Abstract
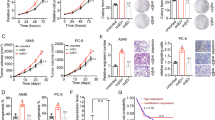
Integrin-linked kinase (ILK) forms a heterotrimeric protein complex with PINCH and PARVIN (IPP) in Focal Adhesions (FAs) that acts as a signaling platform between the cell and its microenvironment regulating important cancer-related functions. We aimed to elucidate the role of ILK in lung adenocarcinoma (LUADC) focusing on a possible link with KRAS oncogene. We used immunohistochemistry on human tissue samples and KRAS-driven LUADC in mice, analysis of large scale publicly available RNA sequencing data, ILK overexpression and pharmacological inhibition as well as knockdown of KRAS in lung cancer cells. ILK, PINCH1 and PARVB (IPP) proteins are overexpressed in human LUADC and KRAS-driven LUADC in mice representing poor prognostic indicators. Genes implicated in ILK signaling are significantly enriched in KRAS-driven LUADC. Silencing of KRAS, as well as, overexpression and pharmacological inhibition of ILK in lung cancer cells provide evidence of a two-way association between ILK and KRAS. Upregulation of PINCH, PARVB and Ras suppressor-1 (RSU1) expression was demonstrated in ILK overexpressing lung cancer cells in addition to a significant positive correlation between these factors in tissue samples, while KRAS silencing downregulates IPP and RSU1. Pharmacological inhibition of ILK in KRAS mutant lung cancer cells suppresses cell growth, migration, EMT and increases sensitivity to platinum-based chemotherapy. ILK promotes an aggressive lung cancer phenotype with prognostic and therapeutic value through functions that involve KRAS, IPP complex and RSU1, rendering ILK a promising biomarker and therapeutic target in lung adenocarcinoma.





Similar content being viewed by others
Data availability
Data generated during and/or analysed during the current study are available from the authors upon request.
Abbreviations
- ECM:
-
Extracellular matrix
- EMT:
-
Epithelial to mesenchymal transition
- ERK1/2:
-
Extracellular regulated kinase
- FFPE:
-
Formalin fixed paraffin embedded
- FA:
-
Focal adhesion
- GSEA:
-
Gene set enrichment analysis
- H-score:
-
Histoscore
- ILK:
-
Integrin-linked kinase
- IPP:
-
ILK-PINCH-PARVIN
- IQ-GAP1:
-
Ras-GTPase activating-like protein 1
- LUADC:
-
Lung adenocarcinoma
- NSCLC:
-
Non small cell lung cancer
- OS:
-
Overall survival
- qPCR:
-
Quantitative PCR
- RSU1:
-
Ras suppressor-1
- WB:
-
Western blot
References
Agalioti T, Giannou AD, Krontira AC, Kanellakis NI, Kati D, Vreka M, Pepe M et al (2017) Mutant KRAS promotes malignant pleural effusion formation. Nat Commun 8:15205. https://doi.org/10.1038/ncomms15205
Attwell S, Mills J, Troussard A, Wu C, Dedhar S (2003) Integration of cell attachment, cytoskeletal localization, and signaling by integrin-linked kinase (ILK), CH-ILKBP, and the tumor suppressor PTEN. Mol Biol Cell 14(12):4813–4825. https://doi.org/10.1091/mbc.e03-05-0308
Bravou V, Klironomos G, Papadaki E, Taraviras S, Varakis J (2006) ILK over-expression in human colon cancer progression correlates with activation of β-catenin, down-regulation of E-cadherin and activation of the Akt–FKHR pathway. J Pathol 208(1):91–99. https://doi.org/10.1002/path.1860
Camp RL, Dolled-Filhart M, Rimm DL (2004) X-tile. Clin Cancer Res 10(21):7252–7259. https://doi.org/10.1158/1078-0432.CCR-04-0713
Cerami E, Gao J, Dogrusoz U, Gross BE, Sumer SO, Aksoy BA, Jacobsen A et al (2012) The CBio cancer genomics portal: an open platform for exploring multidimensional cancer genomics data. Cancer Discov 2(5):401–404. https://doi.org/10.1158/2159-8290.CD-12-0095
Chan BA, Hughes BG (2015) Targeted therapy for non-small cell lung cancer: current standards and the promise of the future. Transl Lung Cancer Res 4(1):36–54. https://doi.org/10.3978/j.issn.2218-6751.2014.05.01
Chen D, Zhang Y, Zhang X, Li J, Han B, Liu S, Wang L, Ling Y, Mao S, Wang X (2013) Overexpression of integrin-linked kinase correlates with malignant phenotype in non-small cell lung cancer and promotes lung cancer cell invasion and migration via regulating epithelial-mesenchymal transition (EMT)-related genes. Acta Histochem 115(2):128–136. https://doi.org/10.1016/j.acthis.2012.05.004
Chu P-C, Yang M-C, Kulp SK, Salunke SB, Himmel LE, Fang C-S, Jadhav AM et al (2016) Regulation of oncogenic KRAS signaling via a novel KRAS-integrin-linked kinase-HnRNPA1 regulatory loop in human pancreatic cancer cells. Oncogene 35(30):3897–3908. https://doi.org/10.1038/onc.2015.458
Chu PC, Kulp SK, Bekaii-Saab T, Chen CS (2018) Targeting integrin-linked kinase to suppress oncogenic KRAS signaling in pancreatic cancer. Small GTPases 9(6):452–456. https://doi.org/10.1080/21541248.2016.1251383
Cutler ML, Bassin RH, Zanoni L, Talbot N (1992) Isolation of Rsp-1, a novel CDNA capable of suppressing v-ras transformation. Mol Cell Biol 12(9):3750–3756. https://doi.org/10.1128/MCB.12.9.3750
Dougherty GW, Jose C, Gimona M, Cutler ML (2008) The Rsu-1-PINCH1-ILK complex is regulated by ras activation in tumor cells. Eur J Cell Biol 87(8–9):721–734. https://doi.org/10.1016/j.ejcb.2008.02.011
Duxbury MS, Ito H, Benoit E, Waseem T, Ashley SW, Whang EE (2005) RNA interference demonstrates a novel role for integrin-linked kinase as a determinant of pancreatic adenocarcinoma cell gemcitabine chemoresistance. Clin Cancer Res 11(9):3433–3438. https://doi.org/10.1158/1078-0432.CCR-04-1510
Edwards LA, Thiessen B, Dragowska WH, Daynard T, Bally MB, Dedhar S (2005) Inhibition of ILK in PTEN-mutant human glioblastomas inhibits PKB/Akt activation, induces apoptosis, and delays tumor growth. Oncogene 24(22):3596–3605. https://doi.org/10.1038/sj.onc.1208427
Elias MC, Pronovost SM, Cahill KJ, Beckerle MC, Kadrmas JL (2012) A crucial role for Ras suppressor-1 (RSU-1) revealed when PINCH and ILK binding is disrupted. J Cell Sci 125(13):3185–3194. https://doi.org/10.1242/jcs.101386
Eslami A, Miyaguchi K, Mogushi K, Watanabe H, Okada N, Shibuya H, Mizushima H, Miura M, Tanaka H (2014) PARVB overexpression increases cell migration capability and defines high risk for endophytic growth and metastasis in tongue squamous cell carcinoma. Br J Cancer 112:338. https://doi.org/10.1038/bjc.2014.590
Ferrer I, Zugazagoitia J, Herbertz S, John W, Paz-Ares L, Schmid-Bindert G (2018) KRAS mutant non-small cell lung cancer: from biology to therapy. Lung Cancer 124:53–64. https://doi.org/10.1016/j.lungcan.2018.07.013
Fielding AB, Lim S, Montgomery K, Dobreva I, Dedhar S (2011) A critical role of integrin-linked kinase, Ch-TOG and TACC3 in centrosome clustering in cancer cells. Oncogene 30(5):521–534. https://doi.org/10.1038/onc.2010.431
Filipenko NR, Attwell S, Roskelley C, Dedhar S (2005) Integrin-linked kinase activity regulates Rac- and Cdc42-mediated actin cytoskeleton reorganization via α-PIX. Oncogene 24(38):5837–5849. https://doi.org/10.1038/sj.onc.1208737
Bravou V, Antonacopoulou A, Papanikolaou S, Nikou S, Lilis I, Giannopoulou E, Kalofonos HP (2015) Focal adhesion proteins α- and β-parvin are overexpressed in human colorectal cancer and correlate with tumor progression. Cancer Investig 33(8):387–397. https://doi.org/10.3109/07357907.2015.1047508
Fuchs BC, Fujii T, Dorfman JD, Goodwin JM, Zhu AX, Lanuti M, Tanabe KK (2008) Epithelial-to-mesenchymal transition and integrin-linked kinase mediate sensitivity to epidermal growth factor receptor inhibition in human hepatoma cells. Cancer Res 68(7):2391–2399. https://doi.org/10.1158/0008-5472.CAN-07-2460
Fukuda K, Gupta S, Chen K, Wu C, Qin J (2009) The pseudoactive site of ILK is essential for its binding to alpha-parvin and localization to focal adhesions. Mol Cell 36(5):819–830. https://doi.org/10.1016/j.molcel.2009.11.028
Fukuda T, Chen K, Shi X, Wu C (2003) PINCH-1 is an obligate partner of integrin-linked kinase (ILK) functioning in cell shape modulation, motility, and survival. J Biol Chem 278(51):51324–51333. https://doi.org/10.1074/jbc.M309122200
Gao J, Aksoy BA, Dogrusoz U, Dresdner G, Gross B, Sumer SO, Sun Y, Jacobsen A, Sinha R, Larsson E, Cerami E (2013) Integrative analysis of complex cancer genomics and clinical profiles using the cBioPortal. Sci Signal 6(269):l1. https://doi.org/10.1126/scisignal.2004088
Gonzalez-Nieves R, DeSantis AI, Cutler ML (2013) Rsu1 contributes to regulation of cell adhesion and spreading by PINCH1-dependent and-independent mechanisms. J Cell Commun Signal 7(4):279–293. https://doi.org/10.1007/s12079-013-0207-5
Hannigan G, Troussard AA, Dedhar S (2005) Integrin-linked kinase: a cancer therapeutic target unique among its ILK. Nat Rev Cancer 5(1):51–63. https://doi.org/10.1038/nrc1524
Huang A-H, Pan S-H, Chang W-H, Hong Q-S, Chen JJW, Sung-Liang Yu (2015) PARVA promotes metastasis by modulating ILK signalling pathway in lung adenocarcinoma. PLoS ONE 10(3):e0118530–e0118530. https://doi.org/10.1371/journal.pone.0118530
Huang C, Li Y, Li Z, Yang Xu, Li Na, Ge Yi, Dong J et al (2019) LIMS1 promotes pancreatic cancer cell survival under oxygen-glucose deprivation conditions by enhancing HIF1A protein translation. Clin Cancer Res 25(13):4091–4103. https://doi.org/10.1158/1078-0432.CCR-18-3533
Jia Z (2015) Role of integrin-linked kinase in drug resistance of lung cancer. OncoTargets Ther 8(June):1561–1565. https://doi.org/10.2147/OTT.S81447
Johnson L, Mercer K, Greenbaum D, Bronson RT, Crowley D, Tuveson DA, Jacks T (2001) Somatic activation of the K-Ras oncogene causes early onset lung cancer in mice. Nature 410(6832):1111–1116. https://doi.org/10.1038/35074129
Kovalevich J, Tracy B, Langford D (2011) PINCH: more than just an adaptor protein in cellular response. J Cell Physiol 226(4):940–947. https://doi.org/10.1002/jcp.22437
Legate KR, Montañez E, Kudlacek O, Füssler R (2006) ILK, PINCH and parvin: the TIPP of integrin signalling. Nat Rev Mol Cell Biol 7(1):20–31. https://doi.org/10.1038/nrm1789
Liang CH, Chiu SY, Hsu IL, Wu YY, Tsai YT, Ke JY, Pan SH et al (2013) α-catulin drives metastasis by activating ILK and driving an Αvβ3 integrin signaling axis. Cancer Res 73(1):428–438. https://doi.org/10.1158/0008-5472.CAN-12-2095
Liu J, Costello PC, Pham N-A, Pintillie M, Jabali M, Sanghera J, Tsao M-S, Johnston MR (2006) Integrin-linked kinase inhibitor KP-392 demonstrates clinical benefitsin an orthotopic human non-small cell lung cancer model. J Thorac Oncol 1(8):771–779. https://doi.org/10.1016/S1556-0864(15)30405-6
Lööf J, Rosell J, Bratthäll C, Doré S, Starkhammar H, Zhang H, Sun X-F (2011) Impact of PINCH expression on survival in colorectal cancer patients. BMC Cancer 11(1):103. https://doi.org/10.1186/1471-2407-11-103
Maziveyi M, Alahari SK (2017) Cell matrix adhesions in cancer: the proteins that form the glue. Oncotarget 8(29):48471–48487. https://doi.org/10.18632/oncotarget.17265
McDonald PC, Fielding AB, Dedhar S (2008) Integrin-linked kinase – essential roles in physiology and cancer biology. J Cell Sci 121(19):3121–3132. https://doi.org/10.1242/jcs.017996
Mongroo PS, Johnstone CN, Naruszewicz I, Leung-Hagesteijn C, Sung RK, Carnio L, Rustgi AK, Hannigan GE (2004) β-parvin inhibits integrin-linked kinase signaling and is downregulated in breast cancer. Oncogene 23(55):8959–8970. https://doi.org/10.1038/sj.onc.1208112
Nagy Á, Lánczky A, Menyhárt O, Győrffy B (2018) Validation of MiRNA prognostic power in hepatocellular carcinoma using expression data of independent datasets. Sci Rep 8(1):9227. https://doi.org/10.1038/s41598-018-27521-y
Nikolopoulos SN, Turner CE (2001) Integrin-linked kinase (ILK) binding to paxillin LD1 motif regulates ILK localization to focal adhesions. J Biol Chem 276(26):23499–23505. https://doi.org/10.1074/jbc.M102163200
Noguchi S, Yasui Y, Iwasaki J, Kumazaki M, Yamada N, Naito S, Akao Y (2013) Replacement treatment with MicroRNA-143 and -145 induces synergistic inhibition of the growth of human bladder cancer cells by regulating PI3K/Akt and MAPK signaling pathways. Cancer Lett 328(2):353–361. https://doi.org/10.1016/j.canlet.2012.10.017
Porcheri C, Suter U, Jessberger S (2014) Dissecting integrin-dependent regulation of neural stem cell proliferation in the adult brain. J Neurosci 34(15):5222–5232. https://doi.org/10.1523/JNEUROSCI.4928-13.2014
Dos Santos N, Habibi G, Wang M, Dunn S (2006) Inhibition of integrin-linked kinase with QLT0267 suppresses the production of urokinase plasminogen activator: a key feature of recurrent breast cancer. Cancer Res 66(8 Supplement):802–802
Schaefer CF, Anthony K, Krupa S, Buchoff J, Day M, Hannay T, Buetow KH (2009) PID: the pathway interaction database. Nucleic Acids Res 37:D674–D679. https://doi.org/10.1093/nar/gkn653
Sepulveda JL, Wu C (2006) The parvins. Cell Mol Life Sci: CMLS 63(1):25–35. https://doi.org/10.1007/s00018-005-5355-1
Siegel RL, Miller KD, Jemal A (2019) Cancer statistics, 2019. CA: A Cancer J Clin 69(1):7–34. https://doi.org/10.3322/caac.21551
Stanchi F, Grashoff C, Yonga CF, Grall D, Fässler R, Van Obberghen-Schilling E (2009) Molecular dissection of the ILK-PINCH-parvin triad reveals a fundamental role for the ILK kinase domain in the late stages of focal-adhesion maturation. J Cell Sci 122(11):1800–1811. https://doi.org/10.1242/jcs.044602
Subramanian A, Tamayo P, Mootha VK, Mukherjee S, Ebert BL, Gillette MA, Paulovich A et al (2005) Gene set enrichment analysis: a knowledge-based approach for interpreting genome-wide expression profiles. Proc Natl Acad Sci 102(43):15545–15550. https://doi.org/10.1073/pnas.0506580102
Takanami I (2005) Increased expression of integrin-linked kinase is associated with shorter survival in non-small cell lung cancer. BMC Cancer 5(January):1. https://doi.org/10.1186/1471-2407-5-1
Travis WD, Brambilla E, Nicholson AG, Yatabe Y, Austin JHM, Beasley MB, Chirieac LR (2015) The 2015 World Health Organization classification of lung tumors: impact of genetic, clinical and radiologic advances since the 2004 classification. J Thorac Oncol 10(9):1243–1260. https://doi.org/10.1097/JTO.0000000000000630
Tseng P-C, Chen C-L, Shan Y-S, Chang W-T, Liu H-S, Hong T-M, Hsieh C-Y, Lin S-H, Lin C-F (2014) An increase in integrin-linked kinase non-canonically confers NF-ΚB-mediated growth advantages to gastric cancer cells by activating ERK1/2. Cell Commun Signal: CCS 12(November):69. https://doi.org/10.1186/s12964-014-0069-3
Tsinias G, Nikou S, Papadas T, Pitsos P, Papadaki H, Bravou V (2018) High PINCH1 expression in human laryngeal carcinoma associates with poor prognosis. Anal Cell Pathol (Amsterdam) 2018(March):2989635. https://doi.org/10.1155/2018/2989635
Tsoumas D, Nikou S, Giannopoulou E, Tsaniras SC, Sirinian C, Maroulis I, Taraviras S, Zolota V, Kalofonos HP, Bravou V (2018) ILK expression in colorectal cancer is associated with EMT, cancer stem cell markers and chemoresistance. Cancer Genomics Proteom 15(2):127–141. https://doi.org/10.21873/cgp.20071
Wang-Rodriguez J, Dreilinger AD, Alsharabi GM, Rearden A (2002) The signaling adapter protein PINCH is up-regulated in the stroma of common cancers, notably at invasive edges. Cancer 95(6):1387–1395. https://doi.org/10.1002/cncr.10878
Wickström SA, Lange A, Montanez E, Fässler R (2010) The ILK/PINCH/parvin complex: the kinase is dead, long live the pseudokinase! EMBO J 29(2):281–291. https://doi.org/10.1038/emboj.2009.376
Widmaier M, Rognoni E, Radovanac K, Azimifar SB, Fässler R (2012) Integrin-linked kinase at a glance. J Cell Sci 125(8):1839–1843. https://doi.org/10.1242/jcs.093864
Wu C-F, Ng K-F, Chen C-S, Chang P-L, Chuang C-K, Weng W-H, Liao S-K, Pang S-T (2010) Expression of parvin-beta is a prognostic factor for patients with urothelial cell carcinoma of the upper urinary tract. Br J Cancer 103(6):852–860. https://doi.org/10.1038/sj.bjc.6605835
Xu H, Cao H, Xiao G (2016) Signaling via PINCH: functions, binding partners and implications in human diseases. Gene 594(1):10–15. https://doi.org/10.1016/j.gene.2016.08.039
Zacharia LC, Stylianopoulos T, Gkretsi V (2017) Ras suppressor-1 (RSU-1) in cancer cell metastasis: friend or foe? Crit Rev Oncogene 22(3–4):249–253. https://doi.org/10.1615/CritRevOncog.2018024231
Zhang Y, Chen Ka, Yizeng Tu, Chuanyue Wu (2004) Distinct roles of two structurally closely related focal adhesion proteins, α-parvins and β-parvins, in regulation of cell morphology and survival. J Biol Chem 279(40):41695–41705. https://doi.org/10.1074/JBC.M401563200
Zhao M, Gao Y, Wang L, Liu S, Han B, Ma L, Ling Y, Mao S, Wang X (2013) Overexpression of integrin-linked kinase promotes lung cancer cell migration and invasion via NF-ΚB-mediated upregulation of matrix metalloproteinase-9. Int J Med Sci 10(8):995–1002. https://doi.org/10.7150/ijms.5963
Zhao X, Xu Z, Wang Z, Wu Z, Gong Y, Zhou L, Xiang Y (2015) RNA silencing of integrin-linked kinase increases the sensitivity of the A549 lung cancer cell line to cisplatin and promotes its apoptosis. Mol Med Rep 12(1):960–966. https://doi.org/10.3892/mmr.2015.3471
Zheng CC, Hu HF, Hong P, Zhang QH, Xu WW, He QY, Li B (2019) Significance of integrin-linked kinase (ILK) in tumorigenesis and its potential implication as a biomarker and therapeutic target for human cancer. Am J Cancer Res 9(1):186–197
Acknowledgments
We thank the Advanced Light Microscopy Facility, University of Patras, for help with microscopy.
Funding
This work was supported by the Basic Research Grant "K. Karatheodori" (E658) of the University of Patras (to Vasiliki Bravou).
Author information
Authors and Affiliations
Contributions
SN: Investigation, Visualization, Writing of original draft; MA, CS, PC, IP, GN: Investigation; FDD: Formal analysis, GTS, HP, VZ, ZL, HPK: resources, writing, reviewing and editing; VB: Conceptualization, Methodology, Funding acquisition, Supervision, Visualization, Writing of original draft, reviewing &editing.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
The study was approved by the Institutional Ethics & Research Committee of the University Hospital of Patras (protocol number 364/01.08.2017). Since archival tissues were used in the study the informed consent process was waived. Mice were bred at the Center for Animal Models of Disease of the Department of Physiology at the Faculty of Medicine of University of Patras, Greece. The animal study was approved by the Veterinary Administration of the Prefecture of Western Greece (#118018/578/30.04.2014) and were conducted in accordance to Directive 2010/63/EU (http://eurlex.europa.eu/legal-content/EN/TXT/?uri=CELEX%3A32010L0063) as described by Agalioti et al. 2017.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Nikou, S., Arbi, M., Dimitrakopoulos, FI.D. et al. Integrin-linked kinase (ILK) regulates KRAS, IPP complex and Ras suppressor-1 (RSU1) promoting lung adenocarcinoma progression and poor survival. J Mol Hist 51, 385–400 (2020). https://doi.org/10.1007/s10735-020-09888-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10735-020-09888-3