Abstract
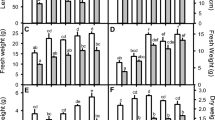
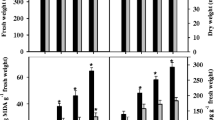
Sorghum bicolor plants were grown in five combinations of NO3 −:NH4 + nutrition (100:0, 75:25, 50:50, 25:75 and 0:100) to test the hypothesis that nitrogen nutrition with ammonium (NH4 +) increases tolerance to salinity. Analyses were performed after 10 days of 75 mM NaCl stress. The K+ contents in the shoots and roots were reduced under salinity stress; however, salt-stressed plants from NO3 −:NH4 + treatments of 100:0 and 0:100 showed higher K+ concentrations in the shoots. Under salinity, Na+ accumulation was severely limited in the presence of NH4 + (0:100), which positively influenced K+/Na+ homeostasis. In parallel, NH4 +-fed plants displayed a substantial accumulation of N-rich amino acids (primarily glutamine and asparagine) in both tissues, which might be fundamental for alleviating NH4 + toxicity. These responses were reflected in favorable gas exchange and growth. Whereas the photosynthetic rate (A) of plants treated with 100:0, 75:25, 50:50 and 25:75 ratios were unaltered or reduced under salinity stress, plants treated with a ratio 0:100 showed increases in A; this phenomenon was strongly correlated with the higher instantaneous carboxylation efficiency of Rubisco (A/Ci) and total N content in the shoots. As a result, although the leaf area and shoot and root dry mass from all nitrogen treatments were severely reduced through stress, the NH4 + supply was effective to alleviate the harmful effects of salinity. The results clearly demonstrate that ammonium nutrition is more advantageous for the growth of S. bicolor under salinity than nitrate nutrition. Our findings suggest that improved plant acclimation induced through NH4 + resulted from increased photosynthesis performance, reduced Na+ toxicity effects and major N-compound content.





Similar content being viewed by others
References
Abdolzadeh A, Shima K, Lambers H, Chiba K (2008) Change in uptake, transport and accumulation of ions in Nerium oleander (rosebay) as affected by different nitrogen sources and salinity. Ann Bot Lond 102:735–746. doi:10.1093/aob/mcn156
Armengaud P, Sulpice R, Miller AJ, Stitt M, Amtmann A, Gibon Y (2009) Multilevel analysis of primary metabolism provides new insights into the role of potassium nutrition for glycolysis and nitrogen assimilation in Arabidobpsis roots. Plant Physiol 150:772–785. doi:10.1104/pp.108.133629
Baethgen WE, Alley MM (1989) A manual colorimetric procedure for measuring ammonium nitrogen in soil and plant Kjeldahl digest. Commun Soil Sci Plant Ann 20:961–969. doi:10.1080/00103628909368129
Britto DT, Kronzucker HJ (2002) NH4 + toxicity in higher plants: a critical review. J Plant Physiol 159:567–584. doi:10.1078/0176-1617-0774
Calatayud Á, Gorbe E, Roca D, Martínez PF (2008) Effect of two nutrient solution temperatures on nitrate uptake, nitrate reductase activity, NH4 + concentration and chlorophyll a fluorescence in rose plants. Environ Exp Bot 64:65–74. doi:10.1016/j.envexpbot.2008.02.003
Cataldo JM, Haroom M, Schrader LE, Youngs VL (1975) Rapid colorimetric determination of nitrate in plant tissue. Commun Soil Sci Plant 6:71–80. doi:10.1080/00103627509366547
Chang B, Yang L, Cong W, Zu Y, Tang Z (2014) The improved resistance to high salinity induced by trehalose is associated with ionic regulation and osmotic adjustment in Catharanthus roseus. Plant Physiol Biochem 77:140–148. doi:10.1016/j.plaphy.2014.02.001
Claussen W, Lenz F (1995) Effect of ammonium and nitrate on net photosynthesis, flower formation, growth and yield of eggplants (Solanum melongena L.). Plant Soil 171:267–274. doi:10.1007/BF00010281
Cuin TA, Shabala S (2007) Compatible solutes reduce ROS-induced potassium efflux in Arabidopsis roots. Plant Cell Environ 30:875–885. doi:10.1111/j.1365-3040.2007.01674.x
Cuin TA, Betts SA, Chalmandrier R, Shabala S (2008) A root’s ability to retain K+ correlates with salt tolerance in wheat. J Exp Bot 59:2697–2706. doi:10.1093/jxb/ern128
Dluzniewska P, Gressler A, Dietrich H, Schnitzler JP, Teuber M, Rennenberg H (2007) Nitrogen uptake and metabolism in Populus x canescens as affected by salinity. New Phytol 173:279–293. doi:10.1111/j.1469-8137.2006.01908.x
Ehlting B, Dluzniewska P, Dietrich H, Selle A, Teuber M, Hänsch R, Gessler A (2007) Interaction of nitrogen nutrition and salinity in Grey poplar (Populus tremula × alba). Plant Cell Environ 30:796–811. doi:10.1111/j.1365-3040.2007.01668.x
Feki K, Quintero FJ, Khoudi H, Leidi EO, Masmoudi K, Pardo JM, Brini F (2014) A constitutively active form of a durum wheat Na+/H+ antiporter SOS1 confers high salt tolerance to transgenic Arabidopsis. Plant Cell Rep 33:277–288. doi:10.1007/s00299-013-1528-9
Fernández-Crespo E, Camañes G, García-Agustín P (2012) Ammonium enhances resistance to salinity stress in citrus plants. J Plant Physiol 169:1183–1191. doi:10.1016/j.jplph.2012.04.011
Frechilla S, Lasa B, Ibarretxe L, Lamsfus C, Aparicio-Tejo P (2001) Pea responses to saline stress is affected by the source of nitrogen nutrition (ammonium or nitrate). Plant Growth Regul 35:171–179. doi:10.1023/A:1014487908495
Garnica M, Houdusse F, Claude YJ, Garcia-Mina JM (2009) Nitrate supply induces changes in polyamine content and ethylene production in wheat plants grown with ammonium. J Plant Physiol 166:363–374. doi:10.1016/j.jplph.2008.06.008
Gill MA, Reisenauer HM (1993) Nature and characterization of ammonium effects on wheat and tomato. Agron J 85:874–879. doi:10.2134/agronj1993.00021962008500040018x
Guo S, Chen G, Zhou Y, Shen Q (2007) Ammonium nutrition increases photosynthesis rate under water stress at early development stage of rice (Oryza Sativa L.). Plant Soil 296:115–124. doi:10.1007/s11104-007-9302-9
Guo XR, Zu YG, Tang ZH (2012) Physiological responses of Catharanthus roseus to different nitrogen forms. Acta Physiol Plant 34:589–598. doi:10.1007/s11738-011-0859-9
Han Y, Yin S, Huang L (2015) Towards plant salinity tolerance-implications from ion transporters and biochemical regulation. Plant Growth Regul 76:13–23. doi:10.1007/s10725-014-9997-6
Hasegawa PM (2013) Sodium (Na+) homeostasis and salt tolerance of plants. Environ Exp Bot 92:19–31. doi:10.1016/j.envexpbot.2013.03.001
Hessini K, Cruz C, Gandour M, Soltani A, Abdelly C (2009) Do reactive oxygen species (ROS) induced by NaCl contribute to ammonium accumulation in Spartina alterniflora? J Plant Nutr Soil Sc 172:851–860. doi:10.1002/jpln.200800315
Hessini K, Hamed KB, Gandour M, Mejri M, Abdelly C, Cruz C (2013) Ammonium nutrition in the halophyte Spartina alterniflora under salt stress: evidence for a priming effect of ammonium? Plant Soil 370:163–173. doi:10.1007/s11104-013-1616-1
Hoagland DR, Arnon DI (1950) The water culture method for growing plants without soil. Calif Agric Exp Sta Circ 347:1–39
Jiang C, Zheng Q, Liu Z, Xu W, Liu L, Zhao G, Long X (2012) Overexpression of Arabidopsis thaliana Na+/H+ antiporter gene enhanced salt resistance in transgenic poplar (Populus X Euramericana ‘Neva’). Trees 26:685–694. doi:10.1007/s00468-011-0635-x
Kant S, Kant P, Lips H, Barak S (2007) Partial substitution of NO3 − by NH4 + fertilization increases ammonium assimilating enzyme activities and reduces the deleterious effects of salinity on the growth of barley. J Plant Physiol 164:303–311. doi:10.1016/j.jplph.2005.12.011
Lasa B, Frechilla S, Aparicio-Tejo PM, Lamsfus C (2002) Role of glutamate dehydrogenase and phosphoenolpyruvate carboxylase activity in ammonium nutrition tolerance in roots. Plant Physiol Biochem 40:969–976. doi:10.1016/S0981-9428(02)01451-1
Li Y, Gao Y, Ding L, Shen Q, Guo S (2009) Ammonium enhances the tolerance of rice seedlings (Oryza sativa L.) to drought condition. Agr Water Manage 96:1746–1750. doi:10.1016/j.agwat.2009.07.008
Li Q, Li BH, Kronzucker HJ, Shi WM (2010) Root growth inhibition by NH4 + in Arabidopsis is mediated by the root tip and is linked to NH4 + efflux and GMPase activity. Plant Cell Environ 34:933–946. doi:10.1111/j.1365-3040.2010.02162.x
Li D, Tian M, Cai J, Jiang D, Cao W, Dai T (2013) Effects of low nitrogen supply on relationships between photosynthesis and nitrogen status at different leaf position in wheat seedlings. Plant Growth Regul 70:257–263. doi:10.1007/s10725-013-9797-4
Lu YX, Li CJ, Zhang FS (2005) Transpiration, potassium uptake and flow in tobacco as affected by nitrogen forms and nutrient levels. Ann Bot 95:991–998. doi:10.1093/aob/mci104
M’rah S, Nebli H, Kaddour R, Mahmoudi H, Lachaâl M, Ouerghi Z (2010) Influence of nitrate-ammonium ratio on growth and nutrition of Arabidopsis thaliana. Plant Soil 336:65–74. doi:10.1007/s11104-010-0445-8
Mansour MMF (2014) The plasma membrane transport systems and adaptation to salinity. J Plant Physiol 171:1787–1800. doi:10.1016/j.jplph.2014.08.016
Marschner H (1995) Mineral nutrition of higher plants. Academic Press, London, p 889
Martínez-Andújar C, Ghanem ME, Albacete A, Pérez-Alfocea F (2013) Response to nitrate/ammonium nutrition of tomato (Solanum lycopersicum L.) plants overexpressing a prokaryotic NH4 +-dependent asparagine synthetase. J Plant Physiol. doi:10.1016/j.jplph.2012.12.011
Miranda RS, Alvarez-Pizarro JC, Araújo CMS, Prisco JT, Gomes-Filho E (2013) Influence of inorganic nitrogen sources on K+/Na+ homeostasis and salt tolerance in sorghum plants. Acta Physiol Plant 35:841–852. doi:10.1007/s11738-012-1128-2
Misra N, Gupta AK (2006) Effect of salinity and different nitrogen sources on the activity of antioxidant enzymes and indole alkaloid content in Catharanthus roseus seedlings. J Plant Physiol 163:11–18. doi:10.1016/j.jplph.2005.02.011
Munns R (2002) Comparative physiology of salt and water stress. Plant Cell Environ 25:239–250. doi:10.1046/j.0016-8025.2001.00808.x
Olías R, Eljakaovi C, Li J, Morales PA, Marín-Manzano MC, Pardo JM, Belver A (2009) The plasma membrane Na+/H+ antiporter SOS1 is essential for salt tolerance in tomato and effects the partitioning of Na+ between plant organs. Plant Cell Environ 32:904–916. doi:10.1111/j.1365-3040.2009.01971.x
Sánchez-Rodríguez E, Rubio-Wilhelmi MM, Ríos JJ, Blasco B, Rosales MÁ, Melgarejo R, Ruiz JM (2011) Ammonia production and assimilation: its importance as a tolerance mechanism during moderate water deficit in tomato plants. J Plant Physiol 168:816–823. doi:10.1016/j.jplph.2010.11.018
Schortemeyer M, Stamp P, Feil B (1997) Ammonium tolerance and carbohydrate status in maize cultivars. Ann Bot Lond 79:25–30. doi:10.1006/anbo.1996.0298
Tester M, Davenport R (2003) Na+ tolerance and Na+ transport in higher plants. Ann Bot Lond 91:503–527. doi:10.1093/aob/mcg058
Türkan I, Demiral T (2009) Recent developments in understanding salinity tolerance. Environ Exp Bot 67:2–9. doi:10.1016/j.envexpbot.2009.05.008
Wang SM, Zhang JL, Liu XS, Li Z, Wu GQ, Cai JY, Flowers TJ, Wang SM (2009) Pucinellia tenuiflora maintains a low Na+ level under salinity by limiting unidirectional Na+ influx resulting in a high selectivity for K+ over Na+. Plant Cell Environ 32:486–496. doi:10.1111/j.1365-3040.2009.01942.x
Weatherburn MW (1967) Phenol-hipochlorite reaction for determination of ammonia. Anal Chem 39:971–974. doi:10.1021/ac60252a045
Xu G, Fan X, Miller AJ (2012) Plant nitrogen assimilation and use efficiency. Annu Rev Plant Biol 63:153–182. doi:10.1146/annurev-arplant-042811-105532
Yemm EW, Cocking EC (1955) The determination of amino-acids with ninhydrin. Analyst 80:209–213. doi:10.1039/AN9558000209
Acknowledgments
The authors are grateful for the financial support provided by the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES), Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) and the Instituto de Ciência e Tecnologia em Salinidade (INCTSal).
Conflict of interest
The authors declare that they have no conflicts of interest.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
de Souza Miranda, R., Gomes-Filho, E., Prisco, J.T. et al. Ammonium improves tolerance to salinity stress in Sorghum bicolor plants. Plant Growth Regul 78, 121–131 (2016). https://doi.org/10.1007/s10725-015-0079-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10725-015-0079-1