Abstract
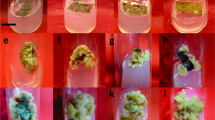
Forsythia saxatilis is an endangered species endemic to Korea. Here, we determined the effects of light quality on the growth and antioxidant activity of F. saxatilis. Six different light conditions were used: white (W10, control); combined light-emitting diodes (LED) of white (W) and far-red (Fr) at 10:1 (W10Fr1); combined LED of red (R) and blue (B) at 5:5 (R5B5); combined LED of R and B at 7:3 (R7B3); combined LED of R, green (G), and B at 7:1:2 (R7G1B2); and a combined LED of R, G, B, and Fr at 7:1:2:1 (R7G1B2Fr1). Forsythia saxatilis shoot formation was the highest with R7G1B2 at 89%, whereas the greatest growth-promoting effects—that is, increase in plant height, leaf length and width, and fresh and dry weights of shoots and roots—were achieved with W10Fr1. The total phenol and flavonoid contents were the highest with R7B3. The activities of 2,2-diphenyl-1-picrylhydrazyl (DPPH) and 2,2′-azino-bis(3-ethylbenzothiazoline-6-sulfonic acid) (ABTS) were the highest with R7B3 treatment. In summary, W10Fr1 treatment was found to be the most effective for the growth of F. saxatilis, whereas R7B3 was confirmed to be the most effective for maximizing antioxidant activity.






Similar content being viewed by others
References
Ahmad M, Lin C, Cashmore AR (1995) Mutations throughout an Arabidopsis blue-light photoreceptor impair blue-light-responsive anthocyanin accumulation and inhibition of hypocotyl elongation. Plant J 8:653–658. https://doi.org/10.1046/j.1365-313X.1995.08050653.x
Bae KH (2000) The medicinal plants of Korea. Kyo-Hak Publishing Co, Seoul, p 260
Bertram L, Lercari B (2000) Phytochrome A and phytochrome B1 control the acquisition of competence for shoot regeneration in tomato hypocotyl. Plant Cell Rep 19:604–609. https://doi.org/10.1007/s002990050780
Bian ZH, Yang QC, Liu WK (2015) Effects of light quality on the accumulation of phytochemicals in vegetables produced in controlled environments: a review. J Sci Food Agric 95:869–877. https://doi.org/10.1002/jsfa.6789
Blois MS (1958) Antioxidant determinations by the use of a stable free radical. Nature 181:1199–1200. https://doi.org/10.1038/1811199a0
Bravo L (1998) Polyphenols: chemistry, dietary sources, metabolism, and nutritional significance. Nutr Rev 56:317–333. https://doi.org/10.1111/j.1753-4887.1998.tb01670.x
Buer CS, Imin N, Djordjevic MA (2010) Flavonoids: new roles for old molecules. J Integr Plant Biol 52:98–111. https://doi.org/10.1111/j.1744-7909.2010.00905.x
Burritt DJ, Leung DW (1996) Organogenesis in cultured petiole explants of Begonia× erythrophylla: the timing and specificity of the inductive stimuli. J Exp Bot 47:557–567. https://doi.org/10.1093/jxb/47.4.557
Burritt DJ, Leung DW (2003) Adventitious shoot regeneration from Begonia× erythrophylla petiole sections is developmentally sensitive to light quality. Physiol Plant 118:289–296. https://doi.org/10.1034/j.1399-3054.2003.00083.x
Carvalho SD, Schwieterman ML, Abrahan CE, Colquhoun TA, Folta KM (2016) Light quality dependent changes in morphology, antioxidant capacity, and volatile production in sweet basil (Ocimum basilicum). Front Plant Sci 7:1328. https://doi.org/10.3389/fpls.2016.01328
Chen CC, Huang MY, Lin KH, Wong SL, Huang WD, Yang CM (2014) Effects of light quality on the growth, development and metabolism of rice seedlings (Oryza sativa L.). Res J Biotechnol 9:15–24
Choi SC, Kim S (2020) A chloroplast genome of Forsythia saxatilis (Nakai) Nakai, an endemic species in Korea. Mitochondrial DNA Part B 5:3725–3727. https://doi.org/10.1080/23802359.2020.1834889
Ciancolini A, Alignan M, Pagnotta MA, Vilarem G, Crinò P (2013) Selection of Italian cardoon genotypes as industrial crop for biomass and polyphenol production. Ind Crops Prod 51:145–151. https://doi.org/10.1016/j.indcrop.2013.08.069
Coley PD, Bryant JP, Chapin FS (1985) Resource availability and plant antiherbivore defense. Science 230:895–899. https://doi.org/10.1126/science.230.4728.895
Dao JP, Kouakou KL, Kouakou C, Cherif M, Ouedraogo MH, Koffi KK, Bi IAZ (2020) Effect of leafy and leafless greenwood, softwood and hardwood cuttings success of Garcinia kola (Heckel). Int J Agric Sci 11:897. https://doi.org/10.4236/as.2020.1110058
Erlund I (2004) Review of the flavonoids quercetin, hesperetin, and naringenin. dietary sources, bioactivities, bioavailability, and epidemiology. Nutr Res 24:851–874. https://doi.org/10.1016/j.nutres.2004.07.005
Gam DT, Khoi PH, Ngoc PB, Linh LK, Hung NK, Anh PTL, Ha CH (2020) LED lights promote growth and flavonoid accumulation of Anoectochilus roxburghii and are linked to the enhanced expression of several related genes. Plants 9:1344. https://doi.org/10.3390/plants9101344
Gupta SD, Jatothu B (2013) Fundamentals and applications of light-emitting diodes (LEDs) in in vitro plant growth and morphogenesis. Plant Biotechnol Rep 7:211–220. https://doi.org/10.1007/s11816-013-0277-0
Han SH, Kim DH, Kim GN, Byun JK (2011) Changes of leaf characteristics, pigment content and photosynthesis of Forsythia saxatilis under two different light intensities. J Korean Soc for Sci 100:609–615
Han SH, Kang HJ, Kim GN, Kim DH (2013) Optimal condition for pollen germination of rare and endangered Forsythia saxatilis. J Korean Soc for Sci 102(1):136–142
Haslam and Edwin (1989) Plant polyphenols: vegetable tannins revisited. CUP Archive
Hassanpour S, Naser M, Behrad E, Mehmandar FB (2011a) Plants and secondary metabolites (Tannins): A Review. Int J for Soil Eros 1:47–53
Hassanpour S, Sadaghian M, Maherisis N, Eshratkhah B, Chaichisemsari M (2011b) Effect of condensed tannin on controlling faecal protein excretion in nematode-infected sheep: in vivo study. J Amer Sci 7:896–900
Heo J, Lee C, Chakrabarty D, Paek K (2002) Growth responses of marigold and salvia bedding plants as affected by monochromic or mixture radiation provided by a light-emitting diode (LED). Plant Growth Regul 38:225–230
Herms DA, Mattson WJ (1992) The dilemma of plants: to grow or defend. Q Rev Biol 67:283–335. https://doi.org/10.1086/417659
Jin AR, Go YC, Young HK, Hye WL, Mi YL, Jae EC, Byoung SK (2010) Application of genetic marker and real-time polymerase chain reaction for discrimination between Forsythia viridissima and Forsythia suspensa. Biol Pharm Bull 33:1133–1137. https://doi.org/10.1248/bpb.33.1133
Jishi T (2018) LED lighting technique to control plant growth and morphology. In Smart Plant Factory, Springer
Johkan M, Shoji K, Goto F, Hashida SN, Yoshihara T (2010) Blue light-emitting diode light irradiation of seedlings improves seedling quality and growth after transplanting in red leaf lettuce. HortScience 45(12):1809–1814
Kadkade P, Seibert M (1977) Phytochrome-regulated organogenesis in lettuce tissue culture. Nature 270:49–50. https://doi.org/10.1038/270049a0
Khattak AB, Zeb A, Bibi N (2008) Impact of germination time and type of illumination on caroteinoid content, protein solubility and in-vitro protein digestibility of chickpea (Cicer arietinum L) sprouts. Food Chem 109:797–801. https://doi.org/10.1016/j.foodchem.2008.01.046
Kim MJ, Kim JY, Jung TK, Choi SW, Yoon KS (2006) Skin anti-aging effect of Forsythia viridissima L. extract. KSBB J 21:444–450
Kim MJ, Im KR, Yoon KS (2009) Anti-inflammatory effects of prescription extracts containing Forsythia viridissima L. J Soc. Cosmet. Sci. Korea. 35:277–285
Kim GN, Han SH, Kim DH, Yun CW, Shin SJ (2013) Optimum light intensity and fertilization effects on physiological activities of Forsythia saxatilis. J Korean Soc for Sci 102:372–381
Kim SH, Cho CH, Yoon HS, Jeon J, Kim SC (2018) Characterization of the complete chloroplast genome of Forsythia saxatilis (Oleaceae), a vulnerable calcicolus species endemic to Korea. Conserv Genet Resour 10:723–726. https://doi.org/10.1007/s12686-017-0910-2
Koes RE, Quattrocchio F, Mol JN (1994) The flavonoid biosynthetic pathway in plants: function and evolution. BioEssays 16:123–132. https://doi.org/10.1002/bies.950160209
Lee JY, Moon H, Kim CJ (2010) Effects of hydroxy pentacyclic triterpene acids from Forsythia viridissima on asthmatic responses to ovalbumin challenge in conscious guinea pigs. Biol Pharm Bull 33:230–237. https://doi.org/10.1248/bpb.33.230
Li Q, Kubota C (2009) Effects of supplemental light quality on growth and phytochemicals of baby leaf lettuce. Environ Exp Bot 67:59–64. https://doi.org/10.1016/j.envexpbot.2009.06.011
Liang XT (2003) Common traditional chinese medicine foundation research. Science Press, Beijing 1:315
Lim H, Lee JG, Lee SH, Kim YS, Kim HP (2008) Anti-inflammatory activity of phylligenin, a lignan from the fruits of Forsythia koreana, and its cellular mechanism of action. J Ethnopharmacol 118:113–117. https://doi.org/10.1016/j.jep.2008.03.016
Manivannan A, Soundararajan P, Halimah N, Ko CH, Jeong BR (2015) Blue LED light enhances growth, phytochemical contents, and antioxidant enzyme activities of Rehmannia glutinosa cultured in vitro. Hortic Environ Biotechnol 56:105–113. https://doi.org/10.1007/s13580-015-0114-1
Moon HK, Suk GY, Kim SC (1997) Micropropagation of a rare species, Forsythia saxatilis N. through tissue culture. J Korean Soc for Sci 86:430–434
Nam TG, Kim DO, Eom SH (2018) Effects of light sources on major flavonoids and antioxidant activity in common buckwheat sprouts. Food Sci Biotechnol 27:169–176
National Institute of Biological Sciences (2013) Endemic species of Korea. National Institute of Biological Sciences. Incheon, , Korea
Naznin MT, Lefsrud M, Gravel V, Hao X (2016) Different ratios of red and blue LED light effects on coriander productivity and antioxidant properties. VIII Int Symp Light Hortic 1134:223–230
Nozue H, Gomi M (2018) Usefulness of broad-spectrum white LEDs to envision future plant factory. Smart Plant Factory, Springer
Oh HE, Yoon A, Park YG (2021) Red light enhances the antioxidant properties and growth of Rubus hongnoensis. Plants 10:2589. https://doi.org/10.3390/plants10122589
Pezeshki SR, DeLaune RD (1994) Rooting of Baldcypress Cuttings. New for 8:381–386
Qu H, Zhang Y, Wang Y, Li B, Sun W (2010) Antioxidant and antibacterial activity of two compounds (forsythiaside and forsythin) isolated from Forsythia suspensa. J Pharm Pharmacol 60:261–266. https://doi.org/10.1211/jpp.60.2.0016
Quettier-Deleu C, Gressier B, Vasseur J, Dine T, Brunet C, Luyckx M, Trotin F (2000) Phenolic compounds and antioxidant activities of buckwheat (Fagopyrum esculentum Moench) hulls and flour. J Ethnopharmacol 72:35–42. https://doi.org/10.1016/s0378-8741(00)00196-3
Rana RS, Sood KK (2011) Effect of cutting diameter and hormonal application on the propagation of Ficus roxburghii Wall. through branch cuttings. Ann for Res 55:69–84
Re R, Pellegrini N, Proteggente A, Pannala A, Yang M, Rice-Evans C (1999) Antioxidant activity applying an improved ABTS radical cation decolorization assay. Free Radic Biol Med 26:1231–1237. https://doi.org/10.1016/s0891-5849(98)00315-3
Saitou T, Kamada H, Harada H (1992) Light requirement for shoot regeneration in horseradish hairy roots. Plant Physiol 99:1336–1341. https://doi.org/10.1104/pp.99.4.1336
Samuolienė G, Sirtautas R, Brazaitytė A, Duchovskis P (2012) LED lighting and seasonality effects antioxidant properties of baby leaf lettuce. Food Chem 134:1494–1499. https://doi.org/10.1016/j.foodchem.2012.03.061
Schmidt S, Zietz M, Schreiner M, Rohn S, Kroh LW, Krumbein A (2010) Genotypic and climatic influences on the concentration and composition of flavonoids in kale (Brassica oleracea var. sabellica). Food Chem 119:1293–1299. https://doi.org/10.1016/j.foodchem.2009.09.004
Takagi K, Kimura M, Harada M, Otsuka Y (1982) Pharmacology of medicinal herbs in east asia. Nanzando, Tokyo, pp 187–188
Takahashi A, Ohnishi T (2004) The significance of the study about the biological effects of solar ultraviolet radiation using the exposed facility on the internal space station. Biol Sci Space 18:255–260. https://doi.org/10.2187/bss.18.255
Thimmaiah SK (1999) Standard methods of biochemical analysis. Kalyani Publishers, Noida
Winkel-Shirley and Brenda (2001) Flavonoid biosynthesis. a colorful model for genetics, biochemistry, cell biology, and biotechnology. Plant Physiol 126:485–493. https://doi.org/10.1104/pp.126.2.485
Yi JM, Shin S, Kim NS, Bang OS (2019) Neuroprotective effects of an aqueous extract of Forsythia viridissima and its major constituents on oxaliplatin-induced peripheral neuropathy. Molecules 24:1177. https://doi.org/10.3390/molecules24061177
Zhang Q, Jia C, Xu H, Wang Y, Zhang M, Huo C, Yu S (2012) Chemical constituents of plants from the genus Forsythia. Mini Rev Org Chem 9:303–318. https://doi.org/10.2174/1570193x11209030303
Zhang Y, Feng F, Chen T, Li Z, Shen QW (2016) Antidiabetic and antihyperlipidemic activities of Forsythia suspensa (Thunb.) Vahl (fruit) in streptozotocin-induced diabetes mice. J Ethnopharmacol 192:256–263. https://doi.org/10.1016/j.jep.2016.07.002
Zhao J, Park YG, Jeong BR (2020) Light quality affects growth and physiology of Carpesium triste Maxim. Cultured in Vitro Agriculture 10:258. https://doi.org/10.3390/agriculture10070258
Funding
This work was supported by a grant from the National Institute of Biological Resources (NIBR), funded by the Ministry of Environment (MOE) of the Republic of Korea (NIBR202115101).
Author information
Authors and Affiliations
Contributions
Conceptualization, YGP. and ARY.; methodology, YGP. and ARY.; formal analysis, ARY. and HEO.; data curation, YGP.; writing—original draft preparation, ARY. and YGP.; and writing—review and editing, YGP. All authors have read and agreed to the published version of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Additional information
Communicated by Pramod Kumar Nagar.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Yoon, A., Oh, H.E. & Park, Y.G. Light quality affects the growth and antioxidant activity of Forsythia saxatilis in cutting propagation. Plant Growth Regul 99, 205–214 (2023). https://doi.org/10.1007/s10725-022-00876-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10725-022-00876-z