Abstract
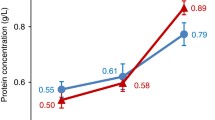
Currently, the definitive diagnosis in breast cancer requires biopsy and histopathology, such the most effective markers are tissue-based. However, the advantages of saliva in collection and storage make it possible for assessing human pathology and contributing to the development of cancer-related biomarkers for clinical application. The present study validated alteration of salivary protein glycopatterns recognized by Bandeiraea simplicifolia lectin I (BS-I) in the saliva of patients with breast diseases using saliva microarrays, and the N/O-glycan profiles of their salivary glycoproteins isolated by the BS-I-magnetic particle conjugates from 259 female subjects (66 healthy volunteers (HV), 65 benign breast cyst or tumor patients (BB), 66 patients with breast cancer in stage I (BC-I) and 62 patients with breast cancer in stage II (BC-II)) were analyzed by MALDI-TOF/TOF-MS. The results showed that the expression level of galactosylated glycans recognized by BS-I was significantly increased in patients with breast cancer compared with HV (p < 0.05). Totally, there were 11/10, 10/19, 7/24 and 7/9 galactosylated N-/O-linked glycans were identified and annotated from the pooled salivary samples of HV, BB, BC-I and BC-II, respectively. One galactosylated N-glycan peak (m/z 2773.977), and 4 galactosylated O-glycan peaks (m/z 868.295, 882.243, 884.270 and 1030.348) were found only in BC-I. These findings could provide pivotal information on galactosylated N/O-linked glycans related to breast cancer, and promote the study of biomarkers for early-stage breast cancer based on precise alterations of galactosylated N/O-glycans in saliva.







Similar content being viewed by others
References
Chen, W., Zheng, R., Baade, P.D., Zhang, S., Zeng, H., Bray, F., Jemal, A., Yu, X.Q., He, J.: Cancer statistics in China, 2015. CA Cancer J. Clin. 66(2), 115–132 (2016)
Sturgeon, C.M., Duffy, M.J., Stenman, U.H., Lilja, H., Brünner, N., Chan, D.W., Babaian, R., Bast RC Jr, Dowell, B., Esteva, F.J., Haglund, C., Harbeck, N., Hayes, D.F., Holten-Andersen, M., Klee, G.G., Lamerz, R., Looijenga, L.H., Molina, R., Nielsen, H.J., Rittenhouse, H., Semjonow, A., Shih IeM, Sibley, P., Sölétormos, G., Stephan, C., Sokoll, L., Hoffman, B.R., Diamandis, E.P., National Academy of Clinical Biochemistry: National Academy of Clinical Biochemistry laboratory medicine practice guidelines for use of tumor markers in testicular, prostate, colorectal, breast, and ovarian cancers. Clin. Chem. 54(12), e11–e79 (2008)
Smith, R.A., Andrews, K., Brooks, D., et al.: Cancer screening in the United States, 2016: a review of current American Cancer Society guidelines and current issues in cancer screening. CA Cancer J. Clin. 66(2), 69–114 (2016)
Marmot, M.G., Altman, D.G., Cameron, D.A., Dewar, J.A., Thompson, S.G., Wilcox, M.: The benefits and harms of breast cancer screening: an independent review. Br. J. Cancer. 108(11), 2205–2240 (2013)
Corbex, M., Burton, R., Sancho-Garnier, H.: Breast cancer early detection methods for low and middle income countries, a review of the evidence. Breast. 21(4), 428–434 (2012)
Ohtsubo, K., Marth, J.D.: Glycosylation in cellular mechanisms of health and disease. Cell. 126(5), 855–867 (2006)
Lebrilla, C.B., An, H.J.: The prospects of glycan biomarkers for the diagnosis of diseases. Mol. BioSyst. 5(1), 17–20 (2009)
Pinho, S.S., Reis, C.A.: Glycosylation in cancer: mechanisms and clinical implications. Nat. Rev. Cancer. 15(19), 540–555 (2015)
Kaur, S., Kumar, S., Momi, N., et al.: Mucins in pancreatic cancer and its microenvironment. Nat Rev Gastroenterol Hepatol. 10(10), 607–620 (2013)
Drake, R.R.: Glycosylation and cancer: moving glycomics to the forefront. Adv. Cancer Res. 126, 1–10 (2015)
RodrÍguez, E., Schetters, S.T.T., van Kooyk, Y.: The tumour glyco-code as a novel immune checkpoint for immunotherapy. Nat Rev Immunol. 18(3), 204–211 (2018)
Sun, S., Shah, P., Eshgh, S.T., et al.: Comprehensive analysis of protein glycosylation by solid-phase extraction of N-linked glycans and glycosite-containing peptides. Nat Biotechnol. 34(1), 84–88 (2016)
Calafat, J., Janssen, H.: Binding of lectins to human mammary tumors: ultrastructural study. Breast Cancer Res Tr. 4(3), 169–179 (1984)
Springer, G.F.: Immunoreactive T and Tn epitopes in cancer diagnosis, prognosis, and immunotherapy. J. Mol. Med. 75(8), 594–602 (1997)
Burchell, J., Durbin, H., Taylor-Papadimitriou, J.: Complexity of expression of antigenic eterminants, recognized by monoclonal antibodies HMFG-1 and HMFG-2, in normal and malignant human mammary epithelial cells. J. Immunol. 131(1), 508–513 (1983)
Jeschke, U.: Can we find breast Cancer via salivary fluid glycosylation analyses? EbioMedicine. 28, 4 (2018)
Gillece-Castro, B.L., Prakobphol, A., Burlingame, A.L., et al.: Structure and bacterial receptor activity of a human salivary proline-rich glycoprotein. J. Biol. Chem. 266(26), 17358–17368 (1991)
Paige, S.Z., Streckfus, C.F.: Salivary analysis in the diagnosis and treatment of breast cancer: a role for the general dentist. Gen. Dent. 55(2), 156–157 (2017)
Ozturk, L.K., Emekli-Alturfan, E., Kasikci, E., et al.: Salivary total sialic acid levels increase in breast cancer patients: a preliminary study. Med. Chem. 7(5), 443–447 (2011)
Scott, D.A., Casadonte, R., Cardinali, B., Spruill, L., Mehta, A.S., Carli, F., Simone, N., Kriegsmann, M., del Mastro, L., Kriegsmann, J., Drake, R.R.: Increases in tumor N-glycan Polylactosamines associated with advanced HER2-positive and triple-negative breast Cancer tissues. Proteomics Clin Appl. 13(1), e1800014 (2019)
Liu, X., Yu, H., Qiao, Y., Yang, J., Shu, J., Zhang, J., Zhang, Z., He, J., Li, Z.: Salivary Glycopatterns as potential biomarkers for screening of early-stage breast Cancer. EbioMedicine. 28, 70–79 (2018)
Kisailus, E.C., Kabat, E.A.: A study of the specificity of Bandeiraea simplicifolia lectin I by competitive-binding assay with blood-group substances and with blood-group a and B active and other oligosaccharides. Carbohydr. Res. 67(1), 243–255 (1978)
Wu, A.M., Song, S.C., Wu, J.H., et al.: Affinity of Bandeiraea (Griffonia) simplicifolia lectin-I, isolectin B4 for Gal alpha 1–4 Gal ligand. Biochem Biophys Res Commun. 216(3), 814–820 (1995)
Qin, Y., Zhong, Y., Zhu, M., Dang, L., Yu, H., Chen, Z., Chen, W., Wang, X., Zhang, H., Li, Z.: Age- and sex-associated differences in the glycopatterns of human salivary glycoproteins and their roles against influenza a virus. J. Proteome Res. 12(6), 2742–2754 (2013)
Zhang, J., Zhong, Y., Zhang, P., du, H., Shu, J., Liu, X., Zhang, H., Guo, Y., Jia, Z., Niu, L., Yang, F., Li, Z.: Identification of abnormal Fucosylated-Glycans recognized by LTL in saliva of HBV-induced chronic hepatitis, cirrhosis, and hepatocellular carcinoma. Glycobiology. 29(3), 242–259 (2019)
Yang, G., Cui, T., Wang, Y., Sun, S., Ma, T., Wang, T., Chen, Q., Li, Z.: Selective isolation and analysis of glycoprotein fractions and their glycomes from hepatocellular carcinoma sera. Proteomics. 13(9), 1481–1498 (2013)
Yang, G., Cui, T., Chen, Q., Ma, T., Li, Z.: Isolation and identification of native membrane glycoproteins from living cell by Concanavalin A-magnetic particle conjugates. Anal. Biochem. 421(1), 339–341 (2012)
Qin, Y., Zhong, Y., Ma, T., et al.: A pilot study of salivary N-glycome in HBV-induced chronic hepatitis, cirrhosis, and hepatocellular carcinoma. Glycoconj. J. 34(4), 523–535 (2016)
Song, X., Ju, H., Lasanajak, Y., Kudelka, M.R., Smith, D.F., Cummings, R.D.: Oxidative release of natural glycans for functional glycomics. Nat. Methods. 13(6), 528–534 (2016)
Ranzinger, R., York, W.S.: GlycomeDB. Methods Mol Biol. 1273, 109–124 (2014)
Damerell, D., Ceroni, A., Maass, K., et al.: Annotation of glycomics MS and MS/MS spectra using the GlycoWorkbench software tool. Methods Mol. Biol. 1273, 3–15 (2015)
Varki, A., Cummings, R.D., Aebi, M., et al.: Symbol nomenclature for graphical representations of glycans. Glycobiology. 25(12), 1323–1324 (2015)
Sh Ju, Y.H., Du, H., et al.: Identification of N- and O-linked glycans recognized by AAL in saliva of patients with atrophic gastritis and gastric cancer. Cancer Biomark. 22(4), 669–681 (2018)
Brooks, S.A., Leathem, A.J.: Expression of alpha-GalNAc glycoproteins by breast cancers. Br. J. Cancer. 71(5), 1033–1038 (1995)
Lomax-Browne, H.J., Robertson, C., Antonopoulos, A., Leathem, A.J.C., Haslam, S.M., Dell, A., Dwek, M.V.: Serum IgA1 shows increased levels of α2,6-linked sialic acid in breast cancer. Interface Focus. 9(2), 20180079 (2019)
Plomp, R., de Haan, H., Bondt, A., et al.: Comparative Glycomics of immunoglobulin a and G from saliva and plasma reveals biomarker potential. Front. Immunol. 9, 2436 (2019)
Parekh, R.B., Dwek, R.A., Sutton, B.J., Fernandes, D.L., Leung, A., Stanworth, D., Rademacher, T.W., Mizuochi, T., Taniguchi, T., Matsuta, K.: Association of rheumatoid arthritis and primary osteoarthritis with changes in the glycosylation pattern of total serum IgG. Nature. 316(6027), 452–457 (1985)
Martin, K., Talukder, R., Hay, F.C., et al.: Characterization of changes in IgG associated oligosaccharide profiles in rheumatoid arthritis, psoriatic arthritis, and ankylosing spondylitis using fluorophore linked carbohydrate electrophoresis. J Rheumatol. 28(7), 1531–1536 (2001)
Ren, S., Zhang, Z., Xu, C., Guo, L., Lu, R., Sun, Y., Guo, J., Qin, R., Qin, W., Gu, J.: Distribution of IgG galactosylation as a promising biomarker for cancer screening in multiple cancer types. Cell Res. 26(8), 963–966 (2016)
Axford, J.S., Sumar, N., Alavi, A., Isenberg, D.A., Young, A., Bodman, K.B., Roitt, I.M.: Changes in normal glycosylation mechanisms in autoimmune rheumatic disease. J. Clin. Invest. 89(3), 1021–1031 (1992)
Bones, J., Byrne, J.C., O'Donoghue, N., McManus, C., Scaife, C., Boissin, H., Nastase, A., Rudd, P.M.: Glycomic and glycoproteomic analysis of serum from patients with stomach cancer reveals potential markers arising from host defense response mechanisms. J. Proteome Res. 10(3), 1246–1265 (2011)
Kolarich, D., Rapp, E., Struwe, W.B., Haslam, S.M., Zaia, J., McBride, R., Agravat, S., Campbell, M.P., Kato, M., Ranzinger, R., Kettner, C., York, W.S.: The minimum information required for a glycomics experiment (MIRAGE) project: improving the standards for reporting mass-spectrometry-based glycoanalytic data. Mol. Cell. Proteomics. 12(4), 991–995 (2013)
York, W.S., Agravat, S., Aokikinoshita, K.F., et al.: Mirage: the minimum information required for a glycomics experiment. Glycobiology. 24(5), 402–406 (2014)
Guile, G.R., Harvey, D.J., O'Donnell, N., Powell, A.K., Hunter, A.P., Zamze, S., Fernandes, D.L., Dwek, R.A., Wing, D.R.: Identification of highly fucosylated N-linked oligosaccharides from the human parotid gland. Eur. J. Biochem. 258(2), 623–656 (1998)
Marino, K., Saldova, R., Adamczyk, B., et al.: Changes in serum N-glycosylation profiles: functional significance and potential for diagnostics. Carbohydr. Chem. 37, 57–93 (2012)
Saldova, R., Asadi Shehni, A., Haakensen, V.D., et al.: Association of N-glycosylation with breast carcinoma and systemic features using high-resolution quantitative UPLC. J Proteome Res. 13(5), 2314–2327 (2014)
Christiansen, M.N., Chik, J., Lee, L., Anugraham, M., Abrahams, J.L., Packer, N.H.: Cell surface protein glycosylation in cancer. Proteomics. 14(4–5), 525–546 (2014)
Arnold, J.N., Saldova, R., Hamid, U.M., Rudd, P.M.: Evaluation of the serum N-linked glycome for the diagnosis of cancer and chronic inflammation. Proteomics. 8(16), 3284–3293 (2008)
Pinho, S.S., Reis, C.A.: Glycosylation in cancer: mechanisms and clinical implications. Nat. Rev. Cancer. 15(9), 540–555 (2015)
Kim, Y.J., Varki, A.: Perspectives on the significance of altered glycosylation of glycoproteins in cancer. Glycoconj. J. 14(5), 569–576 (1997)
Munkley, J.: The role of Sialyl-Tn in Cancer. Int. J. Mol. Sci. 17(3), 275 (2016)
Kannagi, R., Yin, J., Miyazaki, K., et al.: Current relevance of incomplete synthesis and neo-synthesis for cancer-associated alteration of carbohydrate determinants—Hakomori’s concepts revisited. Biochim. Biophys. Acta. 1780(3), 525–531 (2008)
Gill, D.J., Chia, J., Senewiratne, J., Bard, F.: Regulation of O-glycosylation through Golgi-to-ER relocation of initiation enzymes. J. Cell Biol. 189(5), 843–858 (2010)
Pinho, S.S., Reis, C.A.: Glycosylation in cancer: mechanisms and clinical implications. Nat. Rev. Cancer. 15(9), 540–555 (2015)
Hakomori, S., Kannagi, R.: Glycosphingolipids as tumor-associated and differentiation markers. J. Natl. Cancer Inst. 71(2), 231–251 (1983)
Marcos, N.T., Bennett, E.P., Gomes, J., et al.: ST6GalNAc-I controls expression of sialyl-Tn antigen in gastrointestinal tissues. Front. Biosci. (Elite Ed). 3(1443–1455), (2011)
Julien, S., Adriaenssens, E., Ottenberg, K., Furlan, A., Courtand, G., Vercoutter-Edouart, A.S., Hanisch, F.G., Delannoy, P., le Bourhis, X.: ST6GalNAc I expression in MDA-MB-231 breast cancer cells greatly modifies their O-glycosylation pattern and enhances their tumourigenicity. Glycobiology. 16(1), 54–64 (2006)
Funding
This work was supported by the National Natural Science Foundation of China (81372365).
Author information
Authors and Affiliations
Contributions
JY, XL and ZL designed research; JY, LX, HY, JS, MC, HD, JZ, YQ, JH, LN, TM, YH and FY performed research; JY, XL and ZL analyzed data; JY, XL and ZL wrote the paper.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Yang, J., Liu, X., Shu, J. et al. Abnormal Galactosylated–Glycans recognized by Bandeiraea Simplicifolia Lectin I in saliva of patients with breast Cancer. Glycoconj J 37, 373–394 (2020). https://doi.org/10.1007/s10719-020-09910-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10719-020-09910-6