Abstract
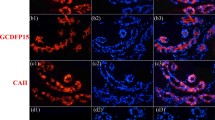
Eccrine sweat glands and hair follicles are two primary skin appendages that serve different functions. Although the two appendages exhibit unique morphological patterns in adults, it is difficult to distinguish them morphologically in the early stages of development and regeneration. To research and compare the development, differentiation and regeneration between eccrine sweat glands and hair follicles/pilosebaceous units, specific antigen markers must be found first. Human skin samples were fixed, paraffin-embedded, and cut. The expression of K5, K7, K8, K14, K27, K31, K73, AE13, α-smooth muscle actin (α-SMA), epithelial membrane antigen (EMA), carcinoembryonic antigen (CEA), Na+/K+-ATPase α and Na+–K+–2Cl cotransporter 1 (NKCC1) in eccrine sweat glands, hair follicles and sebaceous glands was detected by immunofluorescence staining. The results showed that eccrine sweat glands expressed K5, K7, K8, K14, K31, α-SMA, CEA, EMA, Na+/K+-ATPase α and NKCC1, but did not express K27, K73 or K31. Hair follicles expressed K5, K8, K14, K27, K31, K73, α-SMA and AE13, but did not express K7, CEA, Na+/K+-ATPase α or NKCC1. Sebaceous glands expressed K5, K14, K73, and EMA, but did not express K7, K8, K31, α-SMA, CEA, EMA, Na+/K+-ATPase α or NKCC1. We concluded that K7, CEA, Na+/K+-ATPase and NKCC1 can be used as specific markers for eccrine sweat glands, K27 and AE13 can be used as specific markers for hair follicles, and K73 can be used as a specific marker for pilosebaceous unit. These specific markers may contribute to differentiate between eccrine sweat glands and hair follicle/pilosebaceous units.



Similar content being viewed by others
Abbreviations
- α-SMA:
-
Α-smooth muscle actin
- CEA:
-
Carcinoembryonic antigen
- DAPI:
-
4′, 6-diamidino-2-phenylindole
- EMA:
-
Epithelial membrane antigen
- NKCC1:
-
Na+–K+–2Cl cotransporter 1
References
Biggs LC, Mikkola ML (2014) Early inductive events in ectodermal appendage morphogenesis. Semin Cell Dev Biol 25–26:11–21
Frances D, Niemann C (2012) Stem cell dynamics in sebaceous gland morphogenesis in mouse skin. Dev Biol 363:138–146
Fu XB, Sun TZ, Li XK, Sheng ZY (2005) Morphological and distribution characteristics of sweat glands in hypertrophic scar and their possible effects on sweat gland regeneration. Chin Med J (Engl) 118:186–191
Fu X, Fang L, Li X, Cheng B, Sheng Z (2006) Enhanced wound-healing quality with bone marrow mesenchymal stem cells autografting after skin injury. Wound Repair Regen 14:325–335
Fuchs E (2016) Epithelial skin biology: three decades of developmental biology, a hundred questions answered and a thousand new ones to address. Curr Top Dev Biol 116:357–374
Gaide O, Schneider P (2003) Permanent correction of an inherited ectodermal dysplasia with recombinant EDA. Nat Med 9:614–618
Haas M, Forbush B 3rd (2000) The Na-K-Cl cotransporter of secretory epithelia. Annu Rev Physiol 62:515–534
Hammarstrom S (1999) The carcinoembryonic antigen (CEA) family: structures, suggested functions and expression in normal and malignant tissues. Semin Cancer Biol 9:67–81
Jahoda CA, Reynolds AJ, Chaponnier C, Forester JC, Gabbiani G (1991) Smooth muscle alpha-actin is a marker for hair follicle dermis in vivo and in vitro. J Cell Sci 99(Pt 3):627–636
Li B, McKernan K, Shen W (2008) Spatial and temporal distribution patterns of Na-K-2Cl cotransporter in adult and developing mouse retinas. Vis Neurosci 25:109–123
Li H, Zhang X, Zeng S, Li X, Zhang B, Chen L, Lin C, Zhang M, Tang S, Fu X (2015) Combination of keratins and alpha-smooth muscle actin distinguishes secretory coils from ducts of eccrine sweat glands. Acta Histochem 117:275–278
Li H, Li X, Zhang B, Zhang M, Chen W, Tang S, Fu X (2016) Changes in keratins and alpha-smooth muscle actin during three-dimensional reconstitution of eccrine sweat glands. Cell Tissue Res 365:113–122
Li H, Chen L, Zhang M, Zhang B (2017) Foxa1 gene and protein in developing rat eccrine sweat glands. J Mol Histol 48:1–7
Lin CM, Yuan YP, Chen XC, Li HH, Cai BZ, Liu Y, Zhang H, Li Y, Huang K (2015) Expression of Wnt/beta-catenin signaling, stem-cell markers and proliferating cell markers in rat whisker hair follicles. J Mol Histol 46:233–240
Lu CP, Polak L, Rocha AS, Pasolli HA, Chen SC, Sharma N, Blanpain C, Fuchs E (2012) Identification of stem cell populations in sweat glands and ducts reveals roles in homeostasis and wound repair. Cell 150:136–150
Metze D, Luger TA (1996) Ultrastructural localization of carcinoembryonic antigen (CEA) glycoproteins and epithelial membrane antigen (EMA) in normal and neoplastic sweat glands. J Cutan Pathol 23:518–529
Mirmirani P, Tosti A, Goldberg L, Whiting D, Sotoodian B (2019) Frontal fibrosing alopecia: an emerging epidemic. Skin Appendage Disord 5:90–93
Moll I, Moll R (1992) Changes of expression of intermediate filament proteins during ontogenesis of eccrine sweat glands. J Invest Dermatol 98:777–785
Moll R, Moll I, Wiest W (1982) Changes in the pattern of cytokeratin polypeptides in epidermis and hair follicles during skin development in human fetuses. Differentiation 23:170–178
Moll R, Divo M, Langbein L (2008) The human keratins: biology and pathology. Histochem Cell Biol 129:705–733
Morth JP, Pedersen BP, Buch-Pedersen MJ, Andersen JP, Vilsen B, Palmgren MG, Nissen P (2011) A structural overview of the plasma membrane Na+, K+-ATPase and H+-ATPase ion pumps. Nat Rev Mol Cell Biol 12:60–70
Petschnik AE, Fell B, Kruse C, Danner S (2010) The role of alpha-smooth muscle actin in myogenic differentiation of human glandular stem cells and their potential for smooth muscle cell replacement therapies. Expert Opin Biol Ther 10:853–861
Saga K (2002) Structure and function of human sweat glands studied with histochemistry and cytochemistry. Prog Histochem Cytochem 37:323–386
Schon M, Benwood J, O’Connell-Willstaedt T, Rheinwald JG (1999) Human sweat gland myoepithelial cells express a unique set of cytokeratins and reveal the potential for alternative epithelial and mesenchymal differentiation states in culture. J Cell Sci 112(Pt 12):1925–1936
Shikiji T, Minami M, Inoue T, Hirose K, Oura H, Arase S (2003) Keratinocytes can differentiate into eccrine sweat ducts in vitro: involvement of epidermal growth factor and fetal bovine serum. J Dermatol Sci 33:141–150
Sotiropoulou PA, Blanpain C (2012) Development and homeostasis of the skin epidermis. Cold Spring Harb Perspect Biol 4:a008383
Taylor DK, Bubier JA, Silva KA, Sundberg JP (2012) Development, structure, and keratin expression in C57BL/6J mouse eccrine glands. Vet Pathol 49:146–154
Toth BI, Olah A, Szollosi AG, Czifra G, Biro T (2011) “Sebocytes’ makeup”: novel mechanisms and concepts in the physiology of the human sebaceous glands. Pflugers Arch 461:593–606
Wagner VP, Martins MD, Dillenburg CS, Meurer L, Castilho RM, Squarize CH (2015) Histogenesis of keratoacanthoma: histochemical and immunohistochemical study. Oral Surg Oral Med Oral Pathol Oral Radiol 119:310–317
Wang X, Wang X, Liu J, Cai T, Guo L, Wang S, Wang J, Cao Y, Ge J, Jiang Y, Tredget EE, Cao M, Wu Y (2016) Hair follicle and sebaceous gland de novo regeneration with cultured epidermal stem cells and skin-derived precursors. Stem Cells Transl Med 5:1695–1706
Acknowledgements
The manuscript was supported in part by the National Natural Science Foundation of China (81772102, 81471882).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
We declare we have no competing financial, personal or other relationships with other people or organizations.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Cao, L., Chen, L., Li, H. et al. Differential antigen expression between human eccrine sweat glands and hair follicles/pilosebaceous units. J Mol Hist 50, 335–342 (2019). https://doi.org/10.1007/s10735-019-09830-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10735-019-09830-2