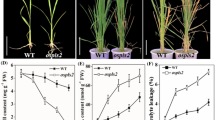
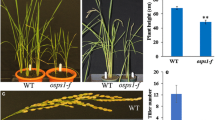
Abstract
Dark-grown seedlings develop skotomorphogenesis. Because of the development of rice direct seeding cultivation systems, there is an increasing need for clarifying the molecular mechanism underlying rice skotomorphogenic development. It has been reported that SRDX motif, LDLDLELRLGFA, was able to convert a transcriptional activator into a strong repressor. In the present study, to explore the functions of PILs in rice skotomorphogenesis, we generated OsPIL11-SRDX and OsPIL16-SRDX transgenic lines by fusing the SRDX transcriptional repressor motif to the C-terminal of two members of the phytochrome interacting factor-like (OsPIL) family in rice (OsPIL11 and OsPIL16). The OsPIL11-SRDX and OsPIL16-SRDX seedlings grown in darkness had constitutively photomorphogenic phenotypes with short coleoptiles and open leaf blades. The results of an RNA sequencing analysis revealed that the dark-grown OsPIL11-SRDX and OsPIL16-SRDX lines had gene expression patterns similar to those of wild-type seedlings grown under red light. Kyoto Encyclopedia of Genes and Genomes pathway enrichment analyses indicated that the expression levels of genes related to photosynthesis, photosynthesis–antenna proteins, and porphyrin and chlorophyll metabolism were up-regulated in the dark-grown OsPIL11-SRDX and OsPIL16-SRDX lines, whereas the expression of genes related to the auxin pathway was down-regulated. In contrast, the expression levels of these photosynthesis-related genes were down-regulated in dark-grown transgenic seedlings overexpressing OsPIL11 or OsPIL16, which had exaggerated skotomorphogenesis. Considered together, our data indicate that OsPIL11 and OsPIL16 primarily function as transcriptional activators, at least in regards to promoting skotomorphogenesis and repressing the expression of photosynthesis-related genes.





Similar content being viewed by others
References
Castillon A, Shen H, Huq E (2007) Phytochrome interacting factors: central players in phytochrome-mediated light signaling networks. Trends Plant Sci 12:514–521. https://doi.org/10.1016/j.tplants.2007.10.001
Chapman EJ, Greenham K, Castillejo C, Sartor R, Bialy A, Sun TP, Estelle M (2012) Hypocotyl transcriptome reveals auxin regulation of growth-promoting genes through GA-dependent and -independent pathways. PLoS ONE 7:e36210. https://doi.org/10.1371/journal.pone.0036210
Chen D, Xu G, Tang W, Jing Y, Ji Q, Fei Z, Lin R (2013) Antagonistic basic helix-loop-helix/bZIP transcription factors form transcriptional modules that integrate light and reactive oxygen species signaling in Arabidopsis. Plant Cell 25:1657–1673. https://doi.org/10.1105/tpc.112.104869
He Y, Li Y, Cui L, Xie L, Zheng C, Zhou G, Zhou J, Xie X (2016) Phytochrome B negatively affects cold tolerance by regulating OsDREB1 gene expression through phytochrome interacting factor-like protein OsPIL16 in rice. Front Plant Sci 7:1963. https://doi.org/10.3389/fpls.2016.01963
Hiratsu K, Ohta M, Matsui K, Ohme-Takagi M (2002) The SUPERMAN protein is an active repressor whose carboxy-terminal repression domain is required for the development of normal flowers. FEBS Lett 514:351–354. https://doi.org/10.1016/s0014-5793(02)02435-3
Hiratsu K, Matsui K, Koyama T, Ohme-Takagi M (2003) Dominant repression of target genes by chimeric repressors that include the EAR motif, a repression domain, in Arabidopsis. Plant J 34:733–739. https://doi.org/10.1046/j.1365-313x.2003.01759.x
Hornitschek P, Kohnen MV, Lorrain S, Rougemont J, Ljung K, López-Vidriero I, Franco-Zorrilla JM, Solano R, Trevisan M, Pradervand S, Xenarios I, Fankhauser C (2012) Phytochrome interacting factors 4 and 5 control seedling growth in changing light conditions by directly controlling auxin signaling. Plant J 71:699–711. https://doi.org/10.1111/j.1365-313X.2012.05033.x
Huq E, Al-Sady B, Hudson M, Kim C, Apel K, Quail PH (2004) PHYTOCHROME-INTERACTING FACTOR 1 is a critical bHLH regulator of chlorophyll biosynthesis. Science 305:1937–1941. https://doi.org/10.1126/science.1099728
Jain M, Nijhawan A, Tyagi AK, Khurana JP (2006) Validation of housekeeping genes as internal control for studying gene expression in rice by quantitative real-time PCR. Biochem Biophys Res Commun 345:646–651. https://doi.org/10.1016/j.bbrc.2006.04.140
Jeong J, Choi G (2013) Phytochrome-interacting factors have both shared and distinct biological roles. Mol Cells 35:371–380. https://doi.org/10.1007/s10059-013-0135-5
Josse EM, Halliday KJ (2008) Skotomorphogenesis: the dark side of light signalling. Curr Biol 18:R1144-1146. https://doi.org/10.1016/j.cub.2008.10.034
Kim K, Shin J, Lee SH, Kweon HS, Maloof JN, Choi G (2011) Phytochromes inhibit hypocotyl negative gravitropism by regulating the development of endodermal amyloplasts through phytochrome-interacting factors. Proc Natl Acad Sci USA 108:1729–1734. https://doi.org/10.1073/pnas.1011066108
Kim K, Jeong J, Kim J, Lee N, Kim ME, Lee S, Chang Kim S, Choi G (2016) PIF1 regulates plastid development by repressing photosynthetic genes in the endodermis. Mol Plant 9:1415–1427. https://doi.org/10.1016/j.molp.2016.08.007
Langmead B, Salzberg SL (2012) Fast gapped-read alignment with Bowtie 2. Nat Methods 9:357–359. https://doi.org/10.1038/nmeth.1923
Lee N, Choi G (2017) Phytochrome-interacting factor from Arabidopsis to liverwort. Curr Opin Plant Biol 35:54–60. https://doi.org/10.1016/j.pbi.2016.11.004
Leivar P, Monte E (2014) PIFs: systems integrators in plant development. Plant Cell 26:56–78. https://doi.org/10.1105/tpc.113.120857
Leivar P, Quail PH (2011) PIFs: pivotal components in a cellular signaling hub. Trends Plant Sci 16:19–28. https://doi.org/10.1016/j.tplants.2010.08.003
Leivar P, Monte E, Oka Y, Liu T, Carle C, Castillon A, Huq E, Quail PH (2008) Multiple phytochrome-interacting bHLH transcription factors repress premature seedling photomorphogenesis in darkness. Curr Biol 18:1815–1823. https://doi.org/10.1016/j.cub.2008.10.058
Leivar P, Tepperman JM, Monte E, Calderon RH, Liu TL, Quail PH (2009) Definition of early transcriptional circuitry involved in light-induced reversal of PIF-imposed repression of photomorphogenesis in young Arabidopsis seedlings. Plant Cell 21:3535–3553. https://doi.org/10.1105/tpc.109.070672
Leivar P, Tepperman JM, Cohn MM, Monte E, Al-Sady B, Erickson E, Quail PH (2012) Dynamic antagonism between phytochromes and PIF family basic helix-loop-helix factors induces selective reciprocal responses to light and shade in a rapidly responsive transcriptional network in Arabidopsis. Plant Cell 24:1398–1419. https://doi.org/10.1105/tpc.112.095711
Li B, Dewey CN (2011) RSEM: accurate transcript quantification from RNA-Seq data with or without a reference genome. BMC Bioinform 12:323. https://doi.org/10.1186/1471-2105-12-323
Li L, Peng W, Liu Q, Zhou J, Liang W, Xie X (2012) Expression patterns of OsPIL11, a phytochrome-interacting factor in rice, and preliminary analysis of its roles in light signal transduction. Rice Sci 19:263–268. https://doi.org/10.1016/S1672-6308(12)60050-X
Li Z, Mo W, Jia L, Xu YC, Tang W, Yang W, Guo YL, Lin R (2019) Rice FLUORESCENT1 is involved in the regulation of chlorophyll. Plant Cell Physiol 60:2307–2318. https://doi.org/10.1093/pcp/pcz129
Liu X, Chen CY, Wang KC, Luo M, Tai R, Yuan L, Zhao M, Yang S, Tian G, Cui Y, Hsieh HL, Wu K (2013) PHYTOCHROME INTERACTING FACTOR3 associates with the histone deacetylase HDA15 in repression of chlorophyll biosynthesis and photosynthesis in etiolated Arabidopsis seedlings. Plant Cell 25:1258–1273. https://doi.org/10.1105/tpc.113.109710
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) Method. Methods 25:402–408. https://doi.org/10.1006/meth.2001.1262
Lorrain S, Trevisan M, Pradervand S, Fankhauser C (2009) Phytochrome interacting factors 4 and 5 redundantly limit seedling de-etiolation in continuous far-red light. Plant J 60:449–461. https://doi.org/10.1111/j.1365-313X.2009.03971.x
Love MI, Huber W, Anders S (2014) Moderated estimation of fold change and dispersion for RNA-seq data with DESeq2. Genome Biol 15:550. https://doi.org/10.1186/s13059-014-0550-8
Matsui K, Umemura Y, Ohme-Takagi M (2008) AtMYBL2, a protein with a single MYB domain, acts as a negative regulator of anthocyanin biosynthesis in Arabidopsis. Plant J 55:954–967. https://doi.org/10.1111/j.1365-313X.2008.03565.x
Mitsuda N, Iwase A, Yamamoto H, Yoshida M, Seki M, Shinozaki K, Ohme-Takagi M (2007) NAC transcription factors, NST1 and NST3, are key regulators of the formation of secondary walls in woody tissues of Arabidopsis. Plant Cell 19:270–280. https://doi.org/10.1105/tpc.106.047043
Murray MG, Thompson WF (1980) Rapid isolation of high molecular weight plant DNA. Nucleic Acids Res 8:4321–4325. https://doi.org/10.1093/nar/8.19.4321
Nakamura Y, Kato T, Yamashino T, Murakami M, Mizuno T (2007) Characterization of a set of phytochrome-interacting factor-like bHLH proteins in Oryza sativa. Biosci Biotechnol Biochem 71:1183–1191. https://doi.org/10.1271/bbb.60643
Nozue K, Harmer SL, Maloof JN (2011) Genomic analysis of circadian clock-, light-, and growth-correlated genes reveals PHYTOCHROME-INTERACTING FACTOR5 as a modulator of auxin signaling in Arabidopsis. Plant Physiol 156:357–372. https://doi.org/10.1104/pp.111.172684
Ohta M, Matsui K, Hiratsu K, Shinshi H, Ohme-Takagi M (2001) Repression domains of class II ERF transcriptional repressors share an essential motif for active repression. Plant Cell 13:1959–1968. https://doi.org/10.1105/tpc.010127
Pham VN, Kathare PK, Huq E (2018) Phytochromes and phytochrome interacting factors. Plant Physiol 176:1025–1038. https://doi.org/10.1104/pp.17.01384
Santner A, Estelle M (2009) Recent advances and emerging trends in plant hormone signalling. Nature 459:1071–1078. https://doi.org/10.1038/nature08122
Shin J, Kim K, Kang H, Zulfugarov IS, Bae G, Lee CH, Lee D, Choi G (2009) Phytochromes promote seedling light responses by inhibiting four negatively-acting phytochrome-interacting factors. Proc Natl Acad Sci USA 106:7660–7665. https://doi.org/10.1073/pnas.0812219106
Stephenson PG, Fankhauser C, Terry MJ (2009) PIF3 is a repressor of chloroplast development. Proc Natl Acad Sci USA 106:7654–7659. https://doi.org/10.1073/pnas.0811684106
Tang W, Wang W, Chen D, Ji Q, Jing Y, Wang H, Lin R (2012) Transposase-derived proteins FHY3/FAR1 interact with PHYTOCHROME-INTERACTING FACTOR1 to regulate chlorophyll biosynthesis by modulating HEMB1 during deetiolation in Arabidopsis. Plant Cell 24:1984–2000. https://doi.org/10.1105/tpc.112.097022
Wu M, Wu J, Gan Y (2020) The new insight of auxin functions: transition from seed dormancy to germination and floral opening in plants. Plant Growth Regul 91:169–174. https://doi.org/10.1007/s10725-020-00608-1
Yamamuro C, Ihara Y, Wu X, Noguchi T, Fujioka S, Takatsuto S, Ashikari M, Kitano H, Matsuoka M (2000) Loss of function of a rice brassinosteroid insensitive1 homolog prevents internode elongation and bending of the lamina joint. Plant Cell 12:1591–1606. https://doi.org/10.1105/tpc.12.9.1591
Yu G, Wang LG, Han Y, He QY (2012) clusterProfiler: an R package for comparing biological themes among gene clusters. OMICS 16:284–287. https://doi.org/10.1089/omi.2011.0118
Zhang Y, Mayba O, Pfeiffer A, Shi H, Tepperman JM, Speed TP, Quail PH (2013) A quartet of PIF bHLH factors provides a transcriptionally centered signaling hub that regulates seedling morphogenesis through differential expression-patterning of shared target genes in Arabidopsis. PLoS Genet 9:e1003244. https://doi.org/10.1371/journal.pgen.1003244
Acknowledgements
We thank Liwen Bianji, Edanz Editing China (www.liwenbianji.cn/ac) for editing the English text of a draft of this manuscript.
Funding
The funded was provided by National Natural Science Foundation of China (Grant Nos: 32070216 and 31700251).
Author information
Authors and Affiliations
Contributions
XX and HZ conceived and designed the research. YL, FZ, CZ, JZ, XM, SN and FC performed the experiments and analyzed the data. YL, FZ, and XX wrote the manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Consent for publication
All authors read this paper and confirm the context.
Additional information
Communicated by Zhong-Hua Chen.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Li, Y., Zhang, F., Zheng, C. et al. Fusion of the SRDX motif to OsPIL11 or OsPIL16 causes rice constitutively photomorphogenic phenotypes in darkness. Plant Growth Regul 96, 157–175 (2022). https://doi.org/10.1007/s10725-021-00767-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10725-021-00767-9