Abstract
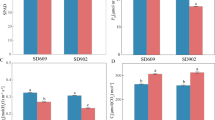
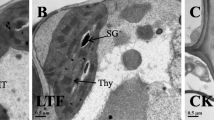
With global climate change in progress, a particular problem is that sudden drops in temperature affect the production of rice. In this study, we explored how to overcome the effects of chilling injury on rice seedlings. Wild-type Dular (WT) plants and Lsi1 (low silicon rice 1) overexpressing Dular plants (Lsi1-OX) were used as research materials. We found that Lsi1 enhanced the antioxidant system and non-structural carbohydrates (NSC) of rice at physiological levels. On this basis, Isobaric Tag for Relative Absolute Quantitation (iTRAQ) technology was used to examine the expression of the rice root proteins in response to low temperature stress. In addition, a total of 433 proteins were differentially expressed in this study, of which 284 were upregulated and 149 were downregulated. These proteins are divided into 24 functions. Moreover, our analysis found that the signaling pathway of the Lsi1-OX plant roots is significantly enhanced under low temperature stress, which in turn, led to the development of resistance reaction and transport pathways, indicating that Lsi1 increases the ability of rice to perceive and transmit low-temperature signals and stimulate the corresponding biochemical processes. In this study, we provided a theoretical basis to understand the mechanism related to the cold resistance of plants and therefor, accelerate the molecular breeding process of low temperature resistant crops.





Similar content being viewed by others
Abbreviations
- WT:
-
Wild-type Dular
- Lsi1-OX:
-
Lsi1 over expressing Dular
- SSC:
-
Soluble sugar content
- SC:
-
Starch content
- NSC:
-
Non-structural carbohydrates
- DEPs:
-
Differentially expressed proteins
- CDPK:
-
Calcium-dependent protein kinase
- NCX:
-
Sodium/calcium exchanger protein
- CBHS:
-
Calmodulin binding heat-shock protein
- RFC:
-
Replication factor C
- ROS:
-
Reactive oxygen species
References
Abbasi NA, Kushad MM (2006) The activities of SOD, POD, and CAT in 'Red Spur Delicious' apple fruit are affected by DPA but not calcium in postharvest drench solutions. J Am Pomol Soc 60(2):84–89
Abbasi F, Onodera H, Toki S, Tanaka H, Komatsu S (2004) OsCDPKI3, a calcium-dependent protein kinase gene from rice, is induced by cold and gibberellin in rice leaf sheath. Plant Mol Biol 55(4):541–552. https://doi.org/10.1007/s11103-004-1178-y
Almadanim MC, Alexandre BM, Rosa MTG, Sapeta H, Leitao AE, Ramalho JC, Lam TT, Negrao S, Abreu IA, Oliveira MM (2017) Rice calcium-dependent protein kinase OsCPK17 targets plasma membrane intrinsic protein and sucrose-phosphate synthase and is required for a proper cold stress response. Plant Cell Environ 40(7):1197–1213. https://doi.org/10.1111/pce.12916
Almadanim MC, Goncalves NM, Rosa MTG, Alexandre BM, Cordeiro AM, Rodrigues M, Saibo NJM, Soares CM, Romao CV, Oliveira MM, Abreu IA (2018) The rice cold-responsive calcium-dependent protein kinase OsCPK17 is regulated by alternative splicing and post-translational modifications. BBA 2:231–246. https://doi.org/10.1016/j.bbamcr.2017.10.010
Azeem S, Li Z, Zheng H, Lin W, Arafat Y, Zhang Z, Lin X, Lin W (2015) Quantitative proteomics study on Lsi1 in regulation of rice (Oryza sativa L.) cold resistance. Plant Growth Regul 78(3):307–323. https://doi.org/10.1007/s10725-015-0094-2
Chen THH, Murata N (2002) Enhancement of tolerance of abiotic stress by metabolic engineering of betaines and other compatible solutes. Curr Opin Plant Biol 5(3):250–257. https://doi.org/10.1016/S1369-5266(02)00255-8
Chen YY, Qian J, You L, Zhang XF, Jiao JX, Liu Y, Zhao J (2018) Subunit interaction differences between the replication factor C complexes in Arabidopsis and rice. Front Plant Sci. https://doi.org/10.3389/Fpls.2018.00779
Chow PS, Landhäusser SM (2004) A method for routine measurements of total sugar and starch content in woody plant tissues. Tree Physiol 24(10):1129–1136. https://doi.org/10.1093/treephys/24.10.1129
Cox J, Mann M (2008) MaxQuant enables high peptide identification rates, individualized p.p.b.-range mass accuracies and proteome-wide protein quantification. Nat Biotechnol 26(12):1367–1372. https://doi.org/10.1038/nbt.1511
Cruz RPD, Sperotto RA, Cargnelutti D, Adamski JM, de FreitasTerra T, Fett JP (2013) Avoiding damage and achieving cold tolerance in rice plants. Food Energy Secur 2(2):96–119. https://doi.org/10.1002/fes3.25
Cui X, Wang Y, Wu J, Han X, Gu X, Lu T, Zhang Z (2019) The RNA editing factor DUA1 is crucial to chloroplast development at low temperature in rice. New Phytol 221(2):834–849. https://doi.org/10.1111/nph.15448
Dai SH, Wei XP, Alfonso AA, Pei LP, Duque UG, Zhang ZH, Babb GM, Beachy RN (2008) Transgenic rice plants that overexpress transcription factors RF2a and RF2b are tolerant to rice Tungro virus replication and disease. Proc Natl Acad Sci USA 105(52):21012–21016. https://doi.org/10.1073/pnas.0810303105
Dubey AK, Kumar N, Kumar A, Ansari MA, Ranjan R, Gautam A, Meenakshi SN, Pandey V, Behera SK, Mallick S, Pande V, Sanyal I (2019) Over-expression of CarMT gene modulates the physiological performance and antioxidant defense system to provide tolerance against drought stress in Arabidopsis thaliana L. Ecotoxicol Environ Saf 171:54–65. https://doi.org/10.1016/j.ecoenv.2018.12.050
Fang CX, Zhang PL, Jian X, Chen WS, Lin HM, Li YZ, Lin WX (2017) Overexpression of Lsi1 in cold-sensitive rice mediates transcriptional regulatory networks and enhances resistance to chilling stress. Plant Sci 262:115–126
Giladi M, Shor R, Lisnyansky M, Khananshvili D (2016) Structure-functional basis of ion transport in sodium-calcium exchanger (NCX) proteins. Int J Mol Sci. https://doi.org/10.3390/Ijms17111949
Gucciardo S, Wisniewski JP, Brewin NJ, Bornemann S (2007) A germin-like protein with superoxide dismutase activity in pea nodules with high protein sequence identity to a putative rhicadhesin receptor. J Exp Bot 58(5):1161–1171. https://doi.org/10.1093/jxb/erl282
Guilbault KR, Brown CS, Friedman JM, Shafroth PB (2012) The influence of chilling requirement on the southern distribution limit of exotic Russian olive (Elaeagnus angustifolia) in western North America. Biol Invasion 14(8):1711–1724. https://doi.org/10.1007/s10530-012-0182-4
Guo XY, Liu DF, Chong K (2018) Cold signaling in plants: insights into mechanisms and regulation. J Integr Plant Biol 60(9):745–756. https://doi.org/10.1111/jipb.12706
Hu WH, Hu GC, Han B (2009) Genome-wide survey and expression profiling of heat shock proteins and heat shock factors revealed overlapped and stress specific response under abiotic stresses in rice. Plant Sci 176(4):583–590. https://doi.org/10.1016/j.plantsci.2009.01.016
Jangam AP, Pathak RR, Raghuram N (2016) Microarray analysis of rice d1 (RGA1) mutant reveals the potential role of G-protein alpha subunit in regulating multiple abiotic stresses such as drought, salinity, heat, and cold. Front Plant Sci. https://doi.org/10.3389/Fpls.2016.00011
Komatsu S, Yang GX, Khan M, Onodera H, Toki S, Yamaguchi M (2007) Over-expression of calcium-dependent protein kinase 13 and calreticulin interacting protein 1 confers cold tolerance on rice plants. Mol Genet Genomics 277(6):713–723. https://doi.org/10.1007/s00438-007-0220-6
Kuk JSS, Burgos NR, Hwang TE, Guh OJ (2003) Antioxidative enzymes offer protection from chilling damage in rice plants. Crop Sci 43(6):2109–2117. https://doi.org/10.2135/cropsci2003.2109
Liu HT, Gao F, Li GL, Han JL, Liu DL, Sun DY, Zhou RG (2008) The calmodulin-binding protein kinase 3 is part of heat-shock signal transduction in Arabidopsis thaliana. Plant J 55(5):760–773. https://doi.org/10.1111/j.1365-313X.2008.03544.x
Ma JF, Mitani N, Nagao S, Konishi S, Tamai K, Iwashita T, Yano M (2004) Characterization of the silicon uptake system and molecular mapping of the silicon transporter gene in rice. Plant Physiol 136(2):3284–3289. https://doi.org/10.1104/pp.104.047365
Ma JF, Tamai K, Yamaji N, Mitani N, Konishi S, Katsuhara M, Ishiguro M, Murata Y, Yano M (2006) A silicon transporter in rice. Nature 440(7084):688–691. https://doi.org/10.1038/nature04590
Manosalva PM, Davidson RM, Liu B, Zhu XY, Hulbert SH, Leung H, Leach JE (2009) A germin-like protein gene family functions as a complex quantitative trait locus conferring broad-spectrum disease resistance in rice. Plant Physiol 149(1):286–296. https://doi.org/10.1104/pp.108.128348
Panda D, Sarkar RK (2014) Mechanism associated with nonstructural carbohydrate accumulation in submergence tolerant rice (Oryza sativa L.) cultivars. J Plant Interact 9(1):62–68. https://doi.org/10.1080/17429145.2012.763000
Ron M (2002) Oxidative stress, antioxidants and stress tolerance. Trends Plant Sci 7(9):405–410. https://doi.org/10.1016/S1360-1385(02)02312-9
Rudd JJ, Franklin-Tong VE (2001) Unravelling response-specificity in Ca2+ signalling pathways in plant cells. New Phytol 151(1):7–33. https://doi.org/10.1046/j.1469-8137.2001.00173.x
Sakamoto T, Deguchi M, Brustolini OJ, Santos AA, Silva FF, Fontes EP (2012) The tomato RLK superfamily: phylogeny and functional predictions about the role of the LRRII-RLK subfamily in antiviral defense. BMC Plant Biol 12(1):229. https://doi.org/10.1186/1471-2229-12-229
Sanders D, Brownlee C, Harper JF (1999) Communicating with calcium. Plant Cell 11(4):691–706. https://doi.org/10.2307/3870893
Sato Y, Masuta Y, Saito K, Murayama S, Ozawa K (2011) Enhanced chilling tolerance at the booting stage in rice by transgenic overexpression of the ascorbate peroxidase gene, OsAPXa. Plant Cell Rep 30(3):399–406. https://doi.org/10.1007/s00299-010-0985-7
Shahbaz M, Abid A, Masood A, Waraich EA (2017) Foliar-applied trehalose modulates growth, mineral nutrition, photosynthetic ability, and oxidative defense system of rice (Oryza sativa L.) under saline stress. J Plant Nutr 40(4):584–599. https://doi.org/10.1080/01904167.2016.1263319
Sinha DK, Atray I, Bentur JS, Nair S (2015) Feeding on resistant rice leads to enhanced expression of defender against apoptotic cell death (OoDAD1) in the Asian rice gall. BMC Plant Biol 15(1):235. https://doi.org/10.1186/S12870-015-0618-Y
Sun J, Zheng TH, Yu J, Wu TT, Wang XH, Chen GM, Tian YL, Zhang H, Wang YH, Terzaghi W, Wang CM, Wan JM (2017) TSV, a putative plastidic oxidoreductase, protects rice chloroplasts from cold stress during development by interacting with plastidic thioredoxin Z. New Phytol 215(1):240–255. https://doi.org/10.1111/nph.14482
Sun RJ, Xu Y, Hou CX, Zhan YH, Liu MQ, Weng XY (2018) Expression and characteristics of rice xylanase inhibitor OsXIP, a member of a new class of antifungal proteins. Biol Plant 62(3):569–578. https://doi.org/10.1007/s10535-018-0787-2
Thimm O, Blasing O, Gibon Y, Nagel A, Meyer S, Kruger P, Selbig J, Muller LA, Rhee SY, Stitt M (2010) MAPMAN: a user-driven tool to display genomics data sets onto diagrams of metabolic pathways and other biological processes. Plant J 37(6):914–939. https://doi.org/10.1111/j.1365-313X.2004.02016.x
Wang XB, Xia M, Chen QS, Wu ZC, Wu P (2002) Identification of a new small GTP-binding protein gene OsRab5a, genomic organization, and expression pattern analysis during nitrate supply and early nutrient starvation in rice (Oryza sativa L.) root. Plant Sci 163(2):273–280. https://doi.org/10.1016/S0168-9452(02)00096-1
Wang Z, Xing SP, Birkenbihl RP, Zachgo S (2009) Conserved functions of Arabidopsis and rice CC-type glutaredoxins in flower development and pathogen response. Mol Plant 2(2):323–335. https://doi.org/10.1093/mp/ssn078
Wu J, Wang Y, Kim ST, Kim SG, Kang KY (2013) Characterization of a newly identified rice chitinase-like protein (OsCLP) homologous to xylanase inhibitor. BMC Biotechnol 13(1):4. https://doi.org/10.1186/1472-6750-13-4
Xiong DL, Yu TT, Ling XX, Fahad S, Peng SB, Li Y, Huang JL (2015) Sufficient leaf transpiration and nonstructural carbohydrates are beneficial for high-temperature tolerance in three rice (Oryza sativa) cultivars and two nitrogen treatments. Funct Plant Biol 42(4):347–356. https://doi.org/10.1071/FP14166
Yang GX, Shen SH, Yang SH, Komatsu S (2003) OsCDPK13, a calcium-dependent protein kinase gene from rice, is induced in response to cold and gibberellin. Plant Physiol Biochem 41(4):369–374. https://doi.org/10.1016/S0981-9428(03)00032-9
Yoshida M, Kawakami A, Sato Y (2007) Analyses of cold tolerance and sugar content of rice seedlings expressing wheat fructosyltransferase. Plant Cell Physiol 48:S237–S237
Zhang ZG, Zhang Q, Wu JX, Zheng X, Zheng S, Sun XH, Qiu QS, Lu TG (2013) Gene knockout study reveals that cytosolic ascorbate peroxidase 2(OsAPX2) plays a critical role in growth and reproduction in rice under drought, salt and cold stresses. PLoS ONE 8(2):e57472. https://doi.org/10.1371/journal.pone.0057472
Zhang JY, Luo W, Zhao Y, Xu YY, Song SH, Chong K (2016) Comparative metabolomic analysis reveals a reactive oxygen species-dominated dynamic model underlying chilling environment adaptation and tolerance in rice. New Phytol 211(4):1295–1310. https://doi.org/10.1111/nph.14011
Zhang ZX, Zhang YP, Zhao H, Huang FL, Zhang ZF, Lin WX (2017) The important functionality of 14-3-3 isoforms in rice roots revealed by affinity chromatography. J Proteomics 158:20–30. https://doi.org/10.1016/j.jprot.2017.02.008
Acknowledgements
This work was sponsored by the National Key Research and Development program of China (2016YFD0300508), Fujian-Taiwan Joint Innovative Centre for Germplasm Resources and Cultivation of Crop (Fujian 2011 program, No. 2015-75), the Natural Foundation of Fujian Higher Education Institutions for Young Scientists (Key Project) (JZ160435) and Science and technology development fund of Fujian Agriculture and Forestry University (KF2015043).
Author information
Authors and Affiliations
Corresponding authors
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Li, Z., Feng, S., Zhan, W. et al. Lsi1 plays an active role in enhancing the chilling tolerance of rice roots. Plant Growth Regul 90, 529–543 (2020). https://doi.org/10.1007/s10725-020-00577-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10725-020-00577-5