Abstract
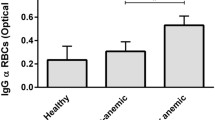
Human C-reactive protein (CRP), as a mediator of innate immunity, removed damaged cells by activating the classical complement pathway. Previous studies have successfully demonstrated that CRPs are differentially induced as glycosylated molecular variants in certain pathological conditions. Affinity-purified CRPs from two most prevalent diseases in India viz. tuberculosis (TB) and visceral leishmaniasis (VL) have differential glycosylation in their sugar composition and linkages. As anemia is a common manifestation in TB and VL, we assessed the contributory role of glycosylated CRPs to influence hemolysis via CRP-complement-pathway as compared to healthy control subjects. Accordingly, the specific binding of glycosylated CRPs with erythrocytes was established by flow-cytometry and ELISA. Significantly, deglycosylated CRPs showed a 7–8-fold reduced binding with erythrocytes confirming the role of glycosylated moieties. Scatchard analysis revealed striking differences in the apparent binding constants (104–105 M−1) and number of binding sites (106–107sites/erythrocyte) for CRP on patients’ erythrocytes as compared to normal. Western blotting along with immunoprecipitation analysis revealed the presence of distinct molecular determinants on TB and VL erythrocytes specific to disease-associated CRP. Increased fragility, hydrophobicity and decreased rigidity of diseased-erythrocytes upon binding with glycosylated CRP suggested membrane damage. Finally, the erythrocyte-CRP binding was shown to activate the CRP-complement-cascade causing hemolysis, even at physiological concentration of CRP (10 μg/ml). Thus, it may be postulated that CRP have a protective role towards the clearance of damaged-erythrocytes in these two diseases.






Similar content being viewed by others
Abbreviations
- αGalNAc:
-
alpha-N-acetyl Galactosamine
- αGlcNAc:
-
alpha-N-acetyl Glucosamine
- α-L-Fuc:
-
alpha-L-fucose
- ABTS:
-
2, 2′-azino-bis (3-ethylbenzthiazole-6-sulfonic acid)
- ANS:
-
8-anilino-1-napthalenesulfonic acid
- BSA:
-
Bovine Serum Albumin
- CaCl2 :
-
Calcium Chloride
- CHAPS:
-
3-(3-cholamidopropyl) dimethylammonio)-1-propanesulfonic acid
- Con A:
-
Concanavalin A
- CRP:
-
C-reactive protein
- CRPTB and CRPVL :
-
Purified CRP from patients with TB and VL
- CRPSigma :
-
CRP purified from human plasma purchased from Sigma Chemical Company
- DAB:
-
3, 3-diaminobenzidine
- DBA:
-
Dolichos biflorus agglutinin
- DIG:
-
Dioxigenin
- DPH:
-
1,6-diphenyl-1, 3, 5-hexatriene
- DSA:
-
Datura stramonium agglutinin
- E:
-
Erythrocytes
- ETB, EVL and EN :
-
Erythrocytes from TB, VL and normal (N) individuals
- EDTA:
-
Ethylene diamine tetra acetic acid
- ELISA:
-
Enzyme-linked immunosorbent assay
- FACS:
-
Fluorescence activated cell sorter
- FITC:
-
Fluorescein isothiocynate
- GLC:
-
Gas liquid chromatography
- GNA:
-
Galanthus nivalis agglutinin
- GVB:
-
Gelatin-veronal-buffered
- HRP:
-
Horseradish peroxidase
- I:
-
Iodine
- IEF:
-
Isoelectric focussing
- IgG:
-
Immunoglobulin G
- kDa:
-
kilo Dalton
- MAA:
-
Maackia amurensis agglutinin
- MAC:
-
Membrane attack complex
- NaCl:
-
Sodium Chloride
- NHS:
-
Normal human serum
- PAGE:
-
Polyacrylamide gel electrophoresis
- PC:
-
Phosphocholine
- PNA:
-
Peanut agglutinin
- SPR:
-
Surface plasmon resonance
- SD:
-
Standard Deviation
- SDS:
-
Sodium Dodecyl sulphate
- SNA:
-
Sambucus nigra agglutinin
- TB:
-
Tuberculosis
- TCA:
-
Trichloro acetic acid
- UEA:
-
Ulex europaeus agglutinin
- VL:
-
Visceral leishmaniasis
- WGA:
-
Wheat germ agglutinin
References
Agrawal, A.: CRP after 2004. Mol. Immunol. 42, 927–930 (2005). doi:10.1016/j.molimm.2004.09.028
Marnell, L., Mold, C., Du Clos, T.W.: C-reactive protein: ligands, receptors and role in inflammation. Clin. Immunol. 117, 104–111 (2005). doi:10.1016/j.clim.2005.08.004
Volanakis, J.E.: Human C-reactive protein: expression, structure, and function. Mol. Immunol. 38, 189–197 (2001). doi:10.1016/S0161-5890(01)00042-6
Gould, J.M., Weiser, J.N.: Expression of C-reactive protein in the human respiratory tract. Infect. Immun. 69, 1747–1754 (2001). doi:10.1128/IAI.69.3.1747-1754.2001
Jabs, W.J., Logering, B.A., Gerke, P., Kreft, B., Wolber, E.M., Klinger, M.H., Fricke, L., Steinhoff, J.: The kidney as a second site of human C-reactive protein formation in vivo. Eur. J. Immunol. 33, 152–161 (2003). doi:10.1002/immu.200390018
Volanakis, J.E., Kaplan, M.H.: Specificity of C-reactive protein for choline phosphate residues of pneumococcal C-polysaccharide. Proc. Soc. Exp. Biol. Med. 136, 612–614 (1971)
Das, T., Mandal, C., Mandal, C.: Protein A-a new ligand for human C-reactive protein. FEBS Lett. 576, 107–113 (2004). doi:10.1016/j.febslet.2004.08.072
Das, T., Sen, A., Kempf, T., Pramanik, S.R., Mandal, C., Mandal, C.: Induction of glycosylation in human C-reactive protein under different pathological conditions. Biochem. J. 373, 345–355 (2003). doi:10.1042/BJ20021701
Das, T., Mandal, C., Mandal, C.: Variations in binding characteristics of glycosylated human C-reactive proteins in different pathological conditions. Glycoconj. J. 20, 537–543 (2004). doi:10.1023/B:GLYC.0000043290.90182.e6
Du Clos, T.W., Marnell, L., Zlock, L.R., Burlingame, R.W.: Analysis of the binding of C-reactive protein to chromatin subunits. J. Immunol. 146, 1220–1225 (1991)
Du Clos, T.W., Zlock, L.T., Marnell, L.: Definition of a C-reactive protein binding determinant on histones. J. Biol. Chem. 266, 2167–2171 (1991)
James, K., Hansen, B., Gewurz, H.: Binding of C-reactive protein to human lymphocytes. II. Interaction with a subset of cells bearing the Fc receptor. J. Immunol. 127, 2545–2550 (1981)
Gewurz, H., Mold, C., Siegel, J., Fiedel, B.: C-reactive protein and the acute phase response. Adv. Intern. Med. 27, 345–372 (1982)
Kaplan, M.H., Volanakis, J.E.: Interaction of C-reactive protein complexes with the complement system. I. Consumption of human complement associated with the reaction of C-reactive protein with pneumococcal C-polysaccharide and with the choline phosphatides, lecithin and sphingomyelin. J. Immunol. 112, 2135–2147 (1974)
McGrath, F.D., Brouwer, M.C., Arlaud, G.J., Daha, M.R., Hack, C.E., Roos, A.: Evidence that complement protein C1q interacts with C-reactive protein through its globular head region. J. Immunol. 176, 2950–2957 (2006)
Kottgen, E., Hell, B., Kage, A., Tauber, R.: Lectin specificity and binding characteristics of human C-reactive protein. J. Immunol. 149, 445–453 (1992)
Arese, P., Turrini, F., Schwarzer, E.: Band 3/complement-mediated recognition and removal of normally senescent and pathological human erythrocytes. Cell. Physiol. Biochem. 16, 133–146 (2005). doi:10.1159/000089839
Lutz, H.U.: Innate immune and non-immune mediators of erythrocyte clearance. Cell Mol Biol Noisy-le-grand 50, 107–116 (2004)
Volanakis, J.E., Clements, W.L., Schrohenlohr, R.E.: C-reactive protein: purification by affinity chromatography and physiochemical characterization. J. Immunol. Methods 23, 285–295 (1978). doi:10.1016/0022-1759(78)90203-X
Christner, R.B., Mortensen, R.F.: Specificity of the binding interaction between human serum amyloid P-component and immobilized human C-reactive protein. J. Biol. Chem. 269, 9760–9766 (1994)
Lowry, O.H., Rosebrough, N.J., Farr, A.L., Randall, R.J.: Protein measurement with the Folin phenol reagent. J. Biol. Chem. 193, 265–275 (1951)
Thompson, D., Pepys, M.B., Wood, S.P.: The physiological structure of human C-reactive protein and its complex with phosphocholine. Structure 7, 169–177 (1999). doi:10.1016/S0969-2126(99)80023-9
Laemmli, U.K.: Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227, 680–685 (1970). doi:10.1038/227680a0
Towbin, H., Staehelin, T., Gordon, J.: Electrophoretic transfer of proteins from polyacrylamide gels to nitrocellulose sheets: procedure and some applications. Proc. Natl. Acad. Sci. USA 76, 4350–4354 (1979). doi:10.1073/pnas.76.9.4350
Hunter, W.M.: Handbook of experimental medicine, Weir, D.M. edition, Blackwell Scientific Publication, Oxford, 14.1–14.3 (1978)
Sloneker, J.H.: Gas-liquid chromatography of alditol acetates, methods in carbohydrate chemistry. In: Whistler, R.L., BeMiller, J.N. (eds.) Academic Press Inc., VI, 20–24 (1972)
Lönngren, J., Svensson, S.: Mass spectrometry in structural analysis of natural carbohydrates. Adv. Carbohydr. Chem. Biochem. 29, 41–106 (1974). doi:10.1016/S0065-2318(08)60248-6
Pal, S., Chatterjee, M., Bhattacharya, D.K., Bandhyopadhyay, S., Mandal, C.: Identification and purification of cytolytic antibodies directed against O-acetylated sialic acid in childhood acute lymphoblastic leukemia. Glycobiology 10, 539–549 (2000). doi:10.1093/glycob/10.6.539
O’ Farrell, P.Z., Goodman, H.M., O’ Farrell, P.Z.: High resolution two-dimensional electrophoresis of basic as well as acidic proteins. Cell 12, 1133–1141 (1977). doi:10.1016/0092-8674(77)90176-3
Coligan E.J., Kruisbeek M.A., Margulies H.D., Shevach M.E., Strober W.: Current protocols in immunology (Wiley Interscience, National Institute of Health, 1993), 32–34 (1993)
Scatchard, G.: The attractions of proteins for small molecules and ions. Ann. N. Y. Acad. Sci. 51, 660–672 (1949). doi:10.1111/j.1749-6632.1949.tb27297.x
Shukla, A.K., Schauer, R.: Fluorimetric determination of unsubstituted and 9(8)-O-acetylated sialic acids in erythrocyte membranes. Hoppe Seylers Z. Physiol. Chem. 363, 255–262 (1982)
Sharma, V., Chatterjee, M., Mandal, C., Sen, S., Basu, D.: Rapid diagnosis of Indian visceral leishmaniasis using achatinin H, a 9-O-acetylated sialic acid binding lectin. Am. J. Trop. Med. Hyg. 58, 551–554 (1998)
Ghosh, S., Bandyopadhyay, S., Bhattacharya, D.K., Mandal, C.: Altered erythrocyte membrane characteristics during anemia in childhood acute lymphoblastic leukemia. Ann. Hematol. 84, 76–84 (2005). doi:10.1007/s00277-004-0933-0
Osmand, A.P., Mortensen, R.F., Siegel, J., Gewurz, H.: Interactions of C-reactive protein with the complement system. III. Complement-dependent passive hemolysis initiated by CRP. J. Exp. Med. 142, 1065–1077 (1975). doi:10.1084/jem.142.5.1065
David, M., Pepys, M.B., Hawkins, P.N.: Metabolic and scintigraphic studies of radioiodinated human C-reactive protein in health and diseases. J. Clin. Invest. 91, 1351–1357 (1993). doi:10.1172/JCI116336
Volanakis, J.E., Kaplan, M.H.: Interaction of C-reactive protein complexes with the complement system. II. Consumption of guinea pig complement by CRP complexes: requirement for human C1q. J. Immunol. 113, 9–17 (1974)
Gabay, C., Kushner, I.: Acute-phase proteins and other systemic responses to inflammation. N. Engl. J. Med. 340, 448–454 (1999). doi:10.1056/NEJM199902113400607
Kushner, I., Rakita, L., Kaplan, M.H.: Studies of acute-phase protein. II. Localization of Cx-reactive protein in heart in induced myocardial infarction in rabbits. J. Clin. Invest. 42, 286–292 (1963). doi:10.1172/JCI104715
Li, P., Mold, C., Du Clos, T.W.: Sublytic complement attack exposes C-reactive protein binding sites on cell membranes. J. Immunol. 152, 2995–3005 (1994)
Narkates, A.J., Volanakis, J.E.: C-reactive protein binding specificities: artificial and natural phospholipid bilayers. Ann. N. Y. Acad. Sci. 389, 172–182 (1982). doi:10.1111/j.1749-6632.1982.tb22135.x
Pepys, M.B., Baltz, M.L.: Acute phase proteins with special reference to C-reactive protein and related proteins (pentaxins) and serum amyloid A protein. Adv. Immunol. 34, 141–212 (1983). doi:10.1016/S0065-2776(08)60379-X
Pepys, M.B.: C-reactive protein fifty years on. Lancet 1, 653–657 (1981). doi:10.1016/S0140-6736(81)91565-8
Ansar, W., Bandyopadhyay, S.M., Chowdhury, S., Habib, S.H., Mandal, C.: Role of C-reactive protein in complement-mediated hemolysis in Malaria. Glycoconj. J. 23, 233–240 (2006). doi:10.1007/s10719-006-7928-0
Ansar, W., Mandal, C., Habib, S.K.H., Roy, S., Mandal, C.: Unraveling the C-reactive protein complement-cascade in destruction of red blood cells: potential pathological implications in Plasmodium falciparum malaria. Cell. Physiol. Biochem. 23, 175–190 (2009). doi:10.1159/000204106
Paul, I., Mandal, C., Allen, A.K., Mandal, C.: Molecular variants of C-reactive proteins from the major carp Catla catla in fresh and polluted aquatic environments. Glycoconj. J. 18, 547–556 (2001). doi:10.1023/A:1019696430477
Mandal, C., Chatterjee, M., Sinha, D.: Investigation of 9-O-acetylated sialoglycoconjugates in childhood acute lymphoblastic leukaemia. Br. J. Haematol. 110, 801–812 (2000). doi:10.1046/j.1365-2141.2000.02105.x
Sinha, D., Chatterjee, M., Mandal, C.: O-acetylation of sialic acids-their detection, biological significance and alteration in diseases. Trends Glycosci. Glycotechnol. 12, 17–33 (2000)
Sinha, D., Mandal, C., Bhattacharya, D.K.: Identification of 9-O acetyl sialoglycoconjugates (9-OAcSGs) as biomarkers in childhood acute lymphoblastic leukemia using a lectin, AchatininH, as a probe. Leukemia 13, 119–125 (1999). doi:10.1038/sj.leu.2401312
Mandal, C., Biswas, M., Nagpurkar, A., Mookerjea, S.: Isolation of a phosphoryl choline-binding protein from the hemolymph of the snail, Achatina fulica. Dev. Comp. Immunol. 15, 227–239 (1991). doi:10.1016/0145-305X(91)90016-R
Lasson, A., Goransson, J.: No microheterogenous changes of plasma C-reactive protein found in man during various diseases. Scand. J. Clin. Lab. Invest. 59, 293–304 (1999). doi:10.1080/00365519950185661
Mandal, C., Srinivasan, G.V., Chowdhury, S., Chandra, S., Mandal, C., Schauer, R., Mandal, C.: High level of sialate-O-acetyltransferase activity in lymphoblasts of childhood acute lymphoblastic leukaemia (ALL): enzyme characterization and correlation with disease status. Glycoconj. J. 26, 57–73 (2009). doi:10.1007/s10719-008-9163-3
Sinha, S., Mandal, C.: Microheterogeneity of C-reactive protein in the sera of fish Labeo rohita induced by metal pollutants. Biochem. Biophys. Res. Commun. 226, 681–687 (1996). doi:10.1006/bbrc.1996.1414
Sinha, S., Mandal, C.N., Allen, A.K., Mandal, C.: Acute phase response of C-reactive protein of Labeo rohita to aquatic pollutants is accompanied by the appearance of distinct molecular forms. Arch. Biochem. Biophys. 369, 139–150 (2001). doi:10.1006/abbi.2001.2592
Paul, I., Mandal, C., Mandal, C.: Effect of environmental pollutants on the C-reactive protein of a freshwater major carp, Catla catla. Dev. Comp. Immunol. 22, 519–532 (1998). doi:10.1016/S0145-305X(98)00031-7
Chatterjee, M., Sharma, V., Sundar, S., Sen, S., Mandal, C.: Identification of antibodies directed against O-acetylated sialic acids in visceral leishmaniasis: its diagnostic and prognostic role. Glycoconj. J. 15, 1141–1147 (1998). doi:10.1023/A:1006963806318
Baltz, M.L., De Beer, F.C., Feinstein, A., Munn, E.A., Fletcher, T.C., Taylor, J., Bruton, C., Clamp, J.R., Davies, A.J.S., Pepys, M.B.: Phylogenetic aspects of C-reactive protein and related protein. Ann. N. Y. Acad. Sci. 389, 49–75 (1982). doi:10.1111/j.1749-6632.1982.tb22125.x
Mold, C., Gurulé, C., Otero, D., Du Clos, T.W.: Complement-dependent binding of C-reactive protein complexes to human erythrocyte CR1. Clin. Immunol. Immunopathol. 8, 153–160 (1996). doi:10.1006/clin.1996.0171
Davies, K.A., Hird, V., Stewart, S., Sivolapenko, G.B., Jose, P., Epenetos, A.A., Walport, M.J.: A study of in vivo immune complex formation and clearance in man. J. Immunol. 144, 4613–4620 (1990)
Kundu, M., Basu, J., Chakrabarti, P.: Chronic myelogenous leukemia: alterations in red cell membrane band 3 and increased IgG binding. Indian J. Biochem. Biophys. 27, 456–459 (1990)
Roy, S.S., Sen, G., Biswas, T.: Role of sulfhydryl groups in band 3 in the inhibition of phosphate transport across erythrocyte membrane in visceral leishmaniasis. Arch. Biochem. Biophys. 436, 121–127 (2005). doi:10.1016/j.abb.2005.01.015
Mukherjee, K., Chowdhury, S., Mondal, S., Mandal, C., Chandra, S., Bhadra, R.K., Mandal, C.: 9-O-acetylated GD3 triggers programmed cell death in mature erythrocytes. Biochem. Biophys. Res. Commun. 362, 651–657 (2007). doi:10.1016/j.bbrc.2007.08.048
Gupta, R.K., Pande, A.H., Gulla, K.C., Gabius, H.J., Hajela, K.: Carbohydrate-induced modulation of cell membrane. VIII. Agglutination with mammalian lectin galectin-1 increases osmofragility and membrane fluidity of trypsinized erythrocytes. FEBS Lett. 580, 1691–1695 (2006). doi:10.1016/j.febslet.2006.02.006
Pande, A.H., Sumati, N., Hajela, N., Hajela, K.: Carbohydrate induced modulation of cell membrane VII. Binding of exogenous lectin increases osmofragility of erythrocytes. FEBS Lett. 427, 21–24 (1998). doi:10.1016/S0014-5793(98)00384-6
Hajela, K., Pande, A.H., Sumati, N.: Carbohydrate induced modulation of cell membrane. VI. Binding of exogenous lectin induces susceptibility of erythrocytes to free radical damage: a spin label study. FEBS Lett. 406, 255–258 (1997). doi:10.1016/S0014-5793(97)00272-X
Volanakis, J.E., Wirtz, K.W.: Interaction of C-reactive protein with artificial phosphatidylcholine bilayers. Nature 281, 155–157 (1979). doi:10.1038/281155a0
Deuticke, B., Lutkemeier, P., Sistemich, M.: Ion selectivity of aqueous leaks induced in the erythrocyte membrane by cross linking of membrane proteins. Biochim. Biophys. Acta 775, 150–160 (1984). doi:10.1016/0005-2736(84)90165-2
Agrawal, A., Volanakis, J.E.: Probing the C1q-binding site on human C-reactive protein by site-directed mutagenesis. J. Immunol. 152, 5404–5410 (1994)
Agrawal, A., Shrive, A.K., Greenhough, T.J., Volanakis, J.E.: Topology and structure of the C1q-binding site on C-reactive protein. J. Immunol. 166, 3998–4004 (2001)
Chava, A.K., Chatterjee, M., Sharma, V., Sundar, S., Mandal, C.: Variable degree of alternative complement pathway-mediated hemolysis in Indian visceral leishmaniasis induced by differential expression of 9-O-acetylated sialoglycans. J. Infect. Dis. 189, 1257–1264 (2004). doi:10.1086/382752
Bandyopadhyay, S., Chatterjee, M., Das, T., Bandyopadhyay, S., Sundar, S., Mandal, C.: Antibodies directed against O-acetylated sialoglycoconjugates accelerate complement activation in Leishmania donovani promastigotes. J. Infect. Dis. 190, 2010–2019 (2004). doi:10.1086/425519
Sharma, V., Chatterjee, M., Sen, G., Kumar, C.A., Mandal, C.: Role of linkage specific 9-O-acetylated sialoglycoconjugates in activation of the alternate complement pathway in mammalian erythrocytes. Glycoconj. J. 17, 887–893 (2000). doi:10.1023/A:1010925414222
Berman, S., Gewurz, H., Mold, C.: Binding of C-reactive protein to nucleated cells leads to complement activation without cytolysis. J. Immunol. 136, 1354–1359 (1986)
Klegeris, A., Singh, E.A., McGeer, P.L.: Effects of C-reactive protein and pentosan polysulphate on human complement activation. Immunology 106, 381–388 (2002). doi:10.1046/j.1365-2567.2002.01425.x
Acknowledgement
This work was supported by the Council of Scientific and Industrial Research (CSIR), I.I.C.B, Department of Science and Technology and the Indian Council of Medical Research, Govt. of India. W.A and S.M receive their Senior Research fellowships from CSIR; S.B from University Grants Commission. We are thankful to Sajal Samanta and Biswajit Khatua; Suchandra Chowdhury, Kaushik Bhattacharya for FACS; Asish Mallick for their help; Samir Roy for SPR and Sandip Chakrabarty for MALDI-TOF analysis.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ansar, W., Mukhopadhyay, S., Habib, S.H. et al. Disease-associated glycosylated molecular variants of human C-reactive protein activate complement-mediated hemolysis of erythrocytes in tuberculosis and Indian visceral leishmaniasis. Glycoconj J 26, 1151–1169 (2009). https://doi.org/10.1007/s10719-009-9236-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10719-009-9236-y