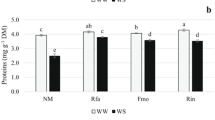
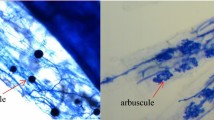
Abstract
Drought is one of the important factors limiting crop growth. Plants can enhance resistance to various stressors by forming symbiotic relationships with arbuscular mycorrhizal fungi (AMF). However, the regulatory mechanism of AMF on the drought tolerance of kenaf remains unclear. Therefore, we studied the effects of inoculating Claroideoglomus etunicatum (C. etunicatum, Ce) on the growth, gaseous exchange, antioxidant enzymes activity, osmotic regulatory substances, endogenous hormones, elemental content, and drought resistance related gene expression in kenaf under different water conditions, as well as the changes in soil enzymes after inoculation with Claroideoglomus etunicatum (Ce). The results showed that the biomass of kenaf inoculated with AMF significantly increased under drought conditions; For the aboveground parts of kenaf, inoculation with AMF improved the gas exchange parameters, and synthesized more osmotic regulatory substances to resist drought stress. At the same time, AMF also enhanced the scavenging ability of reactive oxygen species and reducing the cell damage caused by ROS; For the root of kenaf, AMF promoted the root development, enhanced the ability of plant element and water absorption, and significantly increased the content of IAA and ABA in the roots of kenaf under drought stress, reaching 26.45% and 6.69%, respectively. The content of globulin-related soil proteins (GRSP) were increased after inoculation with AMF, and the soil enzymes activity was improved, making the soil easier to maintain water, for the growth of kenaf; Furthermore AMF regulate the expression of aquaporin related genes (HcPIP1;2, HcPIP2;7) and improved the tolerance of kenaf to drought. Our study is not limited to the direct effects of AMF on plants, but also indirectly affects plants by analyzing the effects of AMF on the physicochemical properties of soil in plant roots. These results indicate that AMF played an important role to promote the growth of kenaf under drought conditions.










Similar content being viewed by others
Data availability
The data generated and/or analyzed during the current study are available from the corresponding author upon reasonable request.
References
Akter S, Khan MS, Smith EN, Flashman E (2021) Measuring ROS and redox markers in plant cells. RSC Chem Biol 2(5):1384–1401. https://doi.org/10.1039/d1cb00071c
Alzahrani Y, Rady MM (2019) Compared to antioxidants and polyamines, the role of maize grain-derived organic biostimulants in improving cadmium tolerance in wheat plants. Ecotoxicol Environ Saf 182:109378. https://doi.org/10.1016/j.ecoenv.2019.109378
Anzano A, Bonanomi G, Mazzoleni S, Lanzotti V (2021) Plant metabolomics in biotic and abiotic stress: a critical overview. Phytochem Rev 21(2):503–524. https://doi.org/10.1007/s11101-021-09786-w
Ayub M, Ashraf MY, Kausar A, Saleem S, Anwar S, Altay V, Ozturk M (2020) Growth and physio-biochemical responses of maize (Zea mays L.) to drought and heat stresses. Plant Biosystems - Int J Dealing all Aspects Plant Biology 155(3):535–542. https://doi.org/10.1080/11263504.2020.1762785
Bahadur A, Batool A, Nasir F, Jiang S, Mingsen Q, Zhang Q, Pan J, Liu Y, Feng H (2019) Mechanistic insights into Arbuscular Mycorrhizal Fungi-mediated Drought stress tolerance in plants. Int J Mol Sci 20(17):4199–4216. https://doi.org/10.3390/ijms20174199
Brookbank BP, Patel J, Gazzarrini S, Nambara E (2021) Role of basal ABA in Plant Growth and Development. Genes (Basel) 12(12):1936–1957. https://doi.org/10.3390/genes12121936
Chandrasekaran M, Paramasivan M (2022) Arbuscular mycorrhizal fungi and antioxidant enzymes in ameliorating drought stress: a meta-analysis. Plant Soil 480(1–2):295–303. https://doi.org/10.1007/s11104-022-05582-3
Chareesri A, De Deyn GB, Sergeeva L, Polthanee A, Kuyper TW (2020) Increased arbuscular mycorrhizal fungal colonization reduces yield loss of rice (Oryza sativa L.) under drought. Mycorrhiza 30(2–3):315–328. https://doi.org/10.1007/s00572-020-00953-z
Chen P, Chen T, Li Z, Jia R, Luo D, Tang M, Lu H, Hu Y, Yue J, Huang Z (2020a) Transcriptome analysis revealed key genes and pathways related to cadmium-stress tolerance in Kenaf (Hibiscus cannabinus L). Ind Crops Prod 158:112970. https://doi.org/10.1016/j.indcrop.2020.112970
Chen W, Meng P, Feng H, Wang C (2020b) Effects of Arbuscular Mycorrhizal Fungi on growth and physiological performance of Catalpa bungei C.A.Mey. Under Drought stress. Forests 11(10):1117–1145. https://doi.org/10.3390/f11101117
Chen Y, Yao Z, Sun Y, Wang E, Tian C, Sun Y, Liu J, Sun C, Tian L (2022) Current studies of the effects of Drought stress on Root exudates and Rhizosphere microbiomes of Crop Plant species. Int J Mol Sci 23(4):2374–2384. https://doi.org/10.3390/ijms23042374
Cheng XF, Wu HH, Zou YN, Wu QS, Kuca K (2021) Mycorrhizal response strategies of trifoliate orange under well-watered, salt stress, and waterlogging stress by regulating leaf aquaporin expression. Plant Physiol Biochem 162:27–35. https://doi.org/10.1016/j.plaphy.2021.02.026
Choudhary S, Wani KI, Naeem M, Khan MMA, Aftab T (2022) Cellular responses, osmotic adjustments, and role of osmolytes in providing salt stress resilience in higher plants: polyamines and nitric oxide crosstalk. J Plant Growth Regul 42(2):539–553. https://doi.org/10.1007/s00344-022-10584-7
Choudhury S, Moulick D, Ghosh D, Soliman M, Alkhedaide A, Gaber A, Hossain A (2022) Drought-Induced oxidative stress in Pearl Millet (Cenchrus americanus L.) at Seedling Stage: Survival mechanisms through Alteration of Morphophysiological and antioxidants Activity. Life 12(8):1171–1183. https://doi.org/10.3390/life12081171
Choukri H, Hejjaoui K, El-Baouchi A, El haddad N, Smouni A, Maalouf F, Thavarajah D, Kumar S (2020) Heat and Drought stress impact on phenology, Grain Yield, and Nutritional Quality of Lentil (Lens culinaris Medikus). Front Nutr 7:596307. https://doi.org/10.3389/fnut.2020.596307
Clarke RC, Merlin MD (2017) Cannabis domestication, breeding history, Present-Day Genetic Diversity, and future prospects. CRC Crit Rev Plant Sci 35(5–6):293–327. https://doi.org/10.1080/07352689.2016.1267498
Fang Y, Xiong L (2015) General mechanisms of drought response and their application in drought resistance improvement in plants. Cell Mol Life Sci 72(4):673–689. https://doi.org/10.1007/s00018-014-1767-0
Friero I, Alarcón MV, Gordillo L, Salguero J (2022) Abscisic acid is involved in several processes associated with root system architecture in maize. Acta Physiol Plant 44(3):28. https://doi.org/10.1007/s11738-022-03360-3
Furutani R, Ohnishi M, Mori Y, Wada S, Miyake C (2022) The difficulty of estimating the electron transport rate at photosystem I. J Plant Res 135(4):565–577. https://doi.org/10.1007/s10265-021-01357-6
Gachene CKK, Karuma AN, Baaru MW (2015) Climate Change and Crop Yield in Sub-Saharan Africa. In: Gachene CKK, Karuma AN, Baaru MW (eds) Sustainable Intensification to Advance Food Security and Enhance Climate Resilience in Africa. pp 165–183. https://doi.org/10.1007/978-3-319-09360-4_8
Hamberger B, Shen Z, Zhang Y-h, Zhang L, Li Y, Sun Y-d, Li Z-y (2020) Changes in the distribution of endogenous hormones in Phyllostachys edulis ‘Pachyloen’ during bamboo shooting. PLoS ONE 15(12):e0241806. https://doi.org/10.1371/journal.pone.0241806
Hasanuzzaman M, Nahar K, Anee TI, Fujita M (2017) Glutathione in plants: biosynthesis and physiological role in environmental stress tolerance. Physiol Mol Biol Plants 23(2):249–268. https://doi.org/10.1007/s12298-017-0422-2
He J-D, Chi G-G, Zou Y-N, Shu B, Wu Q-S, Srivastava AK, Kuča K (2020a) Contribution of glomalin-related soil proteins to soil organic carbon in trifoliate orange. Appl Soil Ecol 154:103592. https://doi.org/10.1016/j.apsoil.2020.103592
He J-D, Zou Y-N, Wu Q-S, Kuča K (2020b) Mycorrhizas enhance drought tolerance of trifoliate orange by enhancing activities and gene expression of antioxidant enzymes. Sci Hort 262:108745–108752. https://doi.org/10.1016/j.scienta.2019.108745
Hsu PK, Dubeaux G, Takahashi Y, Schroeder JI (2021) Signaling mechanisms in abscisic acid-mediated stomatal closure. Plant J 105(2):307–321. https://doi.org/10.1111/tpj.15067
Huang D, Ma M, Wang Q, Zhang M, Jing G, Li C, Ma F (2020) Arbuscular mycorrhizal fungi enhanced drought resistance in apple by regulating genes in the MAPK pathway. Plant Physiol Biochem 149:245–255. https://doi.org/10.1016/j.plaphy.2020.02.020
Huang D, Wang Q, Zou Y, Ma M, Jing G, Ma F, Li C (2021) Silencing MdGH3-2/12 in apple reduces cadmium resistance via the regulation of AM colonization. Chemosphere 269:129407. https://doi.org/10.1016/j.chemosphere.2020.129407
Irato P, Santovito G (2021) Enzymatic and non-enzymatic molecules with antioxidant function. Antioxid (Basel) 10(4):10579–10582. https://doi.org/10.3390/antiox10040579
Jing S, Li Y, Zhu L, Su J, Yang T, Liu B, Ma B, Ma F, Li M, Zhang M (2022) Transcriptomics and metabolomics reveal effect of arbuscular mycorrhizal fungi on growth and development of apple plants. Front Plant Sci 13:1052464–1052479. https://doi.org/10.3389/fpls.2022.1052464
Jongen M, Albadran B, Beyschlag W, Unger S (2022) Can arbuscular mycorrhizal fungi mitigate drought stress in annual pasture legumes? Plant Soil 472(1–2):295–310. https://doi.org/10.1007/s11104-021-05233-z
Kapoor D, Bhardwaj S, Landi M, Sharma A, Ramakrishnan M, Sharma A (2020) The impact of Drought in Plant Metabolism: how to exploit tolerance mechanisms to increase crop production. Appl Sci 10(16):5692–5710. https://doi.org/10.3390/app10165692
Kosakivska IV, Babenko LM, Romanenko KO, Korotka IY, Potters G (2020) Molecular mechanisms of plant adaptive responses to heavy metals stress. Cell Biol Int 45(2):258–272. https://doi.org/10.1002/cbin.11503
Kumar A, Lin H, Li Q, Ruan Y, Cousins D, Li F, Gao S, Jackson K, Wen J, Murray JD, Xu P (2022) Anthocyanin pigmentation as a quantitative visual marker for arbuscular mycorrhizal fungal colonization of Medicago truncatula roots. New Phytol 236(5):1988–1998. https://doi.org/10.1111/nph.18504
Li C, Jia Z, Zhai L, Zhang B, Peng X, Liu X, Zhang J (2021) Effects of Mineral-Solubilizing microorganisms on Root Growth, Soil Nutrient Content, and enzyme activities in the Rhizosphere Soil of Robinia pseudoacacia. Forests 12(1):60–71. https://doi.org/10.3390/f12010060
Li Y, Chu Y, Yao K, Shi C, Deng X, Lin J, Rennenberg H (2022a) Response of sugar metabolism in the cotyledons and roots of Ricinus communis subjected to salt stress. Plant Biol 25(1):62–71. https://doi.org/10.1111/plb.13475
Li Z, Zhang Y, Liu C, Gao Y, Han L, Chu H (2022b) Arbuscular mycorrhizal fungi contribute to reactive oxygen species homeostasis of Bombax ceiba L. under drought stress. Front Microbiol 13:991781. https://doi.org/10.3389/fmicb.2022.991781
Lips SJJ, van Dam JEG (2013) Kenaf Fibre Crop for Bioeconomic Industrial Development. In Kenaf: A Multi-Purpose Crop for Several Industrial Applications: New insights from the Biokenaf Project. pp 105–143. https://doi.org/10.1007/978-1-4471-5067-1_6
Liu C-Y, Hao Y, Wu X-L, Dai F-J, Abd-Allah EF, Wu Q-S, Liu S-R (2023) Arbuscular mycorrhizal fungi improve drought tolerance of tea plants via modulating root architecture and hormones. Plant Growth Regul 102:13–22. https://doi.org/10.1007/s10725-023-00972-8
Liyanage NMN, Chandrasekara BCHWM, Bandaranayake PCG (2021) A CTAB protocol for obtaining high-quality total RNA from cinnamon (Cinnamomum zeylanicum Blume). 3 Biotech 11(4):201. https://doi.org/10.1007/s13205-021-02756-1
Ma Y, Dias MC, Freitas H (2020) Drought and Salinity stress responses and Microbe-Induced Tolerance in plants. Front Plant Sci 11:591911. https://doi.org/10.3389/fpls.2020.591911
Malekzadeh E, Aliasgharzad N, Majidi J, Abdolalizadeh J, Aghebati-Maleki L (2016) Contribution of glomalin to pb sequestration by arbuscular mycorrhizal fungus in a sand culture system with clover plant. Eur J Soil Biol 74:45–51. https://doi.org/10.1016/j.ejsobi.2016.03.003
Mansoor S, Ali Wani O, Lone JK, Manhas S, Kour N, Alam P, Ahmad A, Ahmad P (2022) Reactive oxygen species in plants: from source to Sink. Antioxid (Basel) 11(2):225–238. https://doi.org/10.3390/antiox11020225
Maphuhla NG, Lewu FB, Oyedeji OO (2020) The effects of Physicochemical parameters on Analysed Soil enzyme activity from Alice Landfill Site. Int J Environ Res Public Health 18(1):221–235. https://doi.org/10.3390/ijerph18010221
Mazat JP, Devin A, Ransac S (2020) Modelling mitochondrial ROS production by the respiratory chain. Cell Mol Life Sci 77(3):455–465. https://doi.org/10.1007/s00018-019-03381-1
Meher, Shivakrishna P, Reddy KA, Rao DM (2018) Effect of PEG-6000 imposed drought stress on RNA content, relative water content (RWC), and chlorophyll content in peanut leaves and roots. Saudi J Biol Sci 25(2):285–289. https://doi.org/10.1016/j.sjbs.2017.04.008
Mohammadi Alagoz S, Zahra N, Hajiaghaei Kamrani M, Asgari Lajayer B, Nobaharan K, Astatkie T, Siddique KHM, Farooq M (2022) Role of Root Hydraulics in Plant Drought Tolerance. J Plant Growth Regul 42(10):6228–6243. https://doi.org/10.1007/s00344-022-10807-x
Morales M, Munne-Bosch S (2019) Malondialdehyde: facts and artifacts. Plant Physiol 180(3):1246–1250. https://doi.org/10.1104/pp.19.00405
Muhammad I, Shalmani A, Ali M, Yang QH, Ahmad H, Li FB (2020) Mechanisms regulating the dynamics of Photosynthesis under Abiotic stresses. Front Plant Sci 11:615942. https://doi.org/10.3389/fpls.2020.615942
Mukarram M, Choudhary S, Kurjak D, Petek A, Khan MMA (2021) Drought: sensing, signalling, effects and tolerance in higher plants. Physiol Plant 172(2):1291–1300. https://doi.org/10.1111/ppl.13423
Ozturk M, Turkyilmaz Unal B, García-Caparrós P, Khursheed A, Gul A, Hasanuzzaman M (2020) Osmoregulation and its actions during the drought stress in plants. Physiol Plant 172(2):1321–1335. https://doi.org/10.1111/ppl.13297
Patanè C, Cosentino SL (2013) Yield, water use and radiation use efficiencies of kenaf (Hibiscus cannabinus L.) under reduced water and nitrogen soil availability in a semi-arid Mediterranean area. Eur J Agron 46:53–62. https://doi.org/10.1016/j.eja.2012.12.002
Qamer Z, Chaudhary MT, Du X, Hinze L, Azhar MT (2021) Review of oxidative stress and antioxidative defense mechanisms in Gossypium hirsutum L. in response to extreme abiotic conditions. J Cotton Res 4(1):9. https://doi.org/10.1186/s42397-021-00086-4
Qian K, Wang L, Yin N (2012) Effects of AMF on soil enzyme activity and carbon sequestration capacity in reclaimed mine soil. Int J Min Sci Technol 22(4):553–557. https://doi.org/10.1016/j.ijmst.2012.01.019
Qin M, Zhang Q, Pan J, Jiang S, Liu Y, Bahadur A, Peng Z, Yang Y, Feng H (2019) Effect of arbuscular mycorrhizal fungi on soil enzyme activity is coupled with increased plant biomass. Eur J Soil Sci 71(1):84–92. https://doi.org/10.1111/ejss.12815
Quiroga G, Erice G, Ding L, Chaumont F, Aroca R, Ruiz-Lozano JM (2019) The arbuscular mycorrhizal symbiosis regulates aquaporins activity and improves root cell water permeability in maize plants subjected to water stress. Plant Cell Environ 42(7):2274–2290. https://doi.org/10.1111/pce.13551
Quiroga G, Erice G, Aroca R, Delgado-Huertas A, Ruiz-Lozano JM (2020) Elucidating the possible involvement of Maize aquaporins and Arbuscular Mycorrhizal Symbiosis in the Plant Ammonium and Urea Transport under Drought stress conditions. Plants 9(2):148–164. https://doi.org/10.3390/plants9020148
Rabiei Z, Hosseini SJ, Pirdashti H, Hazrati S (2020) Physiological and biochemical traits in coriander affected by plant growth-promoting rhizobacteria under salt stress. Heliyon 6(10):5321–5329. https://doi.org/10.1016/j.heliyon.2020.e05321
Rajametov SN, Yang EY, Cho MC, Chae SY, Jeong HB, Chae WB (2021) Heat-tolerant hot pepper exhibits constant photosynthesis via increased transpiration rate, high proline content and fast recovery in heat stress condition. Sci Rep 11(1):14328. https://doi.org/10.1038/s41598-021-93697-5
Rapparini F, Peñuelas J (2014) Mycorrhizal Fungi to Alleviate Drought Stress on Plant Growth. In: Rapparini F, Peñuelas J (eds) Use of Microbes for the Alleviation of Soil Stresses, vol 1. pp 21–42. https://doi.org/10.1007/978-1-4614-9466-9_2
Recchia GH, Konzen ER, Cassieri F, Caldas DGG, Tsai SM (2018) Arbuscular Mycorrhizal Symbiosis leads to Differential Regulation of Drought-responsive genes in tissue-specific Root cells of Common Bean. Front Microbiol 9:1339. https://doi.org/10.3389/fmicb.2018.01339
Ren J, Liu X, Yang W, Yang X, Li W, Xia Q, Li J, Gao Z, Yang Z (2021) Rhizosphere soil properties, microbial community, and enzyme activities: short-term responses to partial substitution of chemical fertilizer with organic manure. J Environ Manage 299:113650. https://doi.org/10.1016/j.jenvman.2021.113650
Salvi P, Manna M, Kaur H, Thakur T, Gandass N, Bhatt D, Muthamilarasan M (2021) Phytohormone signaling and crosstalk in regulating drought stress response in plants. Plant Cell Rep 40(8):1305–1329. https://doi.org/10.1007/s00299-021-02683-8
Santander C, Ruiz A, Garcia S, Aroca R, Cumming J, Cornejo P (2020) Efficiency of two arbuscular mycorrhizal fungal inocula to improve saline stress tolerance in lettuce plants by changes of antioxidant defense mechanisms. J Sci Food Agric 100(4):1577–1587. https://doi.org/10.1002/jsfa.10166
Seleiman MF, Al-Suhaibani N, Ali N, Akmal M, Alotaibi M, Refay Y, Dindaroglu T, Abdul-Wajid HH, Battaglia ML (2021) Drought stress impacts on plants and different approaches to alleviate its adverse effects. Plants (Basel) 10(2):259–283. https://doi.org/10.3390/plants10020259
Sheteiwy MS, Abd Elgawad H, Xiong YC, Macovei A, Brestic M, Skalicky M, Shaghaleh H, Alhaj Hamoud Y, El-Sawah AM (2021a) Inoculation with Bacillus amyloliquefaciens and mycorrhiza confers tolerance to drought stress and improve seed yield and quality of soybean plant. Physiol Plant 172(4):2153–2169. https://doi.org/10.1111/ppl.13454
Sheteiwy MS, Ali DFI, Xiong Y-C, Brestic M, Skalicky M, Hamoud YA, Ulhassan Z, Shaghaleh H, AbdElgawad H, Farooq M, Sharma A, El-Sawah AM (2021b) Physiological and biochemical responses of soybean plants inoculated with arbuscular mycorrhizal fungi and Bradyrhizobium under drought stress. BMC Plant Biol 21(1):195. https://doi.org/10.1186/s12870-021-02949-z
Sheteiwy MS, El-Sawah AM, Kobae Y, Basit F, Holford P, Yang H, El-Keblawy A, Abdel-Fattah GG, Wang S, Araus JL, Korany SM, Alsherif EA, AbdElgawad H (2023) The effects of microbial fertilizers application on growth, yield and some biochemical changes in the leaves and seeds of guar (Cyamopsis tetragonoloba L). Food Res Int 172:113122. https://doi.org/10.1016/j.foodres.2023.113122
Singh D, Laxmi A (2015) Transcriptional regulation of drought response: a tortuous network of transcriptional factors. Front Plant Sci 6:895. https://doi.org/10.3389/fpls.2015.00895
Sun H, Liu S, Chen K, Li G (2021) Spectrophotometric determination of chlorophylls in different solvents related to the leaf traits of the main tree species in Northeast China. IOP Conf Series: Earth Environ Sci 836(1):12008–12018. https://doi.org/10.1088/1755-1315/836/1/012008
Tang M, Yue J, Huang Z, Hu Y, Li Z, Luo D, Cao S, Zhang H, Pan J, Wu X, Wu Q, Chen P (2022) Physiological and DNA methylation analysis provides epigenetic insights into chromium tolerance in kenaf. Environ Exp Bot 194:104684. https://doi.org/10.1016/j.envexpbot.2021.104684
Tian H, Jia Z, Liu W, Wei X, Wang H, Bao G, Li J, Zhou Q (2023) Effects of Arbuscular Mycorrhizal Fungi on Growth and Nutrient Accumulation of Oat under Drought conditions. Agronomy 13(10):2580–2592. https://doi.org/10.3390/agronomy13102580
Tiepo AN, Constantino LV, Madeira TB, Gonçalves LSA, Pimenta JA, Bianchini E, de Oliveira ALM, Oliveira HC, Stolf-Moreira R (2020) Plant growth-promoting bacteria improve leaf antioxidant metabolism of drought-stressed neotropical trees. Planta 251(4):83. https://doi.org/10.1007/s00425-020-03373-7
Vicente-Serrano SM, Peña-Angulo D, Beguería S, Domínguez-Castro F, Tomás-Burguera M, Noguera I, Gimeno-Sotelo L, El Kenawy A (2022) Global drought trends and future projections. Philosophical Trans Royal Soc A: Math Phys Eng Sci 380(2238):20210285. https://doi.org/10.1098/rsta.2021.0285
Wang D, Li F, Nong M (2017) Response of yield and water use efficiency to different irrigation levels at different growth stages of Kenaf and crop water production function. Agric Water Manage 179:177–183. https://doi.org/10.1016/j.agwat.2016.06.026
Wang B, Wang Y, Zhang J, Hu C, Jiang J, Li Y, Peng Z (2023) ROS-induced lipid peroxidation modulates cell death outcome: mechanisms behind apoptosis, autophagy, and ferroptosis. Arch Toxicol 97(6):1439–1451. https://doi.org/10.1007/s00204-023-03476-6
Wei H, Li X, He W, Kuang Y, Wang Z, Hu W, Tang M, Chen H (2023) Arbuscular mycorrhizal symbiosis enhances perennial ryegrass growth during temperature stress through the modulation of antioxidant defense and hormone levels. Ind Crops Prod 195:116412. https://doi.org/10.1016/j.indcrop.2023.116412
Windari EA, Ando M, Mizoguchi Y, Shimada H, Ohira K, Kagaya Y, Higashiyama T, Takayama S, Watanabe M, Suwabe K (2021) Two aquaporins, SIP1;1 and PIP1;2, mediate water transport for pollen hydration in the Arabidopsis pistil. Plant Biotechnol (Tokyo) 38(1):77–87. https://doi.org/10.5511/plantbiotechnology.20.1207a
Wu J, Wang J, Hui W, Zhao F, Wang P, Su C, Gong W (2022) Physiology of Plant Responses to Water Stress and related genes: a review. Forests 13(2):324–339. https://doi.org/10.3390/f13020324
Xiao Y, Chen L (2022) Arbuscular mycorrhizal fungi reduce potassium, cadmium and ammonium losses but increases nitrate loss under high intensity leaching events. BMC Plant Biol 22(1):365. https://doi.org/10.1186/s12870-022-03741-3
Xie Q, Essemine J, Pang X, Chen H, Cai W (2020) Exogenous application of abscisic acid to shoots promotes primary root cell division and elongation. Plant Sci 292:110385. https://doi.org/10.1016/j.plantsci.2019.110385
Xu J, Aifen T, Qi J, Wang Y (2020) Bast fibres. In: Xu J, Aifen T, Qi J, Wang Y (eds) Handbook of Natural Fibres. pp 71–92. https://doi.org/10.1016/b978-0-12-818398-4.00005-0
Yan Q, Li X, Xiao X, Chen J, Liu J, Lin C, Guan R, Wang D (2022) Arbuscular mycorrhizal fungi improve the growth and drought tolerance of Cinnamomum migao by enhancing physio-biochemical responses. Ecol Evol 12(7):e9091. https://doi.org/10.1002/ece3.9091
Yang Y, Chen Y, Li W (2008) Arbuscular mycorrhizal fungi infection in desert riparian forest and its environmental implications: a case study in the lower reach of Tarim River. Prog Nat Sci 18(8):983–991. https://doi.org/10.1016/j.pnsc.2008.02.009
Yang Y, He C, Huang L, Ban Y, Tang M (2017) The effects of arbuscular mycorrhizal fungi on glomalin-related soil protein distribution, aggregate stability and their relationships with soil properties at different soil depths in lead-zinc contaminated area. PLoS ONE 12(8):e0182264. https://doi.org/10.1371/journal.pone.0182264
Yang X, Lu M, Wang Y, Wang Y, Liu Z, Chen S (2021) Response mechanism of plants to Drought stress. Horticulturae 7(3):50. https://doi.org/10.3390/horticulturae7030050
Zhang Y, Li Y, Hassan MJ, Li Z, Peng Y (2020) Indole-3-acetic acid improves drought tolerance of white clover via activating auxin, abscisic acid and jasmonic acid related genes and inhibiting senescence genes. BMC Plant Biol 20(1):150. https://doi.org/10.1186/s12870-020-02354-y
Zia R, Nawaz MS, Siddique MJ, Hakim S, Imran A (2021) Plant survival under drought stress: implications, adaptive responses, and integrated rhizosphere management strategy for stress mitigation. Microbiol Res 242:126626. https://doi.org/10.1016/j.micres.2020.126626
Zou YN, Wu QS, Kuča K, Hu Y (2020) Unravelling the role of arbuscular mycorrhizal fungi in mitigating the oxidative burst of plants under drought stress. Plant Biol 23(S1):50–57. https://doi.org/10.1111/plb.13161
Zou YN, Qin QY, Ma WY, Zhou LJ, Wu QS, Xu YJ, Kuca K, Hashem A, Al-Arjani AF, Almutairi KF, Abd-Allah EF (2023) Metabolomics reveals arbuscular mycorrhizal fungi-mediated tolerance of walnut to soil drought. BMC Plant Biol 23(1):118. https://doi.org/10.1186/s12870-023-04111-3
Acknowledgements
This work was funded by the National Natural Science Foundation of China (No.31960368).
Funding
National Natural Science Foundation of China, https://doi.org/10.13039/501100001809.
Author information
Authors and Affiliations
Contributions
Peng Chen: Conceptualization, Writing-review & editing, Supervision, Project administration, Funding acquisition. G. X: Performed the experiments, Writing-original draft, Writing-review & editing, Methodology, Validation, Formalanalysis, Investigation. J. P, Muzammal Rehman: Revised the manuscript. X. L, S. C, C. W, X. W, C. C, J. N, M. W., R. L, R. L: Methodology, Investigation, Validation, Assisted in materials management. All the authors have read and approved the manuscript.
Corresponding author
Ethics declarations
Competing interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Xu, G., Pan, J., Rehman, M. et al. Arbuscular mycorrhizal fungi-mediated drought stress tolerance in kenaf (hibiscus cannabinus l.): a mechanistic approach. Plant Growth Regul (2024). https://doi.org/10.1007/s10725-024-01150-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10725-024-01150-0