Abstract
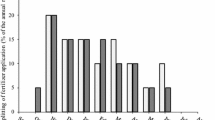
The nitrogen fixing bacterial endophytes Gluconacetobacter diazotrophicus and Herbaspirillum spp. have been proposed to benefit sugarcane (Saccaharum spp. hybrids) growth. Variable populations of these endophytes exist depending upon ontogenic and climatic variations as well. This study investigates the effect of variable chemical nitrogen application in soil on the population of endophytic diazotrophs, acetylene reduction ability of excised roots, plant N-nutrient use efficiency and probable interactions among different parameters in eight commercial sugarcane varieties of subtropical India. Recovery efficiency (RE), agronomic efficiency (AE), partial factor productivity (PFP) and physiologic efficiency (PE) indicators were used for accounting N-nutrient use efficiency. The population of G. diazotrophicus was more at N75 compared to N0 and N150, whereas Herbaspirillum population increased from N0 to N150. ARA was positively correlated with Gluconacetobacter population in rhizosphere and root, whereas it had poor correlation with Herbaspirillum population. Positive correlation of RE and AE with ARA of roots, Gluconacetobacter and Herbaspirillum populations in roots and stems indicate their positive contribution in total nitrogen uptake by the plant per kg of N applied. Average PFP was 808.9 at N75 compared to 408.7 at N150 indicating that N was utilized efficiently at low N input status in sugarcane. Strong positive correlations of AE75 (agronomic efficiency from 75 kg N ha−1 to 150 kg N ha−1) with N-uptake (r 2 = 0.615), cane yield (r 2 = 0.758) and PFP (r 2 = 0.758) and other parameters compared to AE (agronomic efficiency from 0 kg N ha−1 to 75 kg N ha−1 or 150 kg N ha−1) correlations with N-uptake (r 2 = 0.111), cane yield (r 2 = 0.368) and PFP (r 2 = 0.190) indicated that the AE of sugarcane was strongly directed towards producing more cane yield per unit of N fertilizer once the sugarcane plant has established using initial dose of nitrogen and thus AE75 seems to be a more appropriate indicator for accounting N-nutrient use efficiency in sugarcane.

Similar content being viewed by others
References
Baldani IJ, Baldani VLD, Seldin L, Döbereiner J (1986) Characterization of Herbaspirillum seropedicae gen. nov., sp. nov., a root associated nitrogen-fixing bacterium. Int J Syst Bacteriol 36:86–93
Baldani VLD, Baldani IJ, Olvivares FL, Dobereiner J (1992) Identification and ecology of Herbaspirillum seopedicae and the closely related Pseudomonas rubrisubalbicans. Symbiosis 19:65–73
Baldani IJ, Reis VM, Baldani VLD, Dobereiner J (2002) A brief story of nitrogen fixation in sugarcane—reasons for success in Brazil. Funct Plant Biol 29:417–423
Boddey RM (1987) Methods for quantification of nitrogen fixation associated with Gramineae. Crit Rev Plant Sci 6:209–266
Boddey RM, de Oliveira OC, Urquiaga S, Reis VM, Olivares FL, Baldani VLD, Döbereiner J (1995). Biological nitrogen fixation associated with sugarcane and rice: contributions and prospects for improvement. Plant Soil 174:95–209
Bremner JM, Keeney DR (1965) Steam distillation methods for determination of ammonium, nitrate and nitrite. Anal Chem Acta 32:485–495
Cavalcante VA, Döbereiner J (1988) A new acid-tolerant nitrogen fixing bacterium associated with sugarcane. Plant Soil 108:23–31
Döbereiner J Baldani VLD, Reis VM (1995) Endophytic occurrence of diazotrophic bacteria in non-leguminous crops. In: Fendrik I, del Gallo M, Vanderleyden J, de Zamaroczy M (eds) Azospirillum VI and related microorganisms. Springer-Verlag, Heidelberg, Berlin
Fuentes-Ramirez LE, Jimnez-Salgado T, Abarca-Ocampo IR, Caballero-Mellado J (1993) Acetobacter diazotrophicus, an indoleacetic acid producing bacterium isolation from sugarcane cultivars of Mexico. Plant Soil 154:145–150
Good AG, Shrawat AK, Muench DG (2004) Can less yield more? Is reducing nutrient input into the environment compatible with maintaining crop production? Rev Plant Sci 12:597–605
Hardy RWF, Burns RC, Holston RD (1973) Application of the C2H4 assay for measurement of nitrogen fixation. Soil Biol Biochem 24:47–81
Handelsman J (2004) Metagenomics: application of genomics to uncultured microorganisms. Microbiol Mol Biol Rev 68:669–685
Hoefsloot G, Termorshuizen AJ, Watt DA, Cramer MD (2005) Biological nitrogen fixation is not a major contributor to the nitrogen demand of a commercial grown South African sugarcane cultivar. Plant Soil 277:85–96
James EK, Reis VM, Olivares F, Baldani JI, Döbereiner J (1994) Infection of sugarcane by the nitrogen fixing bacterium Acetobacter diazotrophicus. J Exp Bot 45:757–766
James EK, Oliveres FL (1998) Infection and colonization of sugarcane and other graminaceous plants by endophytic diazotrophs. Crit Rev Plant Sci 17:77–119
James EK, Oliveres FL, Olivera ALM, Reis B Jr, Silva LG, Reis VM (2001) Further observations on the interaction between sugarcane and Gluconacetobacter diazotrophicus under laboratory and greenhouse conditions. J Exp Bot 52:747–760
Leininger S, Urich T, Schloter M, Schwark L, Qi J, Nicol GW, Prosser JI, Schuster SC, Schleper C (2006) Archaea predominate among ammonia oxidizing prokaryotes in soils. Nature 442:806–809
Li RP, MacRae IC (1991) Specific association of diazotrophic acetobacters with sugarcane. Soil Biol Biochem 23:999–1002
Muthukumarasamy R, Revathi G, Lakshminarasimhan C (1999a) Diazotrophic association in sugarcane cultivation in South India. Trop Agric (Trinidad) 76:171–178
Muthukumarasamy R, Revathi G, Lakshminarasimhan C (1999b) Influence of N-fertilization on the isolation of Acetobacter diazotrophicus and Herbaspirillum spp. from Indian sugarcane varieties. Biol Fertil Soils 29:157–164
Muthukumarasamy R, Revathi G, Seshadri S, Lakshminarasimhan C (2002a) Gluconacetobacter diazotrophicus (syn. Acetobacter diazotrophicus), a promising diazotrophic endophyte in tropics. Curr Sci 83:137–145
Muthukumarasamy R, Revathi G, Loganathan P (2002b) Effect of inorganic N on the population, in vitro colonization and morphology of Acetobacter diazotrophicus (syn. Gluconacetobacter diazotrophicus). Plant Soil 243:91–102
Olivares FL, Baldani VLD, Reis VM, Baldani JI, Dobereiner J (1996) Occurrence of endophytic diazotroph Herbaspirillum spp. in roots, stems and leaves predominantly in gramineae. Biol Fertil Soils 21:197–200
Oliveira ALM, Urquiaga S, Döbereiner J, Baldani JI (2002) The effect of inoculating endophytic N2-fixing bacteria on micropropagated sugarcane plants. Plant Soil 242:205–215
Page AL, Miller RH, Keeney DR (eds) (1982) Methods of soil analysis. Part 2: Chemical and microbiological properties. American Society of Agronomy, Madison, Wisconsin, USA
Reis dos FB Jr, Reis VM, Urquiaga S, Döbereiner J (2000) Influence of nitrogen fertilization on the population of diazotrophic Herbaspirillum spp. and Gluconacetobacter diazotrophicus in sugarcane (Saccharum spp.). Plant Soil 219:153–159
Sevilla M, de Oliveira A, Baldani I, Kennedy C (1998) Contributions of the bacterial endophyte Acetobacter diazotrophicus to sugarcane nutrition: a preliminary study. Symbiosis 25:181–191
Snedecor GW, Cochran WG (1967). Statistical methods. Oxford and IBH Publishing Co., India
Suman A, Shasany AK, Singh M, Shahi HN, Gaur A, Khanuja SPS (2001) Molecular assessment of diversity among endophytic diazotrophs isolated from subtropical Indian sugarcane. World J Microbiol Biotechnol 17:39–45
Suman A, Gaur A, Shrivastava AK, Yadav RL (2005) Improving sugarcane growth and nutrient uptake by inoculating Gluconacetobacter diazotrophicus. Plant Growth Regul 47:155–162
Urquiaga S, Cruz KHS, Boddey RM (1992). Contribution of nitrogen fixation to sugarcane: nitrogen 15 and nitrogen balance estimates. Soil Sci Soc Am J 56:105–114
Walkley A, Black IA (1934) An examination of the Degtjareff method for determining soil organic matter and a proposed modification of the chromic acid titration method. Soil Sci 37:29–38
Yoneyama T, Muraoka T, Kim TH, Dacanay EV, Nakanishi Y (1997) The natural 15N abundance of sugarcane and neighboring plants in Brazil, the Philippines and Miyako (Japan). Plant Soil 189:239–244
Acknowledgements
Authors thank Dr. Rajesh Verma and Mr. Ashok, for their help in the experimentation and analysis. Financial assistance by Department of Biotechnology, New Delhi, through INM project is gratefully acknowledged.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Suman, A., Shrivastava, A.K., Gaur, A. et al. Nitrogen use efficiency of sugarcane in relation to its BNF potential and population of endophytic diazotrophs at different N levels. Plant Growth Regul 54, 1–11 (2008). https://doi.org/10.1007/s10725-007-9219-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10725-007-9219-6