Abstract
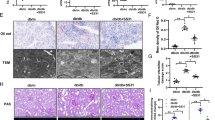
Methylglyoxal (MG), a metabolic intermediate of glycolysis is a precursor for endogeneous production of advanced glycation end-products. The increased production of MG have negative influence over the structure and function of different biomolecules and thus plays an important role in the pathogenesis of diabetic cardiac complications. Retinoic acid (RA), an active metabolite of vitamin A, has a major role in preventing cardiac remodeling and ventricular fibrosis. Hence, the objective of the present study was to determine whether rats administered with all-trans retinoic acid (RA) could attenuate MG induced pathological effects. Wistar rats were divided into 4 groups. Group 1 rats were kept as control; Group 2 rats were administrated with MG (75 mg/kg/day) for 8 weeks. Group 3 rats were given RA (Orally, 1.0 mg/kg/day) along with MG; Group 4 rats received RA alone. Cardiac antioxidant status, induction of fibrosis, AGE receptor (RAGE) and cytokines expression was evaluated in the heart tissues. Administration of MG led to depletion of antioxidant enzymes, induction of fibrosis (p < 0.001), up-regulated expression of RAGE (3.5 fold), TGF-β (4.4 fold), SMAD2 (3.7 fold), SMAD3 (6.0 fold), IL-6 (4.3 fold) and TNF-α (5.5 fold) in the heart tissues compared to control rats. Moreover, the exogenous administration of MG caused significant (p < 0.001) increase in the circulating CML levels. Whereas, RA treatment prevented the induction of fibrosis and restored the levels of cytokines and RAGE expression. Methylglyoxal-induced fibrosis can lead to pathological effects in the heart tissues. RA attenuates the effects of MG in the heart, suggesting that it can be of added value to usual diabetic therapy.








Similar content being viewed by others
Abbreviations
- ALK4, ALK5:
-
Activin receptor-like kinase
- CML:
-
Carboxy methyl lysine
- GA:
-
Gallic acid
- GAPDH:
-
Glyceraldehydes-3-phosphate dehydrogenase
- GSH:
-
Glutathione
- IL-1, IL-6:
-
Interleukin
- TGF-β:
-
Transforming growth factor beta
- TNF-α:
-
Tumor necrosis factor alpha
References
Turk, Z.: Glycotoxines, carbonyl stress and relevance to diabetes and its complications. Physiol. Res. 59(2), 147–156 (2010)
Rodrigues, T., Matafome, P., Santos-Silva, D., Sena, C., Seica, R.: Reduction of methylglyoxal-induced glycation by pyridoxamine improves adipose tissue microvascular lesions. J. Diabetes Res. 2013, 690650 (2013). doi:10.1155/2013/690650
Rabbani, N., Thornalley, P.J.: Methylglyoxal, glyoxalase 1 and the dicarbonyl proteome. Amino Acids 42(4), 1133–1142 (2012). doi:10.1007/s00726-010-0783-0
Price, C.L., Knight, S.C.: Advanced glycation: a novel outlook on atherosclerosis. Curr. Pharm. Des. 13(36), 3681–7 (2007). doi:10.2174/138161207783018608
Negre-Salvayre, A., Salvayre, R., Auge, N., Pamplona, R., Portero-Otin, M.: Hyperglycemia and glycation in diabetic complications. Antioxid. Redox Signal. 11(12), 3071–3109 (2009). doi:10.1089/ARS.2009.2484
Manolescu, D.C., Jankowski, M., Danalache, B.A., Wang, D., Broderick, T.L., Chiasson, J.L., Gutkowska, J.: All-trans retinoic acid stimulates gene expression of the cardioprotective natriuretic peptide system and prevents fibrosis and apoptosis in cardiomyocytes of obese ob/ob mice. Appl. Physiol. Nutr. Metab. 39(10), 1127–1136 (2014). doi:10.1139/apnm-2014-0005
Palm-Leis, A., Singh, U.S., Herbelin, B.S., Olsovsky, G.D., Baker, K.M., Pan, J.: Mitogen-activated protein kinases and mitogen-activated protein kinase phosphatases mediate the inhibitory effects of all-trans retinoic acid on the hypertrophic growth of cardiomyocytes. J. Biol. Chem. 279(52), 54905–54917 (2004). doi:10.1074/jbc.M407383200
Vistoli, G., De Maddis, D., Cipak, A., Zarkovic, N., Carini, M., Aldini, G.: Advanced glycoxidation and lipoxidation end products (AGEs and ALEs): an overview of their mechanisms of formation. Free Radic. Res. 47(Suppl 1), 3–27 (2013). doi:10.3109/10715762.2013.815348
Matafome, P., Sena, C., Seica, R.: Methylglyoxal, obesity, and diabetes. Endocrine 43(3), 472–484 (2013). doi:10.1007/s12020-012-9795-8
Ramasamy, R., Yan, S.F., Schmidt, A.M.: Advanced glycation endproducts: from precursors to RAGE: round and round we go. Amino Acids 42(4), 1151–1161 (2012). doi:10.1007/s00726-010-0773-2
Umadevi, S., Gopi, V., Elangovan, V.: Regulatory mechanism of gallic acid against advanced glycation end products induced cardiac remodeling in experimental rats. Chem. Biol. Interact. 208, 28–36 (2014). doi:10.1016/j.cbi.2013.11.013
Gkogkolou, P., Bohm, M.: Advanced glycation end products: key players in skin aging? Dermatoendocrinol. 4(3), 259–270 (2012). doi:10.4161/derm.22028
Giacco, F., Brownlee, M.: Oxidative stress and diabetic complications. Circ. Res. 107(9), 1058–1070 (2010). doi:10.1161/CIRCRESAHA.110.223545
Larson, R.S., Tallman, M.S.: Retinoic acid syndrome: manifestations, pathogenesis, and treatment. Best Pract. Res. Clin. Haematol. 16(3), 453–461 (2003)
Guo, X., Wang, X.F.: Signaling cross-talk between TGF-beta/BMP and other pathways. Cell Res. 19(1), 71–88 (2009). doi:10.1038/cr.2008.302
Wrighton, K.H., Lin, X., Feng, X.H.: Phospho-control of TGF-beta superfamily signaling. Cell Res. 19(1), 8–20 (2009). doi:10.1038/cr.2008.327
Miyazawa, K., Shinozaki, M., Hara, T., Furuya, T., Miyazono, K.: Two major Smad pathways in TGF-β superfamily signalling. Genes Cell. 7(12), 1191–1204 (2002). doi:10.1046/j.1365-2443.2002.00599.x
Ott, C., Jacobs, K., Haucke, E., Navarrete Santos, A., Grune, T., Simm, A.: Role of advanced glycation end products in cellular signaling. Redox Biol. 2, 411–429 (2014). doi:10.1016/j.redox.2013.12.016
Zhao, J., Randive, R., Stewart, J.A.: Molecular mechanisms of AGE/RAGE-mediated fibrosis in the diabetic heart. World J. Diabetes 5(6), 860–867 (2014). doi:10.4239/wjd.v5.i6.860
Heo, Y.J., Oh, H.J., Jung, Y.O., Cho, M.L., Lee, S.Y., Yu, J.G., Park, M.K., Kim, H.R., Lee, S.H., Park, S.H., Kim, H.Y.: The expression of the receptor for advanced glycation end-products (RAGE) in RA-FLS is induced by IL-17 via Act-1. Arthritis Res. Ther. 13(4), R113 (2011). doi:10.1186/ar3398
Singh, V.P., Bali, A., Singh, N., Jaggi, A.S.: Advanced glycation end products and diabetic complications. Korean J. Physiol. Pharmacol. 18(1), 1–14 (2014). doi:10.4196/kjpp.2014.18.1.1
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interests
The authors declare no conflict of interest.
Ethical approval
Experimental procedures involving the use of animals or animal tissue were performed in accordance with the Indian National Law on animal care and use and approved by the Institutional Animal Ethics Committee at the SASTRA University.
Rights and permissions
About this article
Cite this article
Subramanian, U., Nagarajan, D. All-Trans Retinoic Acid supplementation prevents cardiac fibrosis and cytokines induced by Methylglyoxal. Glycoconj J 34, 255–265 (2017). https://doi.org/10.1007/s10719-016-9760-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10719-016-9760-5