Abstract
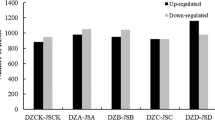
Winter rapeseed seedlings are susceptible to low temperature during overwintering in Northwest China, leading to reduced crops production. Freezing stress is one of the main environmental stresses in Northwest China from late autumn to early spring, an eventful period for overwinter survival rate of winter rapeseed. However, the molecular mechanism of freezing tolerance formation is still very backward in winter rapeseed. In this study, using a pair of freezing-sensitive and freezing-resistant cultivars NQF24 and NTS57, the exhaustive effects of freezing stress on freezing tolerance formation were evaluated by analyzing leaf at the levels of transcriptome, proteome, physiology and ultrastructure. There were 8497 and 7358 differentially expressed genes (DEGs) and 418 and 573 differentially abundant proteins (DAPs) identified in the leaf of NQF24 and NTS57 under freezing stress, respectively. Function enrichment analysis showed that most of the enriched DEGs and DAPs were associated with plant hormones signal transduction, fatty acid metabolism, ribosome, plant-pathogen interaction and secondary metabolites biosynthesis. Freezing tolerance is formed by enhanced signals transduction, increased the biosynthesis of protein and secondary metabolites, enhanced reactive oxygen species (ROS) scavenging, more osmolytes, lower lipid peroxidation, and stronger cell stability. These results can be taken as selection indicators in freezing tolerance breeding program in rapeseed.








Similar content being viewed by others
Abbreviations
- ABA:
-
Abscisic acid
- ACO:
-
1-Aminocyclopropane-1-carboxylate oxidase
- ACS:
-
1-Aminocyclopropane-1-carboxylate synthase
- ABR:
-
Abscisic acid receptor
- ARP:
-
Auxin responsive protein
- ASS:
-
ATP synthase subunit
- ATP:
-
Auxin transporter protein
- BR:
-
Brassinosteroid
- BRI:
-
Brassinosteroid insensitive
- BZR:
-
Brassinazole-resistant
- CAT:
-
Catalase
- CCMT:
-
Caffeic acid O-methyltransferase
- CCO:
-
Cytochrome c oxidase
- CDPK:
-
Ca2+-dependent protein kinase
- CIPK:
-
CBL-interacting serine/threonine-protein kinase
- CP:
-
Chaperone protein
- DAP:
-
Differentially abundant protein
- DDA:
-
Data-dependent acquisition
- DEG:
-
Differentially expressed gene
- DIA:
-
Data-independent acquisition
- EIN:
-
Ethylene insensitive protein
- ERF:
-
Ethylene response factor
- FPKM:
-
Fragments per kilobase per million
- GH3:
-
GH3 auxin-responsive promoter
- GO:
-
Gene Ontology
- HSF:
-
Heat stress transcription factor
- HSP:
-
Heat shock protein
- JA:
-
Jasmonic acid
- KEGG:
-
Kyoto Encyclopedia of Genes and Genomes
- LOX:
-
Lipoxygenases
- MAPK:
-
Mitogen-activated protein kinase
- MDA:
-
Malondialdehyde
- PCA:
-
Principal component analysis
- PIF:
-
Phytochrome interacting factor
- POD:
-
Peroxidase
- PP2C:
-
Protein phosphatase 2C
- RLK:
-
Receptor-like kinase
- ROS:
-
Reactive oxygen species
- SAMT:
-
S-Adenosylmethionine-dependent methyltransferase
- SHCT:
-
Shikimate O-hydroxycinnamoyltransferase
- SnRK:
-
SNF1-related protein kinase
- SOD:
-
Superoxide dismutase
- SPK:
-
Serine/threonine-protein kinase
- TEM:
-
Transmission electron microscope
- TF:
-
Transcription factor
References
Baxter A, Mittler R, Suzuki N (2014) ROS as key players in plant stress signaling. J Exp Bot 65:1229–1240
Binns D, Dimmer E, Huntley R, Barrell D, O’Donovan C, Apweiler R (2009) QuickGO: a web-based tool for Gene Ontology searching. Bioinformatics 25(22):3045–3046
Bruderer R, Bernhardt OM, Gandhi T, Miladinović SM, Cheng LY, Messner S, Ehrenberger T, Zanotelli V, Butscheid Y, Escher C, Vitek O, Rinner O, Reiter L (2015) Extending the limits of quantitative proteome profiling with data-independent acquisition and application to acetaminophen-treated three-dimensional liver microtissues. Mol Cell Proteomics 14(5):1400–1410
Chen MX, Zhu FY, Wang FZ, Ye NH, Gao B, Chen X, Zhao SS, Fan T, Cao YY, Liu TY, Su ZZ, Xie LJ, Hu QJ, Wu HJ, Xiao S, Zhang J, Liu YG (2019) Alternative splicing and translation play important roles in hypoxic germination in rice. J Exp Bot 70(3):817–833
Dahan J, Tcherkez G, Macherel D, Benamar A, Belcram K (2014) Disruption of the CYTOCHROME C OXIDASE DEFICIENT1 gene leads to cytochrome c oxidase depletion and reorchestrated respiratory metabolism in arabidopsis. Plant Physiol 166:1788–1802
Deng S, Sun J, Zhao R, Ding M, Zhang Y, Sun Y, Wang W, Tan Y, Liu D, Ma X, Hou P, Wang M, Lu C, Shen X, Chen S (2015) Populus euphratica APYRASE2 enhances cold tolerance by modulating vesicular trafficking and extracellular ATP in arabidopsis plants. Plant Physiol 169(1):530–548
Ding Y, Li H, Zhang X, Xie Q, Gong Z, Yang S (2015) OST1 kinase modulates freezing tolerance by enhancing ICE1 stability in arabidopsis. Dev Cell 32:278–289
Ding Y, Lv J, Shi Y, Gao J, Hua J, Song C, Gong Z, Yang S (2019a) EGR2 phosphatase regulates OST1 kinase activity and freezing tolerance in arabidopsis. EMBO J 38:e99819
Ding Y, Shi Y, Yang S (2019b) Advances and challenges in uncovering cold tolerance regulatory mechanisms in plants. New Phytol 222(4):1690–1704
Egertson JD, Kuehn A, Merrihew GE, Bateman NW, MacLean BX, Ting YS, Canterbury JD, Marsh DM, Kellmann M, Zabrouskov V, Wu CC, MacCoss MJ (2013) Multiplexed MS/MS for improved data-independent acquisition. Nat Methods 10(8):744–756
Fang P, Yan M, Chi C, Wang M, Zhou Y, Zhou J, Shi K, Xia X, Foyer CH, Yu J (2019) Brassinosteroids act as a positive regulator of photoprotection in response to chilling stress. Plant Physiol 180(4):2061–2076
Frank M, Cortleven A, Novák O, Schmülling T (2020) Root-derived trans-zeatin cytokinin protects Arabidopsis plants against photoperiod stress. Plant Cell Environ 43(11):2637–2649
Gong Z, Xiong L, Shi H, Yang S, Herrera-Estrella LR, Xu G, Chao DY, Li J, Wang PY, Qin F, Li J, Ding Y, Shi Y, Wang Y, Yang Y, Guo Y, Zhu JK (2020) Plant abiotic stress response and nutrient use efficiency. Sci China Life Sci 63(5):635–674
Guo XY, Liu DF, Chong K (2018) Cold signaling in plants: insights into mechanisms and regulation. J Integr Plant Biol 60:745–756
He CG, Gualerzi CO (2021) The ribosome as a switchboard for bacterial stress response. Front Microbiol 11:619038
Hu Y, Jiang Y, Han X, Wang H, Pan J, Yu D (2017) Jasmonate regulates leaf senescence and tolerance to cold stress: crosstalk with other phytohormones. J Exp Bot 68(6):1361–1369
Huang YC, Niu CY, Yang CR, Jinn TL (2016) The heat stress factor HSFA6b connects ABA signaling and ABA-mediated heat responses. Plant Physiol 172(2):1182–1199
Huang H, Liu B, Liu L, Song S (2017) Jasmonate action in plant growth and development. J Exp Bot 68(6):1349–1359
Jamshidi Goharrizi K, Baghizadeh A, Kalantar M, Fatehi F (2019) Combined effects of salinity and drought on physiological and biochemical characteristics of pistachio rootstocks. Sci Hortic 261:108970
Jamshidi Goharrizi K, Amirmahani F, Salehi F (2020a) Assessment of changes in physiological and biochemical traits in four pistachio rootstocks under drought, salinity and drought + salinity stresses. Physiol Plant 168(4):973–989
Jamshidi Goharrizi K, Moosavi SS, Amirmahani F, Salehi F, Nazari M (2020b) Assessment of changes in growth traits, oxidative stress parameters, and enzymatic and non-enzymatic antioxidant defense mechanisms in Lepidium draba plant under osmotic stress induced by polyethylene glycol. Protoplasma 257(2):459–473
Jamshidi Goharrizi K, Geoffrey M, Ghotbzadeh Kermani S, Heidarinezhad A, Salehi F (2021) Short-term cold stress affects physiological and biochemical traits of pistachio rootstocks. S Afr J Bot 141:90–98
Jia Y, Ding Y, Shi Y, Zhang X, Gong Z, Yang S (2016) The cbfs triple mutants reveal the essential functions of CBFs in cold acclimation and allow the definition of CBF regulons in arabidopsis. New Phytol 212:345–353
Jiang B, Shi Y, Zhang X, Xin X, Qi L, Guo H, Li J, Yang S (2017) PIF3 is a negative regulator of the CBF pathway and freezing tolerance in Arabidopsis. Proc Natl Acad Sci 114(32):6695–6702
Kanehisa M, Sato Y, Kawashima M, Furumichi M, Tanabe M (2016) KEGG as a reference resource for gene and protein annotation. Nucleic Acids Res 44(1):457–462
Kazan K (2015) Diverse roles of jasmonates and ethylene in abiotic stress tolerance. Trends Plant Sci 20(4):219–229
Kim D, Pertea G, Trapnell C, Pimentel H, Kelley R, Salzberg SL (2013) TopHat2: accurate alignment of transcriptomes in the presence of insertions, deletions and gene fusions. Genome Biol 14(4):R36
Kim SH, Kim HS, Bahk S, An J, Yoo Y, Kim JY, Chung WS (2017) Phosphorylation of the transcriptional repressor MYB15 by mitogen-activated protein kinase 6 is required for freezing tolerance in arabidopsis. Nucleic Acids Res 45(11):6613–6627
Klusch N, Murphy BJ, Mills DJ, Yildiz O, Kuhlbrandt W (2017) Structural basis of proton translocation and force generation in mitochondrial ATP synthase. Elife 6:e33274
Li H, Ye K, Shi Y, Cheng J, Zhang X, Yang S (2017) BZR1 positively regulates freezing tolerance via CBF-dependent and CBF-independent pathways in arabidopsis. Mol Plant 10:545–559
Li B, Liu Y, Cui XY, Fu JD, Zhou YB, Zheng WJ, Lan JH, Jin LG, Chen M, Ma YZ, Xu ZS, Min DH (2019) Genome-wide characterization and expression analysis of soybean TGA transcription factors identified a novel TGA gene involved in drought and salt tolerance. Front Plant Sci 10:549
Lin Q, Xie Y, Guan W, Duan Y, Wang Z, Sun C (2019) Combined transcriptomic and proteomic analysis of cold stress induced sugar accumulation and heat shock proteins expression during postharvest potato tuber storage. Food Chem 297:124991
Liu Z, Jia Y, Ding Y, Shi Y, Li Z, Guo Y, Gong Z, Yang S (2017) Plasma membrane CRPK1-mediated phosphorylation of 14-3-3 proteins induces their nuclear import to fine-tune CBF signaling during cold response. Mol Cell 66:117–128
Ma Y, Dai X, Xu Y, Luo W, Zheng X, Zeng D, Pan Y, Lin X, Liu H, Zhang D (2015) COLD1 confers chilling tolerance in rice. Cell 160:1209–1221
Ma N, Hu C, Wan L, Hu Q, Xiong J, Zhang C (2017) Strigolactones improve plant growth, photosynthesis, and alleviate oxidative stress under salinity in rapeseed (Brassica napus L.) by regulating gene expression. Front Plant Sci 8:1671
Ma X, Chen C, Yang M, Dong X, Lv W, Meng Q (2018) Cold-regulated protein (SlCOR413IM1) confers chilling stress tolerance in tomato plants. Plant Physiol Biochem 124:29–39
Ma J, Chen T, Wu S, Yang C, Bai M, Shu K, Li K, Zhang G, Jin Z, He F, Hermjakob H, Zhu Y (2019a) iProX: an integrated proteome resource. Nucleic Acids Res 47(1):1211–1217
Ma L, Coulter JA, Liu L, Zhao Y, Chang Y, Pu Y, Zeng X, Xu Y, Wu J, Fang Y, Bai J, Sun W (2019b) Transcriptome analysis reveals key cold-stress-responsive genes in winter rapeseed (Brassica rapa L.). Int J Mol Sci 20(5):1071
Moin M, Bakshi A, Saha A, Dutta M, Madhav SM, Kirti PB (2016) Rice ribosomal protein large subunit genes and their spatio-temporal and stress regulation. Front Plant Sci 7:1284
Nowicka B, Ciura J, Szymańska R, Kruk J (2018) Improving photosynthesis, plant productivity and abiotic stress tolerance current trends and future perspectives. Plant Physiol 231:415–433
Palm DM, Agostini A, Averesch V, Girr P, Werwie M, Takahashi S, Satoh H, Jaenicke E, Paulsen H (2018) Chlorophyll a/b binding-specificity in water-soluble chlorophyll protein. Nat Plants 4(11):920–929
Park S, Lee CM, Doherty CJ, Gilmour SJ, Kim Y, Thomashow MF (2015) Regulation of the arabidopsis CBF regulon by a complex low temperature regulatory network. Plant J 82:193–207
Pu Y, Liu L, Wu J, Zhao Y, Bai J, Ma L, Yue J, Jin J, Niu Z, Fang Y, Sun W (2019) Transcriptome profile analysis of winter rapeseed (Brassica napus L.) in response to freezing stress, reveal potentially connected events to freezing stress. Int J Mol Sci 20(11):2771
Ramakrishna A, Ravishankar GA (2011) Influence of abiotic stress signals on secondary metabolites in plants. Plant Signal Behav 6(11):1720–1731
Ranjan R, Kumar N, Gautam A, Kumar Dubey A, Pandey SN, Mallick S (2020) Chlorella sp. modulates the glutathione mediated detoxification and S-adenosylmethionine dependent methyltransferase to counter arsenic toxicity in Oryza sativa L. Ecotoxicol Environ Saf 208:111418
Ray PD, Huang BW, Tsuji Y (2012) Reactive oxygen species (ROS) homeostasis and redox regulation in cellular signaling. Cell Signal 24:981–990
Scarpeci TE, Zanor MI, Valle EM (2008) Investigating the role of plant heat shock proteins during oxidative stress. Plant Signal Behav 3:856–857
She J, Han Z, Kim TW, Wang J, Cheng W, Chang J, Shi S, Wang J, Yang M, Wang ZY, Chai J (2011) Structural insight into brassinosteroid perception by BRI1. Nature 474:472–476
Shi Y, Tian S, Hou L, Huang X, Zhang X, Guo H, Yang S (2012) Ethylene signaling negatively regulates freezing tolerance by repressing expression of CBF and type-A ARR genes in arabidopsis. Plant Cell 24:2578–2595
Shi Y, Ding Y, Yang S (2018) Molecular regulation of CBF signaling in cold acclimation. Trends Plant Sci 23:623–637
Song L, Liu Z, Tong J, Xiao L, Ma H, Zhang H (2015) Comparative proteomics analysis reveals the mechanism of fertility alternation of thermosensitive genic male sterile rice lines under low temperature inducement. Proteomics 15(11):1884–1905
Toyooka K, Sato M, Kutsuna N, Nagata N (2014) Development of high resolution TEM image acquisition system by using high-pressure freezing method. Plant Morphol 26:3–8
Trapnell C, Williams BA, Pertea G, Mortazavi A, Kwan G, van Baren MJ, Salzberg SL, Wold BJ, Pachter L (2010) Transcript assembly and quantification by RNA-Seq reveals unannotated transcripts and isoform switching during cell differentiation. Nat Biotechnol 28:511–515
Wang Y, Dai Y, Tao X, Wang JZ, Cheng HY, Yang H, Ma XR (2016) Heat shock factor genes of tall fescue and perennial ryegrass in response to temperature stress by RNA-Seq analysis. Front Plant Sci 6:1226
Wei J, Liu X, Li L, Zhao H, Liu S, Yu X, Shen Y, Zhou Y, Zhu Y, Shu Y, Ma H (2020) Quantitative proteomic, physiological and biochemical analysis of cotyledon, embryo, leaf and pod reveals the effects of high temperature and humidity stress on seed vigor formation in soybean. BMC Plant Biol 20:127
Wei J, Zheng G, Yu X, Liu S, Dong X, Cao X, Fang X, Li H, Jin J, Mi W, Liu Z (2021) Comparative transcriptomics and proteomics analyses of leaves reveals a freezing stress-responsive molecular network in winter rapeseed (Brassica rapa L.). Front Plant Sci 12:664311
Xia XJ, Fang PP, Guo X, Qian XJ, Zhou J, Shi K, Zhou YH, Yu JQ (2018) Brassinosteroid-mediated apoplastic H2O2-glutaredoxin 12/14 cascade regulates antioxidant capacity in response to chilling in tomato. Plant Cell Environ 41:1052–1064
Xie Y, Chen P, Yan Y, Bao C, Li X, Wang L, Shen X, Li H, Liu X, Niu C, Zhu C, Fang N, Shao Y, Zhao T, Yu J, Zhu J, Xu L, van Nocker S, Ma F, Guan Q (2018) An atypical R2R3 MYB transcription factor increases cold hardiness by CBF-dependent and CBF-independent pathways in apple. New Phytol 218(1):201–218
Xu H, Martinoia E, Szabo I (2015) Organellar channels and transporters. Cell Calcium 58(1):1–10
Xu Y, Zeng X, Wu J, Zhang F, Li C, Jiang J, Wang Y, Sun W (2018) iTRAQ-based quantitative proteome revealed metabolic changes in winter turnip rape (Brassica rapa L.) under cold stress. Int J Mol Sci 19(11):3346
Yan L, Zhai Q, Wei J, Li S, Wang B, Huang T, Du M, Sun J, Kang L, Li CB, Li C (2013) Role of tomato lipoxygenase D in wound-induced jasmonate biosynthesis and plant immunity to insect herbivores. PLoS Genet 9(12):e1003964
Yan MY, Xie DL, Cao JJ, Xia XJ, Shi K, Zhou YH, Zhou J, Foyer CH, Yu JQ (2020) Brassinosteroid-mediated reactive oxygen species are essential for tapetum degradation and pollen fertility in tomato. Plant J 102(5):931–947
Zeng X, Xu Y, Jiang J, Zhang F, Ma L, Wu D, Wang Y, Sun W (2018) iTRAQ-based comparative proteomic analysis of the roots of two winter Turnip Rapes (Brassica rapa L.) with different freezing-tolerance. Int J Mol Sci 19(12):4077
Zhang CY, Zhang ZY, Li JH, Li F, Liu HH, Yang WS, Chong K, Xu YY (2017) OsMAPK3 phosphorylates OsbHLH002/OsICE1 and inhibits its ubiquitination to activate OsTPP1 and enhances rice chilling tolerance. Dev Cell 43:731–743
Zhao Y, Dong W, Zhang N, Ai X, Wang M, Huang Z, Xiao L, Xia G (2014) A wheat allene oxide cyclase gene enhances salinity tolerance via jasmonate signaling. Plant Physiol 164(2):1068–1076
Zhao C, Zhang Z, Xie S, Si T, Li Y, Zhu JK (2016) Mutational evidence for the critical role of CBF transcription factors in cold acclimation in arabidopsis. Plant Physiol 171:2744–2759
Zhao C, Wang P, Si T, Hsu CC, Wang L, Zayed O, Yu Z, Zhu Y, Dong J, Tao WA, Zhu JK (2017) MAP kinase cascades regulate the cold response by modulating ICE1 protein stability. Dev Cell 43:618–629
Zhu J (2016) Review abiotic stress signaling and responses in plants. Cell 167:313–324
Acknowledgements
The authors gratefully acknowledge the partial financial support from the Young Doctoral Fund of Gansu (2021QB-035), Industrial Support Plan Project of Gansu (2021CYZC-46), Scientific Research Start-up Funds for Openly-recruited Doctors of Gansu Agricultural University (GAU-KYQD-2019-25), Ministry of Science and Technology of China (2018YFD0100500), National Natural Science Foundation of China (31660404) and the Special Funds for the Central Government to Guide Local Technological Development of China (ZCYD-2020-2-3).
Author information
Authors and Affiliations
Contributions
This work presented here was finished by all authors. JW performed the multi-omics joint analysis and wrote the manuscript; GZ executed the physiological and biochemical assays; XD and SL participated in the proteomic data analysis; HL and YW cultivated all plant samples; ZL designed the experiments and revised the paper. All authors have read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing financial interests.
Additional information
Communicated by Vaclav Motyka.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
10725_2021_763_MOESM1_ESM.jpg
Supplementary file1 (JPG 723 kb) Fig S1. The repeatability analysis among three biological replicates of NFC, NFT, NSC and NST in transcriptome (A) and proteome (B), respectively
10725_2021_763_MOESM2_ESM.jpg
Supplementary file2 (JPG 1918 kb) Fig S2. Pearson’s correlation coefficient among three biological replicates of NFC, NFT, NST and NST in transcriptome
10725_2021_763_MOESM3_ESM.jpg
Supplementary file3 (JPG 1509 kb) Fig S3. Pearson’s correlation coefficient between three biological replicates of NFC, NFT, NST and NST in proteome
10725_2021_763_MOESM4_ESM.jpg
Supplementary file4 (JPG 576 kb) Fig S4. GO classification analysis of the specifically detected DEGs (A) and DAPs (B) in NFC_NFT
10725_2021_763_MOESM5_ESM.jpg
Supplementary file5 (JPG 582 kb) Fig S5. GO classification analysis of the specifically detected DEGs (A) and DAPs (B) detected in NSC_NST
10725_2021_763_MOESM6_ESM.xlsx
Supplementary file6 (XLSX 25216 kb) Table S1. The proteins database of Brassica napus downloaded from the NCBI, and the list of all genes and proteins identified in transcriptome and proteome, respectively
10725_2021_763_MOESM7_ESM.xlsx
Supplementary file7 (XLSX 7637 kb) Table S2. DEGs identified in leaves between the stressed and control rapeseed cultivar NF and NS, respectively
10725_2021_763_MOESM8_ESM.xlsx
Supplementary file8 (XLSX 1753 kb) Table S3. DAPs identified in leaves between the stressed and control rapeseed cultivar NF and NS, respectively
10725_2021_763_MOESM11_ESM.xlsx
Supplementary file11 (XLSX 1444 kb) Table S6. Differentially expressed TFs identified in leaves between the control and the stressed rapeseed cultivar NF and NS, respectively, by transcriptome
Rights and permissions
About this article
Cite this article
Wei, J., Zheng, G., Dong, X. et al. Integration of transcriptome and proteome analysis reveals the mechanism of freezing tolerance in winter rapeseed. Plant Growth Regul 96, 103–118 (2022). https://doi.org/10.1007/s10725-021-00763-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10725-021-00763-z