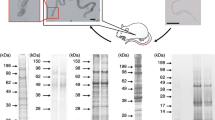
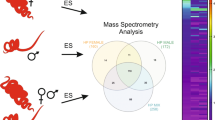
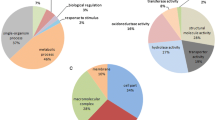
Abstract
Parasitic helminths and pest insects are organisms with great ecological importance, having direct or indirect detrimental effects on people’s lives worldwide. Several reports in literature indicate that the glycan repertoire of parasites plays important roles in host-parasite interactions and modulation and evasion of the host immune system, while insect glycans are essential for their survival, growth and development. Although glycosylation is the result of a highly conserved machinery, differences between species and between different stages of one organism’s life cycle occur. This review provides insight into recent glycomics studies both for helminths and insects, focussing on sex differences and the role of carbohydrate structures in reproduction. Information on the differential N-glycosylation process between males and females can generate a better understanding of the biology and physiology of these economic important organisms, and can contribute to the discovery of novel anti-fecundity vaccine candidates and drug targets, as well as in the elaboration of innovative pest management strategies.

Similar content being viewed by others
References
van Die, I., Cummings, R.D.: Glycan gimmickry by parasitic helminths: a strategy for modulating the host immune response? Glycobiology. 20, 2–12 (2010)
Jiménez-Castells, C., Vanbeselaere, J., Kohlhuber, S., Ruttkowski, B., Joachim, A., Paschinger, K.: Gender and developmental specific N-glycomes of the porcine parasite Oesophagostomum dentatum. Biochim. Biophys. Acta Gen. Subj. (2016). https://doi.org/10.1016/j.bbagen.2016.10.011
Ghosh, S.: Sialylation and sialyltransferase in insects. Glycoconj. J. 35, 433–441 (2018)
Hokke, C.H., van Diepen, A.: Helminth glycomics – glycan repertoires and host-parasite interactions. Mol. Biochem. Parasitol. 215, 47–57 (2016)
Walski, T., De Schutter, K., Van Damme, E.J.M., Smagghe, G.: Diversity and functions of protein glycosylation in insects. Insect Biochem. Mol. Biol. (2017). https://doi.org/10.1016/j.ibmb.2017.02.005
Prasanphanich, N.S., Mickum, M.L., Heimburg-Molinaro, J., Cummings, R.D.: Glycoconjugates in host-helminth interactions. Front. Immunol. (2013). https://doi.org/10.3389/fimmu.2013.00240
Schiller, B., Hykollari, A., Yan, S., Paschinger, K., Wilson, I.B.H.: Complicated N-linked glycans in simple organisms. Biol. Chem. 393, 661–673 (2012)
Walski, T., Van Damme, E.J.M., Smargiasso, N., Christiaens, O., De Pauw, E., Smagghe, G.: Protein N-glycosylation and N-glycan trimming are required for postembryonic development of the pest beetle Tribolium castaneum. Sci. Rep. (2016). https://doi.org/10.1038/srep35151
Katoh, T., Tiemeyer, M.: The N’s and O’s of Drosophila glycoprotein glycobiology. Glycoconj. J. 30, 57–66 (2013)
North, S.J., Koles, K., Hembd, C., Morris, H.R., Dell, A., Panin, V.M., Haslam, S.M.: Glycomic studies of Drosophila melanogaster embryos. Glycoconj. J. 23, 345–354 (2006)
Fabini, G., Freilinger, A., Altmann, F., Wilson, I.B.: Identification of core α1,3-fucosylated glycans and cloning of the requisite fucosyltransferase cDNA from Drosophila melanogaster. J. Biol. Chem. 276, 28058–28067 (2001)
Rendić, D., Sharrow, M., Katoh, T., Overcarsh, B., Nguyen, K., Kapurch, J., Aoki, K., Wilson, I.B.H., Tiemeyer, M.: Neural-specific α3-fucosylation of N-linked glycans in the Drosophila embryo requires fucosyltransferase A and influences developmental signaling associated with O-glycosylation. Glycobiology. 20, 1353–1365 (2010)
Wuhrer, M., Koeleman, C.A.M., Fitzpatrick, J.M., Hoffman, K.F., Deelder, A.M., Hokke, C.H.: Gender-specific expression of complex-type N-glycans in schistosomes. Glycobiology. 16, 991–1006 (2006)
Kim, B., Suo, B., Emmons, S.W.: Gene function prediction based on developmental transcriptomes of the two sexes in C. elegans. Cell Rep. 17, 917–928 (2016)
Loukas, A., Mullin, N.P., Tetteh, K.K.A., Moens, L., Maizels, R.M.: A novel C-type lectin secreted by a tissue-dwelling parasitic nematode. Curr. Biol. 9, 825–828 (1999)
Loukas, A., Maizels, R.M.: Helminth C-type lectins and host-parasite interactions. Parasitol. Today. 16, 333–339 (2000)
Brown, A.C., Harrison, L.M., Kapulkin, W., Jones, B.F., Sinha, A., Savage, A., Villalon, N., Cappello, M.: Molecular cloning and characterization of a C-type lectin from Ancylostoma ceylanicum: evidence for a role in hookworm reproductive physiology. Mol. Biochem. Parasitol. 151, 141–147 (2007)
Vanbeselaere, J., Yan, S., Joachim, A., Paschinger, K., Wilson, I.B.H.: The parasitic nematode Oesophagostomum dentatum synthesizes unusual glycosaminoglycan-like O-glycans. Glycobiology. 28, 474–481 (2018)
Martini, F., Eckmair, B., Štefanić, S., Jin, C., Garg, M., Yan, S., Jiménez-Castells, C., Hykollari, A., Neupert, C., Venco, L., Silva, D.V., Wilson, I.B.H., Paschinger, K.: Highly modified and immunoactive N-glycans of the canine heartworm. Nat. Commun. (2019). https://doi.org/10.1038/s41467-018-07948-7
Cai, P., Liu, S., Piao, X., Hou, N., Gobert, G.N., McManus, D.P., Chen, Q.: Comprehensive transcriptome analysis of sex-biased expressed genes reveals discrete biological and physiological features of male and female Schistosoma japonicum. PLoS Negl. Trop. Dis. (2016). https://doi.org/10.1371/journal.pntd.0004684
Hokke, C.H., Fitzpatrick, J.M., Hoffman, K.F.: Integrating transcriptome, proteome and glycome analyses of Schistosoma biology. Trends Parasitol. (2007). https://doi.org/10.1016/j.pt.2007.02.007
Fitzpatrick, J.M., Johnston, D.A., Williams, G.W., Williams, D.J., Freeman, T.C., Dunne, D.W., Hoffmann, K.F.: An oligonucleotide microarray for transcriptome analysis of Schistosoma mansoni and its application/use to investigate gender-associated gene expression. Mol. Biochem. Parasitol. 141, 1–13 (2005)
Liu, F., Lu, J., Hu, W., Wang, S., Cui, S., Chi, M., Yan, Q., Wang, X., Song, H., Xu, X., Wang, J., Zhang, X., Zhang, X., Wang, Z., Xue, C., Brindley, P.J., McManus, D.P., Yang, P., Feng, Z., Chen, Z., Han, Z.: New perspectives on host-parasite interplay by comparative transcriptomic and proteomic analysis of Schistosoma japonicum. PLoS Pathog. (2006). https://doi.org/10.1371/journal.ppat.0020029
Zhang, M., Hong, Y., Han, Y., Han, H., Peng, J., Qiu, C., Yang, J., Lu, K., Fu, Z., Lin, J.: Proteomic analysis of tegument-exposed proteins of female and male Schistosoma japonicum worms. J. Proteome Res. 12, 5260–5270 (2013)
Khoo, K., Chatterjee, D., Caulfield, J.P., Morris, H.R., Dell, A.: Structural mapping of the glycans from the egg glycoproteins of Schistosoma mansoni and Schistosoma japonicum: identification of novel core structures and terminal sequences. Glycobiology. 7, 663–677 (1997)
Smit, C.H., van Diepen, A., Nguyen, D.L., Wuhrer, M., Hoffman, K.F., Deelder, A.M., Hokke, C.H.: Glycomic analysis of life stages of the human parasite Schistosoma mansoni reveals developmental expression profiles of functional and antigenic glycan motifs. Mol. Cell. Proteomics. 14, 1750–1769 (2015)
Sanderson, M.J.: Phylogenetic signal in the eukaryotic tree of life. Science. 321, 121–123 (2008)
Aoki, K., Perlman, M., Lim, J.M., Cantu, R., Wells, L., Tiemeyer, M.: Dynamic developmental elaboration of N-linked glycan complexity in the Drosophila melanogaster embryo. J. Biol. Chem. 282, 9127–9142 (2007)
Ten Hagen, K.G., Zhang, L., Tian, E., Zhang, Y.: Glycobiology on the fly: developmental and mechanistic insights from Drosophila. Glycobiology. 19, 102–111 (2009)
Kurz, S., Aoki, K., Jin, C., Karlsson, N.G., Tiemeyer, M., Wilson, I.B.H., Paschinger, K.: Targeted release and fractionation reveal glucoronylated and sulphated N- and O-glycans in larvae of dipteran insects. J. Proteome. 126, 172–188 (2015)
Dönitz, J., Schmitt-Engel, C., Grossmann, D., Gerischer, L., Tech, M., Schoppmeier, M., Klingler, M., Bucher, G.: iBeetle-base: a database for RNAi phenotypes in the red flour beetle Tribolium castaneum. Nucleic Acids Res. 43, D720–D725 (2014)
Stanton, R., Hykollari, A., Eckmair, B., Malzl, D., Dragosits, M., Palmberger, D., Wang, P., Wilson, I.B.H., Paschinger, K.: The underestimated N-glycomes of lepidopteran species. Biochim. Biophys. Acta. 1861, 699–714 (2017)
Kajihura, H., Hamaguchi, Y., Mizushima, H., Misaki, R., Fujiyama, K.: Sialylation potentials of the silkworm, Bombyx mori; B. mori possesses an active α2,6-sialyltransferase. Glycobiology. 25, 1441–1453 (2015)
Mabashi-Asazuma, H., Sohn, B.H., Kim, Y.S., Kuo, C.W., Khoo, K.H., Kucharski, C.A., Fraser, M.J.J., Jarvis, D.L.: Targeted glycoengineering extends the protein N-glycosylation pathway in the silkworm silk gland. Insect Biochem. Mol. Biol. 65, 20–27 (2015)
Soya, S., Sahar, U., Karaçali, S.: Monosaccharide profiling of silkworm (Bombyx mori L.) nervous system during development and aging. Invertebr. Neurosci. (2016). https://doi.org/10.1007/s10158-016-0191-6
Kubelka, V., Altmann, F., Staudacher, E., Tretter, V., März, L., Hård, K., Kamerling, J.P., Vliegenthart, J.F.: Primary structures of the N-linked carbohydrate chains from honeybee venom phospholipase A2. Eur. J. Biochem. 213, 1193–1204 (1993)
Kubelka, V., Altmann, F., März, L.: The asparagine-linked carbohydrate of honeybee venom hyaluronidase. Glycoconj. J. 12, 77–83 (1995)
Scheys, F., De Schutter, K., Shen, Y., Yu, N., Smargiasso, N., De Pauw, E., Van Damme, E.J.M., Smagghe, G.: The N-glycome of the hemipteran pest insect Nilaparvata lugens reveals unexpected sex differences. Insect Biochem. Mol. Biol. (2019). https://doi.org/10.1016/j.ibmb.2019.01.008
Cattaneo, F., Pasini, M.E., Intra, J., Matsumoto, M., Briani, F., Hoshi, M., Perotti, M.E.: Identification and expression analysis of Drosophila melanogaster genes encoding beta-hexosaminidases of the sperm plasma membrane. Glycobiology. 16, 786–800 (2006)
Cattaneo, F., Pasini, M.E., Perotti, M.E.: Glycosidases are present on the surface of Drosophila melanogaster spermatozoa. Mol. Reprod. Dev. 48, 276–281 (1997)
Cattaneo, F., Ogiso, M., Hoshi, M., Perotti, M.E., Pasini, M.E.: Purification and characterization of the plasma membrane glycosidases of Drosophila melanogaster spermatozoa. Insect Biochem. Mol. Biol. 32, 929–941 (2002)
Intra, J., Cenni, F., Perotti, M.E.: An α-L-fucosidase potentially involved in fertilization is present on Drosophila spermatozoa surface. Mol. Reprod. Dev. 73, 1149–1158 (2006)
Intra, J., De Caro, D., Perotti, M.E., Perotti, M.E.: Glycosidases in the plasma membrane of Ceratitis capitata spermatozoa. Insect Biochem. Mol. Biol. 41, 90–100 (2011)
Pasini, M.E., Intra, J., Gomulski, L.M., Calvenzani, V., Petroni, K., Briani, F., Perotti, M.E.: Identification and expression profiling of Ceratitis capitata genes coding for β-hexosaminidases. Gene. 473, 44–56 (2010)
Stephens, K., Thaler, C.D., Cardullo, R.A.: Characterization of plasma membrane associated type II α-D-mannosidase and β-N-acetylglucosaminidase of Aquarius remigis sperm. Insect Biochem. Mol. Biol. 60, 78–85 (2015)
Intra, J., Cenni, F., Pavesi, G., Pasini, M.E., Perotti, M.E.: Interspecific analysis of the glycosidases of the sperm plasma membrane in Drosophila. Mol. Reprod. Dev. 76, 85–100 (2009)
Perotti, M., Cattaneo, F., Pasini, M.E., Vernì, F., Hackstein, J.H.P.: Male sterile mutant casanova gives clues to mechanisms of sperm-egg interactions in Drosophila melanogaster. Mol. Reprod. Dev. 60, 248–259 (2001)
Intra, J., Veltri, C., De Caro, D., Perotti, M.E., Pasini, M.E.: In vitro evidence for the participation of Drosophila melanogaster sperm β-N-acetylglucosaminidases in the interactions with glycans carrying terminal N-acetylglucosamine residues on the egg's envelopes. Arch. Insect Biochem. Physiol. (2017). https://doi.org/10.1002/arch.21403
Intra, J., Concetta, V., De Caro, D., Perotti, M.E., Pasini, M.E.: Drosophila sperm surface alpha-L-fucosidase interacts with the egg coats through its core fucose residues. Insect Biochem. Mol. Biol. 63, 133–143 (2015)
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
The authors declare that they have no conflicts of interest.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Scheys, F., Van Damme, E.J.M. & Smagghe, G. Let’s talk about sexes: sex-related N-glycosylation in ecologically important invertebrates. Glycoconj J 37, 41–46 (2020). https://doi.org/10.1007/s10719-019-09866-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10719-019-09866-2