Abstract
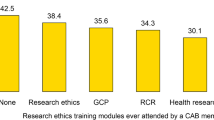
The community representative plays a very important role in an institutional review board (IRB) but there is sparse data about their understanding of their role in an IRB. This study was conducted to assess perceptions of community members serving on IRBs of one region in India. A validated questionnaire (Cohen’s kappa coefficient of 0.84) was administered to community members of IRBs in a prospective cross-sectional study. The questions related to demography, perceptions of their role in the IRB, experiences while serving on the IRBs, difficulties faced by them, and suggestions to improve their contributions. Of the 56 IRBs contacted, all 15 of the public institutions shared details of their community members, while only 26 of the 41 IRBs of private institutions responded. When questioned about why they joined the IRB, one third of the respondents said either that there was “no specific reason” for joining or that they accepted the invitation to serve on the insistence of other members of the IRBs or the head of their institution. All except one felt that community members were needed for optimal functioning. Six participants said that they did not review informed consent documents (ICD), and 10 were unaware that their presence was vital for quorum to be met. Twenty-eight said they participated actively in meetings and did not feel intimidated by the presence of clinicians. Twelve reported difficulties in understanding medical terminologies in ICDs. Although the majority of participants were aware about their role in an IRB, some did not review important documents such as ICDs. Restricted participation by private hospital IRBs was a striking observation in our study. There is a need to define eligibility criteria and develop targeted training modules for community members to ensure their effective contribution to effective oversight of clinical research.
Similar content being viewed by others
References
Allison, R., Abbott, L., & Wichman, A. (2008). Roles and experiences of non-scientist institutional review board members at the National Institutes of Health. IRB, 30(5), 8–13.
Anderson, E. (2006). A qualitative study of non-affiliated, non-scientist institutional review board members. Accountability in Research, 13(2), 135–155.
Council for International Organizations of Medical Sciences (CIOMS). (2002). International ethical guidelines for biomedical research involving human subjects. Geneva, Switzerland: Retrieved October 5, 2014 from http://www.cioms.ch/publications/layout_guide2002.pdf.
Ethics committee registration. Central Drugs Standard Control Organization. Retrieved January 15, 2014 from http://www.cdsco.nic.in/forms/list.aspx?lid=1859&Id=1.
Indian Council of Medical Research (ICMR). (2006). Ethical guidelines for biomedical research on human participants. Retrieved October 5, 2014 from http://www.icmr.nic.in/ethical_guidelines.pdf.
International Conference on Harmonization, Guideline for Good Clinical Practice. (1996). Retrieved October 5, 2014 from http://www.ich.org/fileadmin/Public_Web_Site/ICH_Products/Guidelines/Efficacy/E6/E6_R1_Guideline.pdf.
Jadhav, M., & Bhatt, A. (2013). Ethics in clinical research in India: A survey of clinical research professionals’ perceptions. Perspectives in Clinical Research, 4(1), 4–8.
Klitzman, R. (2012). Institutional review board community members: Who are they, what do they do, and whom do they represent? Academic Medicine, 87(7), 975–981.
Lemmens, T., & Thompson, A. (2001). Noninstitutional commercial review boards in North America: A critical appraisal and comparison with IRBs. IRB, 23(2), 1–12.
Lidz, C., Simon, L., Seligowski, A., Myers, S., Gardner, W., Candilis, P., et al. (2012). The participation of community members on medical institutional review boards. Journal of Empirical Research on Human Research Ethics, 7(1), 1–6.
Operational guidelines for ethics committees that review biomedical research, TDR/ETHICS/2000.1.Retrieved October 5, 2014 from http://www.fercap-sidcer.org/publications/pdf/201202/FERCAP-16-WHO%20EC%20Operational%20Guidelines.pdf.
Schedule, Y. (Drugs and Cosmetic Act 1940; amendment 20th January 2005). Retrieved October 5, 2014 from http://dbtbiosafety.nic.in/act/schedule_y.pdf.
Sengupta, S., & Lo, B. (2003). The roles and experiences of nonaffiliated and non-scientist members of institutional review boards. Academic Medicine, 78(2), 212–218.
Title 45 Code of Federal Regulations, Pt. 46.107(d). (2009 ed). Protection of human subjects, Retrieved October 5, 2014 from http://www.hhs.gov/ohrp/policy/ohrpregulations.pdf.
University of Southern California Office for the Protection of Research Subjects. (2013). What it takes to be an IRB community member. Retrieved October 5, 2014 from https://oprs.usc.edu/files/2013/05/Community-Member-Booklet-5.1.13.pdf.
Viera, A., & Garrett, J. (2005). Understanding interobserver agreement: The kappa statistic. Family Medicine, 37(5), 360–363.
World Medical Association Declaration of Helsinki. (2008). Ethical principles for medical research involving human subjects. Retrieved October 5, 2014 from http://www.wma.net/en/30publications/10policies/b3/17c.pdf.
Acknowledgments
The authors acknowledge with gratitude the contribution of Dr. Kannan Sridharan in the statistical analysis of the study data.
Conflict of interest
None.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kuyare, M.S., Marathe, P.A., Kuyare, S.S. et al. Perceptions and Experiences of Community Members Serving on Institutional Review Boards: A Questionnaire Based Study. HEC Forum 27, 61–77 (2015). https://doi.org/10.1007/s10730-014-9263-3
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10730-014-9263-3