W, Kroon AA, Fuss-Lejeune MJ, de Leeuw PW (2005) Genetic contribution to the acute effects of angiotensin II type 1 receptor blockade. J Hypertens 23(4):753–758
Sookoian S, Castano G, Garcia SI, Viudez P, Gonzalez C, Pirola CJ (2005) A1166C angiotensin II type 1 receptor gene polymorphism may predict hemodynamic response to losartan in patients with cirrhosis and portal hypertension. Am J Gastroenterol 100(3):636–642
Nakayama M, Nakano H, Tsuboi N, Kurosawa T, Tsuruta Y, Iwasaki Y et al (2005) The effect of angiotensin receptor blockade ARB on the regression of left ventricular hypertrophy in hemodialysis patients: comparison between patients with D allele and non-D allele ACE gene polymorphism. Clin Nephrol 64(5):358–363
Hallberg P, Lind L, Michaelsson K, Karlsson J, Kurland L, Kahan T et al (2003) B2 bradykinin receptor (B2BKR) polymorphism and change in left ventricular mass in response to antihypertensive treatment: results from the Swedish Irbesartan Left Ventricular Hypertrophy Investigation versus Atenolol (SILVHIA) trial. J Hypertens 21(3):621–624
Heart Failure Society Of A. HFSA 2006 (2006) Comprehensive Heart Failure Practice Guideline. J Card Fail 12(1):e1–e2
Cicoira M, Zanolla L, Rossi A, Golia G, Franceschini L, Cabrini G et al (2001) Failure of aldosterone suppression despite angiotensin-converting enzyme (ACE) inhibitor administration in chronic heart failure is associated with ACE DD genotype. J Am Coll Cardiol 37(7):1808–1812
Cicoira M, Rossi A, Bonapace S, Zanolla L, Perrot A, Francis DP et al (2004) Effects of ACE gene insertion/deletion polymorphism on response to spironolactone in patients with chronic heart failure. Am J Med 116(10):657–661
Ohmichi N, Iwai N, Uchida Y, Shichiri G, Nakamura Y, Kinoshita M (1997) Relationship between the response to the angiotensin converting enzyme inhibitor imidapril and the angiotensin converting enzyme genotype. Am J Hypertens 10(8):951–955
Stavroulakis GA, Makris TK, Krespi PG, Hatzizacharias AN, Gialeraki AE, Anastasiadis G et al (2000) Predicting response to chronic antihypertensive treatment with fosinopril: the role of angiotensin-converting enzyme gene polymorphism. Cardiovasc Drugs Ther 14(4):427–432
Ueda S, Meredith PA, Morton JJ, Connell JM, Elliott HL (1998) ACE (I/D) genotype as a predictor of the magnitude and duration of the response to an ACE inhibitor drug (enalaprilat) in humans. Circulation 98(20):2148–2153
Arnett DK, Davis BR, Ford CE, Boerwinkle E, Leiendecker-Foster C, Miller MB et al (2005) Pharmacogenetic association of the angiotensin-converting enzyme insertion/deletion polymorphism on blood pressure and cardiovascular risk in relation to antihypertensive treatment: the Genetics of Hypertension-Associated Treatment (GenHAT) study. Circulation 111(25):3374–3383
Harrap SB, Tzourio C, Cambien F, Poirier O, Raoux S, Chalmers J et al (2003) The ACE gene I/D polymorphism is not associated with the blood pressure and cardiovascular benefits of ACE inhibition. Hypertension 42(3):297–303
Fan X, Wang Y, Sun K, Zhang W, Yang X, Wang S et al (2007) Polymorphisms of ACE2 gene are associated with essential hypertension and antihypertensive effects of Captopril in women. Clin Pharmacol Ther 82(2):187–196
Su X, Lee L, Li X, Lv J, Hu Y, Zhan S et al (2007) Association between angiotensinogen, angiotensin II receptor genes, and blood pressure response to an angiotensin-converting enzyme inhibitor. Circulation 115(6):725–732
Yu HM, Lin SG, Liu GZ, Zhang YQ, Ma WJ, Deng CY (2006) Associations between CYP11B2 gene polymorphisms and the response to angiotensin-converting enzyme inhibitors. Clin Pharmacol Ther 79(6):581–589
Risch N, Burchard E, Ziv E, Tang H (2002) Categorization of humans in biomedical research: genes, race and disease. Genome Biol 3(7):comment 2007
Cohn JN, Pfeffer MA, Rouleau J, Sharpe N, Swedberg K, Straub M et al (2003) Adverse mortality effect of central sympathetic inhibition with sustained-release moxonidine in patients with heart failure (MOXCON). Eur J Heart Fail 5(5):659–667
Bozkurt B, Torre-Amione G, Warren MS, Whitmore J, Soran OZ, Feldman AM et al (2001) Results of targeted anti-tumor necrosis factor therapy with etanercept (ENBREL) in patients with advanced heart failure. Circulation 103(8):1044–1047
Gheorghiade M, Gattis WA, O’Connor CM, Adams KF Jr, Elkayam U, Barbagelata A et al (2004) Effects of tolvaptan, a vasopressin antagonist, in patients hospitalized with worsening heart failure: a randomized controlled trial. JAMA 291(16):1963–1971
Gheorghiade M, Konstam MA, Burnett JC Jr, Grinfeld L, Maggioni AP, Swedberg K et al (2007) Short-term clinical effects of tolvaptan, an oral vasopressin antagonist, in patients hospitalized for heart failure: the EVEREST Clinical Status Trials. JAMA 297(12):1332–1343
Kaye DM, Krum H (2007) Drug discovery for heart failure: a new era or the end of the pipeline? Nat Rev Drug Discov 6(2):127–139
Roses AD (2004) Pharmacogenetics and drug development: the path to safer and more effective drugs. Nat Rev Genet 5(9):645–656
Effects of enalapril on mortality in severe congestive heart failure. Results of the Cooperative North Scandinavian Enalapril Survival Study (CONSENSUS) (1987) The CONSENSUS Trial Study Group. N Engl J Med 316(23):1429–1435
Acknowledgments
This work was supported in part by National Institutes of Health P50 HL077113 (ALB), GM74492 (IZ), American Heart Association Heartland Affiliate 0655496Z (ALB) and Florida/Puerto Rico Affiliate 0435278B (IZ).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Beitelshees, A.L., Zineh, I. Renin–angiotensin–aldosterone system (RAAS) pharmacogenomics: implications in heart failure management. Heart Fail Rev 15, 209–217 (2010). https://doi.org/10.1007/s10741-008-9092-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10741-008-9092-z