Abstract
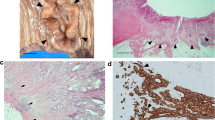
The purpose of this study was to evaluate the expression of Hedgehog (HH) signaling molecules (SHH and GLI-1) by cancer-associated fibroblasts (CAF) in oral squamous cell carcinoma (OSCC). Immunohistochemistry was used to detect molecular HH signaling and CAF-related protein expression, including α-SMA and S100A4, in 70 samples of human OSCC. The colocalization of α-SMA and S100A4 with SHH was also evaluated by double-staining. In vitro study was performed using primary normal oral fibroblast (NOF) and CAF through immunofluorescence and Western Blot for CAF-proteins, SHH, and GLI-1. Forty-five cases (64.28%) were positive for α-SMA exclusively in tumor stroma, and S100A4 was identified in the cytoplasm of CAFs in 94.28% (n = 66) of the cases. With respect to stromal cells, 64 (91.43%) OSCC cases were positive for SHH, and 31 were positive for GLI-1 (44.29%); positive correlations were found between SHH and α-SMA (p < 0.0001, φ = 0.51), as well as between SHH and S100A4 (p = 0.087, φ = 0.94). Protein expression of SHH and GLI-1 was observed in primary CAFs and NOFs. Although SHH was found to be localized in the cellular cytoplasm of both cell types, GLI-1 was present only in the nuclei of CAF. Our results indicate that CAFs are not only potential sources of HH ligands in tumor stroma, but may also respond to HH signaling through nuclear GLI-1 activation. We further observed that elevated SHH expression by OSCC cells was associated with higher CAF density, reinforcing the chemoattractant role played by these molecules.





Similar content being viewed by others
References
Abe Y, Tanaka N (2016) The hedgehog signaling networks in lung cancer: the mechanisms and roles in tumor progression and implications for cancer therapy. Biomed Res Int. https://doi.org/10.1155/2016/7969286
Adrien J, Bertolus C, Gambotti L et al (2014) Why are head and neck squamous cell carcinoma diagnosed so late? influence of health care disparities and socio-economic factors. Oral Oncol 50:90–97. https://doi.org/10.1016/j.oraloncology.2013.10.016
Amakye D, Jagani Z, Dorsch M (2013) Unraveling the therapeutic potential of the Hedgehog pathway in cancer. Nat Med 19:1410–1422. https://doi.org/10.1038/nm.3389
Ansari SS, Sharma AK, Zepp M et al (2018) Upregulation of cell cycle genes in head and neck cancer patients may be antagonized by erufosine’s down regulation of cell cycle processes in OSCC cells. Oncotarget 9:5797–5810. https://doi.org/10.18632/oncotarget.23537
Armas-López L, Zúñiga J, Arrieta O, Ávila-Moreno F (2017) The Hedgehog-GLI pathway in embryonic development and cancer: Implications for pulmonary oncology therapy. Oncotarget 8:60684–60703. https://doi.org/10.18632/oncotarget.19527
Bailey JM, Swanson BJ, Hamada T et al (2008) Sonic hedgehog promotes desmoplasia in pancreatic cancer. Clin Cancer Res 14:5995–6004. https://doi.org/10.1158/1078-0432.CCR-08-0291
Bray F, Ferlay J, Soerjomataram I (2018) Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. https://doi.org/10.3322/caac.21492
Cavicchioli Buim ME, Gurgel CAS, Gonçalves Ramos EA et al (2011) Activation of sonic hedgehog signaling in oral squamous cell carcinomas: a preliminary study. Hum Pathol 42:1484–1490. https://doi.org/10.1016/j.humpath.2010.12.015
Cerami E, Gao J, Dogrusoz U et al (2012) The cBio cancer genomics portal: an open platform for exploring multidimensional cancer genomics data: figure 1. Cancer Discov 2:401–404. https://doi.org/10.1158/2159-8290.CD-12-0095
Chi AC, Day TA, Neville BW (2015) Oral cavity and oropharyngeal squamous cell carcinoma-an update. CA Cancer J Clin 65:401–421. https://doi.org/10.3322/caac.21293
Cirillo N, Hassona Y, Celentano A et al (2017) Cancer-associated fibroblasts regulate keratinocyte cell-cell adhesion via TGF-β-dependent pathways in genotype-specific oral cancer. Carcinogenesis 38:76–85. https://doi.org/10.1093/carcin/bgw113
Costea DE, Hills A, Osman AH et al (2013) Identification of two distinct carcinoma-associated fibroblast subtypes with differential tumor-promoting abilities in oral squamous cell carcinoma. Cancer Res 73:3888–3901. https://doi.org/10.1158/0008-5472.CAN-12-4150
Davis J (1971) Elementary survey analysis. Englewood cliffs. Prentice-Hall, NJ.
de FValverde L, de APereira T, Dias RB et al (2016) Macrophages and endothelial cells orchestrate tumor-associated angiogenesis in oral cancer via hedgehog pathway activation. Tumor Biol 37:9233–9241. https://doi.org/10.1007/s13277-015-4763-6
Dias RB, De Faro VL, Sales CBS et al (2016) Enhanced expression of hedgehog pathway proteins in oral epithelial dysplasia. Appl Immunohistochem Mol Morphol 24:596–602. https://doi.org/10.1097/PAI.0000000000000225
Dourado MR, Korvala J, Åström P et al (2019) Extracellular vesicles derived from cancer-associated fibroblasts induce the migration and invasion of oral squamous cell carcinoma. J Extracell Vesicles. https://doi.org/10.1080/20013078.2019.1578525
Gao J, Aksoy BA, Dogrusoz U et al (2013) Integrative analysis of complex cancer genomics and clinical profiles using the cBioPortal. Sci Signal. https://doi.org/10.1126/scisignal.2004088
Gascard P, Tlsty TD (2016) Carcinoma-associated fibroblasts: orchestrating the composition of malignancy. Genes Dev 30:1002–1019. https://doi.org/10.1101/gad.279737.116
Göritz C, Dias DO, Tomilin N et al (2011) A pericyte origin of spinal cord scar tissue. Science 333:238–242. https://doi.org/10.1126/science.1203165
Habib JG, O’Shaughnessy JA (2016) The hedgehog pathway in triple-negative breast cancer. Cancer Med 5:2989–3006. https://doi.org/10.1002/cam4.833
Hanahan D, Coussens LM (2012) Accessories to the crime: functions of cells recruited to the tumor microenvironment. Cancer Cell 21:309–322. https://doi.org/10.1016/j.ccr.2012.02.022
Horn A, Palumbo K, Cordazzo C et al (2012) Hedgehog signaling controls fibroblast activation and tissue fibrosis in systemic sclerosis. Arthritis Rheum 64:2724–2733. https://doi.org/10.1002/art.34444
IARC (2005) WHO histological classification of tumours of the oral cavity and oropharynx. In: Barnes L (ed) Pathology and genetics of head and neck tumours. IARC Press, France, pp 163–181
Jotzu C, Alt E, Welte G et al (2011) Adipose tissue derived stem cells differentiate into carcinoma-associated fibroblast-like cells under the influence of tumor derived factors. Cell Oncol 34:55–67. https://doi.org/10.1007/s13402-011-0012-1
Kalluri R (2016) The biology and function of fibroblasts in cancer. Nat Rev Cancer 16:582–598. https://doi.org/10.1038/nrc.2016.73
Kayamori K, Katsube KI, Sakamoto K et al (2016) NOTCH3 s induced in cancer-associated fibroblasts and promotes angiogenesis in oral squamous cell carcinoma. PLoS ONE 11:e0154112. https://doi.org/10.1371/journal.pone.0154112
Kellermann MG, Sobral LM, Da Silva SD et al (2007) Myofibroblasts in the stroma of oral squamous cell carcinoma are associated with poor prognosis. Histopathology 51:849–853. https://doi.org/10.1111/j.1365-2559.2007.02873.x
Kellermann MG, Sobral LM, da Silva SD et al (2008) Mutual paracrine effects of oral squamous cell carcinoma cells and normal oral fibroblasts: induction of fibroblast to myofibroblast transdifferentiation and modulation of tumor cell proliferation. Oral Oncol 44:509–517. https://doi.org/10.1016/j.oraloncology.2007.07.001
Kidd S, Spaeth E, Watson K et al (2012) Origins of the tumor microenvironment: quantitative assessment of adipose-derived and bone marrow-derived stroma. PLoS ONE. https://doi.org/10.1371/journal.pone.0030563
Kuroda H, Kurio N, Shimo T et al (2017) Oral squamous cell carcinoma-derived sonic hedgehog promotes angiogenesis. Anticancer Res 37:6731–6737
Lin NN, Wang P, Zhao D et al (2017) Significance of oral cancer-associated fibroblasts in angiogenesis, lymphangiogenesis, and tumor invasion in oral squamous cell carcinoma. J Oral Pathol Med 46:21–30. https://doi.org/10.1111/jop.12452
McMillan R, Matsui W (2012) Molecular pathways: the hedgehog signaling pathway in cancer. Natl Institutes Heal 18:4883–4888. https://doi.org/10.1158/1078-0432.CCR-11-2509
Mpekris F, Papageorgis P, Polydorou C et al (2017) Sonic-hedgehog pathway inhibition normalizes desmoplastic tumor microenvironment to improve chemo- and nanotherapy. J Control Release 261:105–112. https://doi.org/10.1016/j.jconrel.2017.06.022
Nayak A, Siddharth S, Das S et al (2017) Nanoquinacrine caused apoptosis in oral cancer stem cells by disrupting the interaction between GLI1 and β catenin through activation of GSK3β. Toxicol Appl Pharmacol 330:53–64. https://doi.org/10.1016/j.taap.2017.07.008
Öhlund D, Elyada E, Tuveson D (2014) Fibroblast heterogeneity in the cancer wound. J Exp Med 211:1503–1523. https://doi.org/10.1084/jem.20140692
Orimo A, Gupta PB, Sgroi DC et al (2005) Stromal fibroblasts present in invasive human breast carcinomas promote tumor growth and angiogenesis through elevated SDF-1/CXCL12 secretion. Cell 121:335–348. https://doi.org/10.1016/j.cell.2005.02.034
Otsuka A, Levesque MP, Dummer R, Kabashima K (2015) Hedgehog signaling in basal cell carcinoma. J Dermatol Sci 78:95–100. https://doi.org/10.1016/j.jdermsci.2015.02.007
Peiris-Pagès M, Sotgia F, Lisanti M (2015) Chemotherapy induces the cancer-associated fibroblast phenotype, activating paracrine Hedgehog-GLI signalling in breast cancer cells. Oncotarget 6:10728–10745. https://doi.org/10.18632/oncotarget.3828
Quante M, Tu SP, Tomita H et al (2011) Bone marrow-derived myofibroblasts contribute to the mesenchymal stem cell niche and promote tumor growth. Cancer Cell 19:257–272. https://doi.org/10.1016/j.ccr.2011.01.020
Ramos-Vega V, Venegas Rojas B, Donoso Torres W (2020) Immunohistochemical analysis of cancer-associated fibroblasts and podoplanin in head and neck cancer. Med Oral Patol Oral y Cir Bucal. https://doi.org/10.4317/medoral.23335
Santi A, Kugeratski FG, Zanivan S (2018) Cancer associated fibroblasts: the architects of stroma remodeling. Proteomics 18:1–15. https://doi.org/10.1002/pmic.201700167
Shin K, Lim A, Zhao C et al (2015) Hedgehog signaling restrains bladder cancer progression by eliciting stromal production of urothelial differentiation factors. Cancer Cell 26:521–533. https://doi.org/10.1016/j.ccell.2014.09.001.Hedgehog
Sobral LM, Bufalino A, Lopes MA et al (2011) Myofibroblasts in the stroma of oral cancer promote tumorigenesis via secretion of activin A. Oral Oncol 47:840–846. https://doi.org/10.1016/j.oraloncology.2011.06.011
Spivak-Kroizman TR, Hostetter G, Posner R et al (2013) Hypoxia triggers hedgehog-mediated tumor-stromal interactions in pancreatic cancer. Cancer Res 73:3235–3247. https://doi.org/10.1158/0008-5472.CAN-11-1433
Takabatake K, Shimo T, Murakami J et al (2019) The role of sonic hedgehog signaling in the tumor microenvironment of oral squamous cell carcinoma. Int J Mol Sci. https://doi.org/10.3390/ijms20225779
Takahashi H, Sakakura K, Kudo T et al (2017) Cancer-associated fibroblasts promote an immunosuppressive microenvironment through the induction and accumulation of protumoral macrophages. Oncotarget 8:8633–8647. https://doi.org/10.18632/oncotarget.14374
The World Health Organization (2001) Declaration of Helsinki: World medical association Declaration of Helsinki. Bull world Heal Organ 79:373–374
Theunissen JW, de Sauvage FJ (2009) Paracrine hedgehog signaling in cancer. Cancer Res 69:6007–6010. https://doi.org/10.1158/0008-5472.CAN-09-0756
Valenti G, Quinn HM, Heynen GJJE et al (2017) Cancer stem cells regulate cancer-associated fibroblasts via activation of hedgehog signaling in mammary gland tumors. Cancer Res 77:2134–2147. https://doi.org/10.1158/0008-5472.CAN-15-3490
Vered M, Shnaiderman-Shapiro A, Zlotogorski-Hurvitz A et al (2019) Cancer-associated fibroblasts in the tumor microenvironment of tongue carcinoma is a heterogeneous cell population. Acta Histochem 121:151446. https://doi.org/10.1016/j.acthis.2019.151446
Wu F, Zhang Y, Sun B et al (2017) Hedgehog signaling: from basic biology to cancer therapy. Cell Chem Biol 24:252–280. https://doi.org/10.1016/j.chembiol.2017.02.010
Zhou B, Chen WL, Wang YY et al (2014) A role for cancer-associated fibroblasts in inducing the epithelial-to-mesenchymal transition in human tongue squamous cell carcinoma. J Oral Pathol Med 43:585–592. https://doi.org/10.1111/jop.12172
Acknowledgements
The authors thank Andris K. Walter for providing English translation and consulting services.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and publication of this article: This work was supported by the National Council for Scientific and Technological Development (CNPq), [Grant No. 308595/2016‐5].
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Guimaraes, V.S.N., Vidal, M.T.A., de Faro Valverde, L. et al. Hedgehog pathway activation in oral squamous cell carcinoma: cancer-associated fibroblasts exhibit nuclear GLI-1 localization. J Mol Hist 51, 675–684 (2020). https://doi.org/10.1007/s10735-020-09913-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10735-020-09913-5