Abstract
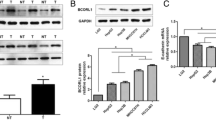
More and more studies have shown that Branched chain amino acid transaminase 1 (BCAT1) is involved in the occurrence and development of a variety of tumors. However, the mechanism of its occurrence and development in hepatocellular carcinoma (HCC) remains unclear. Here, we demonstrated the relationship between BCAT1 and AKT signaling pathway, as well as EMT, and the clinical significance of BCAT1 by using BCAT1 expression in 5 cell lines and 113 liver cancer and non-liver cancer tissue samples. The results showed that the expression of AKT was positively correlated with BCAT1 in HCC tissues, and BCAT1 could promote the progression of HCC cells through the AKT signaling pathway. Clinical analysis and Bioinformatics technology analysis revealed that BCAT1 was correlated with poor prognosis, and BCAT1 expression in the HCC tissues was evidently correlated with tumor number, vascular invasion, Edmondson grade and TNM stage (P < 0.05). In vitro studies showed that BCAT1 increased the invasion and migration of in MHCC-97H cells a d Huh7 cells. By inhibiting the expression of the BCAT1 gene, we detected the corresponding changes in the expression levels of Twist, E-cadherin and Vimentin, confirming that BCAT1 may promote the invasion and migration of HCC cells through epithelial-mesenchymal transformation (EMT). Overall, BCAT1 can activate AKT signaling pathway and EMT to promote the development and metastasis of HCC cells. this study may provide new ideas and directions for cancer diagnosis and treatment.








Similar content being viewed by others
Data availability
The data used to support the findings of this study are available from the corresponding authors upon request.
References
An Y, Wang Q, Zhang G, Sun F, Zhang L, Li H et al (2020) OSlihc: an online prognostic biomarker analysis tool for hepatocellular carcinoma. Front Pharmacol 11:875. https://doi.org/10.15252/embj.2021108647
Bishnupuri KS, Alvarado DM, Khouri AN, Shabsovich M, Chen B, Dieckgraefe BK et al (2019) IDO1 and kynurenine pathway metabolites activate PI3K-AKT signaling in the neoplastic colon epithelium to promote cancer cell proliferation and inhibit apoptosis. Cancer Res 79(6):1138–1150. https://doi.org/10.1158/0008-5472.CAN-18-0668
Brabletz S, Schuhwerk H, Brabletz T, Stemmler MP (2021) Dynamic EMT: a multi-tool for tumor progression. EMBO J 40(18):e108647. https://doi.org/10.15252/embj.2021108647
Caines A, Selim R, Salgia R (2020) The changing global epidemiology of hepatocellular carcinoma. Clin Liver Dis 24(4):535–547. https://doi.org/10.1016/j.cld.2020.06.001
Chen B, Hu Z, Li B, Lin X, Luo Z, Hu Z (2019) The expressions of Hedgehog and PI3K-AKT pathway components correlate with invasion and metastasis in hepatocellular carcinoma. Int J Clin Exp Pathol 12(6):2381–2388
Chen B, Hu Z, Chen B, Li B (2020) Effects and mechanism of lanthanum citrate on the proliferation and apoptosis of hepatocellular carcinoma cell line MHCC-97H. Turk J Gastroenterol 31(3):264–271. https://doi.org/10.5152/tjg.2020.18800
Fattahi S, Amjadi-Moheb F, Tabaripour R, Ghossein A, Akhavan-Niaki H (2020) PI3K/AKT/mTOR signaling in gastric cancer: epigenetics and beyond. Life Sci 262:118513. https://doi.org/10.1016/j.lfs.2020.118513
Gao YX, Yang TW, Yin JM, Yang PX, Kou BX, Chai MY et al (2020) Progress and prospects of biomarkers in primary liver cancer (Review). Int J Oncol 57(1):54–66. https://doi.org/10.3892/ijo.2020.5035
Go M, Shin E, Jang SY, Nam M, Hwang GS, Lee SY (2022) BCAT1 promotes osteoclast maturation by regulating branched-chain amino acid metabolism. Exp Mol Med 54(6):825–833. https://doi.org/10.1038/s12276-022-00775-3
Hattori A, Tsunoda M, Konuma T, Kobayashi M, Nagy T, Glushka J et al (2017) Cancer progression by reprogrammed BCAA metabolism in myeloid leukaemia. Nature 545(7655):500–504. https://doi.org/10.1038/nature22314
He D, Gao J, Zheng L, Liu S, Ye L, Lai H et al (2021) TGF-β inhibitor RepSox suppresses osteosarcoma via the JNK/Smad3 signaling pathway. Int J Oncol 59(5):84. https://doi.org/10.3892/ijo.2021.5264
Honda M, Takehana K, Sakai A, Tagata Y, Shirasaki T, Nishitani S et al (2011) Malnutrition impairs interferon signaling through mTOR and FoxO pathways in patients with chronic hepatitis C. Gastroenterology 141(1):128-140.e1-2. https://doi.org/10.1053/j.gastro.2011.03.051
Hu Z, Xie F, Hu A, Xu M, Liu Y, Zhang J et al (2020) Silencing glioma-associated oncogene homolog 1 suppresses the migration and invasion of hepatocellular carcinoma in vitro. Oncol Lett 20(5):228. https://doi.org/10.3892/ol.2020.12091
Khan MA, Jain VK, Rizwanullah M, Ahmad J, Jain K (2019) PI3K/AKT/mTOR pathway inhibitors in triple-negative breast cancer: a review on drug discovery and future challenges. Drug Discov Today 24(11):2181–2191. https://doi.org/10.1016/j.drudis.2019.09.001
Lambert AW, Weinberg RA (2021) Linking EMT programmes to normal and neoplastic epithelial stem cells. Nat Rev Cancer 21(5):325–338. https://doi.org/10.1038/s41568-021-00332-6
Le Couteur DG, Solon-Biet SM, Cogger VC, Ribeiro R, de Cabo R, Raubenheimer D et al (2020) Branched chain amino acids, aging and age-related health. Ageing Res Rev 64:101198. https://doi.org/10.1016/j.arr.2020.101198
Lee SK, Lee SW, Jang JW, Bae SH, Choi JY, Yoon SK (2021) Immunological markers, prognostic factors and challenges following curative treatments for hepatocellular carcinoma. Int J Mol Sci 22(19):10271. https://doi.org/10.3390/ijms221910271
Lin XM, Tan ST, Fu L, Dong QZ (2020) BCAT1 overexpression promotes proliferation, invasion, and Wnt signaling in non-small cell lung cancers. Onco Targets Ther 13:3583–3594. https://doi.org/10.2147/OTT.S237306
Liu QQ, Yang PH, Tu KS, Zhang HG, Zheng X, Yao YM et al (2014) TPX2 knockdown suppressed hepatocellular carcinoma cell invasion via inactivating AKT signaling and inhibiting MMP2 and MMP9 expression. Chin J Cancer Res 26(4):410–417. https://doi.org/10.3978/j.issn.1000-9604.2014.08.01
Liu YX, Dong WB, Shao J, Wang YB, Zhou MY, Sun HP (2017) Branched-chain amino acid negatively regulates KLF15 expression via PI3K-AKT pathway. Front Physiol 8:853. https://doi.org/10.3389/fphys.2017.00853
Liu QQ, Liu YW, Xie YK, Zhang JH, Song CX, Wang JZ et al (2021) Amplification of DDR2 mediates sorafenib resistance through NF-κB/c-Rel signaling in hepatocellular carcinoma. Cell Biol Int 45(9):1906–1916. https://doi.org/10.1002/cbin.11625
Luo L, Sun W, Zhu W, Li S, Zhang W, Xu X et al (2021) BCAT1 decreases the sensitivity of cancer cells to cisplatin by regulating mtor-mediated autophagy via branched-chain amino acid metabolism. Cell Death Dis 12(2):169. https://doi.org/10.1038/s41419-021-03456-7
Mao L, Chen J, Lu X, Yang C, Ding Y, Wang MM et al (2021) Proteomic analysis of lung cancer cells reveals a critical role of BCAT1 in cancer cell metastasis. Theranostics 11(19):9705–9720. https://doi.org/10.7150/thno.61731
Martin SB, Reiche WS, Fifelski NA, Schultz AJ, Stanford SJ, Martin AA et al (2020) Leucine and branched-chain amino acid metabolism contribute to the growth of bone sarcomas by regulating AMPK and mTORC1 signaling. Biochem J 477(9):1579–1599. https://doi.org/10.1042/BCJ20190754
Peng L, Wen L, Shi QF, Gao F, Huang B, Meng J et al (2020) Scutellarin ameliorates pulmonary fibrosis through inhibiting NF-κB/NLRP3-mediated epithelial-mesenchymal transition and inflammation. Cell Death Dis 11(11):978. https://doi.org/10.1038/s41419-020-03178-2
Ren JY, Gu YH, Cui XW, Long MM, Wang W, Wei CJ, Wang ZC et al (2020) Protein tyrosine phosphatase receptor S acts as a metastatic suppressor in malignant peripheral nerve sheath tumor via profilin 1-induced epithelial-mesenchymal transition. Front Cell Dev Biol 8:582220. https://doi.org/10.3389/fcell.2020.582220
Saxena K, Jolly MK, Balamurugan K (2020) Hypoxia, partial EMT and collective migration: emerging culprits in metastasis. Transl Oncol 13(11):100845. https://doi.org/10.1016/j.tranon.2020.100845
Shafei MA, Forshaw T, Davis J, Flemban A, Qualtrough D, Dean S et al (2020) BCATc modulates crosstalk between the PI3K/AKT and the Ras/ERK pathway regulating proliferation in triple negative breast cancer. Oncotarget 11(21):1971–1987. https://doi.org/10.18632/oncotarget.27607
Shu X, Zhan PP, Sun LX, Yu L, Liu J, Sun LC et al (2021) BCAT1 activates PI3K/AKT/mTOR pathway and contributes to the angiogenesis and tumorigenicity of gastric cancer. Front Cell Dev Biol 9:659260. https://doi.org/10.3389/fcell.2021.659260
Toyokawa Y, Koonthongkaew J, Takagi H (2021) An overview of branched-chain amino acid aminotransferases: functional differences between mitochondrial and cytosolic isozymes in yeast and human. Appl Microbiol Biotechnol 105(21–22):8059–8072. https://doi.org/10.1007/s00253-021-11612-4
Uddin GM, Karwi QG, Pherwani S, Gopal K, Wagg CS, Biswas D et al (2021) Deletion of BCATm increases insulin-stimulated glucose oxidation in the heart. Metabolism 124:154871. https://doi.org/10.1016/j.metabol.2021.154871
Wang ZQ, Faddaoui A, Bachvarova M, Plante M, Gregoire J, Renaud MC et al (2015) BCAT1 expression associates with ovarian cancer progression: possible implications in altered disease metabolism. Oncotarget 6(31):31522–31543. https://doi.org/10.18632/oncotarget.5159
Wang H, Wang F, Ouyang W, Jiang X, Wang Y (2021) BCAT1 overexpression regulates proliferation and c-Myc/GLUT1 signaling in head and neck squamous cell carcinoma. Oncol Rep 45(5):52. https://doi.org/10.3892/or.2021.8003
Xu M, Liu Q, Jia Y, Tu K, Yao Y, Liu Q et al (2016) BCAT1 promotes tumor cell migration and invasion in hepatocellular carcinoma. Oncol Lett 12(4):2648–2656. https://doi.org/10.3892/ol.2016.4969
Xu Y, Yu W, Yang T, Zhang M, Liang C, Cai X et al (2018) Overexpression of BCAT1 is a prognostic marker in gastric cancer. Hum Pathol 75:41–46. https://doi.org/10.1016/j.humpath.2018.02.003
Yang JD, Hainaut P, Gores GJ, Amadou A, Plymoth A, Roberts LR (2019a) A global view of hepatocellular carcinoma: trends, risk, prevention and management. Nat Rev Gastroenterol Hepatol 16(10):589–604. https://doi.org/10.1038/s41575-019-0186-y
Yang P, Chen S, Zhong G, Wang Y, Kong W, Wang Y (2019b) ResolvinD1 attenuates high-mobility group box 1-induced epithelial-to-mesenchymal transition in nasopharyngeal carcinoma cells. Exp Biol Med (maywood) 244(18):1608–1618. https://doi.org/10.1177/1535370219885320
Yang Q, Xie B, Tang H, Meng W, Jia C, Zhang X et al (2019c) Minichromosome maintenance 3 promotes hepatocellular carcinoma radioresistance by activating the NF-κB pathway. J Exp Clin Cancer Res 38(1):263. https://doi.org/10.1186/s13046-019-1241-9
Yi L, Fan X, Li J, Yuan F, Zhao J, Nistér M, Yang X (2021) Enrichment of branched chain amino acid transaminase 1 correlates with multiple biological processes and contributes to poor survival of IDH1 wild-type gliomas. Aging (albany NY) 13(3):3645–3660. https://doi.org/10.18632/aging.202328
Zeng B, Zhang X, Zhao J, Wei Z, Zhu H, Fu M et al (2019) The role of DNMT1/hsa-miR-124-3p/BCAT1 pathway in regulating growth and invasion of esophageal squamous cell carcinoma. BMC Cancer 19(1):609. https://doi.org/10.1186/s12885-019-5815-x
Zhang L, Han JQ (2019) Branched-chain amino acid transaminase 1 (BCAT1) promotes the growth of breast cancer cells through improving mTOR-mediated mitochondrial biogenesis and function. Biochem Biophys Res Commun 486(2):224–231. https://doi.org/10.1016/j.bbrc.2017.02.101
Zhang B, Xu F, Wang K, Liu M, Li J, Zhao Q et al (2021) BCAT1 knockdown-mediated suppression of melanoma cell proliferation and migration is associated with reduced oxidative phosphorylation. Am J Cancer Res 11(6):2670–2683
Zheng YH, Hu WJ, Chen BC, Grahn TH, Zhao YR, Bao HL et al (2016) BCAT1, a key prognostic predictor of hepatocellular carcinoma, promotes cell proliferation and induces chemoresistance to cisplatin. Liver Int 36(12):1836–1847. https://doi.org/10.1111/liv.13178
Zhu WJ, Shao YQ, Peng Y (2017) MicroRNA-218 inhibits tumor growth and increases chemosensitivity to CDDP treatment by targeting BCAT1 in prostate cancer. Mol Carcinog 56(6):1570–1577. https://doi.org/10.1002/mc.22612
Zhu Q, Yu X, Zhou ZW, Luo M, Zhou C, He ZX, Chen Y, Zhou SF (2018) A quantitative proteomic response of hepatocellular carcinoma Hep3B cells to danusertib, a pan-Aurora kinase inhibitor. J Cancer 9(12):2061–2071. https://doi.org/10.7150/jca.20822
Zhu ZW, Achreja A, Meurs N, Animasahun O, Owen S, Mittal A et al (2020) Tumour-reprogrammed stromal BCAT1 fuels branched-chain ketoacid dependency in stromal-rich PDAC tumours. Nat Metab 2(8):775–792. https://doi.org/10.1038/s42255-020-0226-5
Acknowledgements
The principal authors are very grateful for the patients who participated in the study.
Funding
The present study was in part supported by the National Natural Science Foundation of China (Grant No. 82060505), Science and Technology Projects of Ganzhou City (Grant Nos. GZ2017ZSF053 and GZ2017ZSF264), Science and technology research project of the Education Department of Jiangxi Province (Grant No. GJJ201516). Key Project of Key Research and Development Plan of Jiangxi Province (Grant No. 20212BBG71013).
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Experiments was designed by QL. Data collection and analysis were performed by YD, XW, SL and AL. The first draft of the manuscript was written by YD and XW. The manuscript was revised by BX and XH. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
All the authors have no relevant financial or non-financial interests to disclose.
Ethical approval
This article has been reviewed and approved by the ethics board of The First Affiliated Hospital of Gannan Medical University.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ding, Y., Wang, X., Lu, S. et al. BCAT1, as a prognostic factor for HCC, can promote the development of liver cancer through activation of the AKT signaling pathway and EMT. J Mol Histol 54, 25–39 (2023). https://doi.org/10.1007/s10735-022-10108-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10735-022-10108-3