Abstract
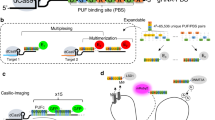
The biological function of chromatin bases on its spatial organization and dynamic activities in different situations. Labeling and tracing of genomic sequences has been a huge challenge in studying the spatial dynamics of chromatin. We reported the development of ‘all-in-one Casilio system (Aio-Casilio)’, a new system that enables the labeling of endogenous genomic loci. The Aio-Casilio system consists of the dCas9 protein, mClover fused with the Pumilio and FBF proteins RNA-binding domain (PUF domain) and an U6-sgRNA appended with multiple PUF-binding site(s). Here we showed that the Aio-Casilio system is robust tool for imaging of repetitive elements in telomeres and major satellite in N2A cells. Furthermore, we developed a PBTon–Aio-Casilio System, which enables the visualization of repetitive sequences in mES cells. However, this system failed to establish a labeled ES cell line when we attempted to establish a stable labeling cell line for living cell image. This Aio-Casilio imaging tool has potential to significantly improve the capacity to study the conformation of chromosomes in living cells.



Similar content being viewed by others
References
Abil Z, Denard CA, Zhao H (2014) Modular assembly of designer PUF proteins for specific post-transcriptional regulation of endogenous RNA. J Biol Eng 8:7. doi:10.1186/1754-1611-8-7
Anton T, Bultmann S, Leonhardt H, Markaki Y (2014) Visualization of specific DNA sequences in living mouse embryonic stem cells with a programmable fluorescent CRISPR/Cas system. Nucleus 5:163–172. doi:10.4161/nucl.28488
Barrangou R et al (2007) CRISPR provides acquired resistance against viruses in prokaryotes. Science 315:1709–1712. doi:10.1126/science.1138140
Belmont AS, Li G, Sudlow G, Robinett C (1999) Visualization of large-scale chromatin structure and dynamics using the lac operator/lac repressor reporter system. Methods Cell Biol 58:203–222
Chen Y, Varani G (2013) Engineering RNA-binding proteins for biology. FEBS J 280:3734–3754. doi:10.1111/febs.12375
Chen B et al (2013) Dynamic imaging of genomic loci in living human cells by an optimized CRISPR/Cas system. Cell 155:1479–1491. doi:10.1016/j.cell.2013.12.001
Cheng AW et al (2013) Multiplexed activation of endogenous genes by CRISPR-on, an RNA-guided transcriptional activator system. Cell Res 23:1163–1171 doi:10.1038/cr.2013.122
Cheng AW et al (2016) Casilio: a versatile CRISPR–Cas9–Pumilio hybrid for gene regulation and genomic labeling. Cell Res 26:254–257. doi:10.1038/cr.2016.3
Christian M et al (2010) Targeting DNA double-strand breaks with TAL effector nucleases. Genetics 186:757–761. doi:10.1534/genetics.110.120717
Cong L et al (2013) Multiplex genome engineering using CRISPR/Cas systems. Science 339:819–823. doi:10.1126/science.1231143
Cui C, Shu W, Li P (2016) Fluorescence in situ hybridization: cell-based genetic diagnostic and research applications. Front Cell Dev Biol 4:89 doi:10.3389/fcell.2016.00089
Deng W, Shi X, Tjian R, Lionnet T, Singer RH (2015) CASFISH: CRISPR/Cas9-mediated in situ labeling of genomic loci in fixed cells. Proc Natl Acad Sci USA 112:11870–11875. doi:10.1073/pnas.1515692112
Gilbert LA et al (2013) CRISPR-mediated modular RNA-guided regulation of transcription in eukaryotes. Cell 154:442–451. doi:10.1016/j.cell.2013.06.044
Jiang W, Marraffini LA (2015) CRISPR-Cas: new tools for genetic manipulations from bacterial immunity systems. Annu Rev Microbiol 69:209–228. doi:10.1146/annurev-micro-091014-104441
Konermann S et al (2013) Optical control of mammalian endogenous transcription and epigenetic states. Nature 500:472–476. doi:10.1038/nature12466
Lindhout BI, Fransz P, Tessadori F, Meckel T, Hooykaas PJ, van der Zaal BJ (2007) Live cell imaging of repetitive DNA sequences via GFP-tagged polydactyl zinc finger proteins. Nucleic Acids Res 35:e107. doi:10.1093/nar/gkm618
Ma H, Naseri A, Reyes-Gutierrez P, Wolfe SA, Zhang S, Pederson T (2015) Multicolor CRISPR labeling of chromosomal loci in human cells. Proc Natl Acad Sci USA 112:3002–3007. doi:10.1073/pnas.1420024112
Miller JC et al (2007) An improved zinc-finger nuclease architecture for highly specific genome editing. Nat Biotechnol 25:778–785. doi:10.1038/nbt1319
Miyanari Y, Ziegler-Birling C, Torres-Padilla ME (2013) Live visualization of chromatin dynamics with fluorescent TALEs. Nat Struct Mol Biol 20:1321–1324 doi:10.1038/nsmb.2680
Park TS, Han JY (2012) piggyBac transposition into primordial germ cells is an efficient tool for transgenesis in chickens. Proc Natl Acad Sci USA 109:9337–9341. doi:10.1073/pnas.1203823109
Perez EE et al (2008) Establishment of HIV-1 resistance in CD4+T cells by genome editing using zinc-finger nucleases. Nat Biotechnol 26:808–816. doi:10.1038/nbt1410
Perez-Pinera P et al (2013) RNA-guided gene activation by CRISPR–Cas9-based transcription factors. Nat Methods 10:973–976. doi:10.1038/nmeth.2600
Qi LS, Larson MH, Gilbert LA, Doudna JA, Weissman JS, Arkin AP, Lim WA (2013) Repurposing CRISPR as an RNA-guided platform for sequence-specific control of gene expression. Cell 152:1173–1183. doi:10.1016/j.cell.2013.02.022
Qin P et al (2017) Live cell imaging of low- and non-repetitive chromosome loci using CRISPR–Cas9. Nat Commun 8:14725 doi:10.1038/ncomms14725
Wang S, Su JH, Zhang F, Zhuang X (2016) An RNA-aptamer-based two-color CRISPR labeling system. Sci Rep 6:26857 doi:10.1038/srep26857
Yuan M, Webb E, Lemoine NR, Wang Y (2016) CRISPR–Cas9 as a powerful tool for efficient creation of oncolytic viruses. Viruses 8:72. doi:10.3390/v8030072
Zhang F, Cong L, Lodato S, Kosuri S, Church GM, Arlotta P (2011) Efficient construction of sequence-specific TAL effectors for modulating mammalian transcription. Nat Biotechnol 29:149–153. doi:10.1038/nbt.1775
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Zhang, S., Song, Z. Aio-Casilio: a robust CRISPR–Cas9–Pumilio system for chromosome labeling. J Mol Hist 48, 293–299 (2017). https://doi.org/10.1007/s10735-017-9727-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10735-017-9727-2