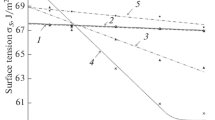
The granulometric compositions of native and acid-activated rocks containing aluminosilicates are compared. It is shown that in water rocks gradually dissolve and the particle size decreases simultaneously, which is associated with the disaggregation of large particles, diffusion of water into the interparticle space, and hydrolysis of aluminosilicates. The effect of acid treatment on the adsorption-structural characteristics of diatomite was investigated. It is shown that analysis of the particle-size distribution can be used for rapid assessment of the performance of acid-activated sedimentary rocks.




Similar content being viewed by others
References
C. N. Rhodes and D. R. Brown, “Structural characterization and optimization of acid-treated montmorillonite and high-porosity silica supports for ZnCl2 alkylation catalysts,” J. Chem. Soc., Faraday Trans., 88(15), 2269 – 2274 (1992).
L. I. Bel’chinskaya, O. Yu. Strelnikova, N. A. Khodosova, and F. Ressner, “Adsorption-structural, ion-exchange, and catalytic characteristics of the native and modified sorbent of the Sokirnitskii deposit,” Khim., Fiz. ta Tekhnol. Poverkhni, No. 4, 420 – 426 (2013).
E. Srasra, F. Bergaya, H. Van Damme, and N. K. Ariguib, “Surface properties of an activated bentonite colorization of rape-seed oils,” Appl. Clay Sci., 4(5 – 6), 411 – 421 (1989).
G. E. Christidis, P. W. Scott, and A. C. Dunham, “Acid activation and bleaching capacity of bentonites from the islands of Milos and Chios, Aegean, Greece,” Appl. Clay Sci., 12(4), 329 – 347 (1997).
G. E. Christidis and S. Kosiari, “Decolorization of vegetable oils: a study of the mechanism of adsorption of β-carotene by an acid-activated bentonite from Cyprus,” Clays and Clay Minerals, 51(3), 327 – 333 (2003).
F. K. Hymore, “Effects of some additives on the performance of acid-activated clays in the bleaching of palm oil,” Appl. Clay Sci., 10(5), 379 – 385 (1996).
I. Mrad, A. Ghorbel, D. Tichit, and J. F. Lambert, “Optimization of the preparation of an Al-pillared clay: thermal stability and surface acidity,” Appl. Clay Sci., 12(4), 349 – 364 (1997).
I. Benito, C. Blanco, M. Martínez, et al., “Thermogravimetric analysis for acidity determination in pillared clays,” J. Thermal Anal. Calorim., 55(2), 461 – 466 (1999).
A. V. Bondarenko, Yu. Ya. Filonenko, L. I. Belchinskaya, et al., “Regulation of the composition of the surface layer of montmorillonite by acid activation,” Zh. Fiz. Khim., 79(7), 1280 – 1284 (2005).
L. Rožić, T. Novaković, and S. Petrović, “Modeling and optimization process parameters of acid activation of bentonite by response surface methodology,” Appl. Clay Sci., 48(1 – 2), 154 – 158 (2010).
L. I. Belchinskaya, K. A. Kozlov, A. V. Bondarenko, et al., “Investigation of structural and adsorption characteristics on activation and modification of native silicates,” Sorbts. Khromatograf. Prots., 7(4), 571 – 576 (2007).
P. Komadel, D. Schmidt, J. Madejová, and B. Èíèel, “Alteration of smectites by treatments with hydrochloric acid and sodium carbonate solutions,” Appl. Clay Sci., 5(2), 113 – 122 (1990).
L. I. Belchinskaya, N. A. Khodosova, O. Yu. Strelnikova, and G. A. Petukhova, “Regulation of sorption processes on natural nanoporous aluminosilicates. 1. Acid and base modification,” Fizikokhimiya Poverkh. Zashch. Mater., 51(5), 487 – 494 I2015O.
O. M. Mdivnishvili, Crystalline Basis for Regulating the Properties of Native Sorbents [in Russian], Metsniereba, Tbilisi (1983).
N. F. Chelishchev, Ion-Exchange Properties of Minerals [in Russian], Nauka, Moscow (1973).
J. B. Jones and E. R. Segnit, “The nature of opal I. Nomenclature and constituent phases,” J. Geol. Soc. Australia, 18, 57 – 68 (1971).
V. V. Nasedkin, N. M. Boeva, I. A. Garbuzova, et al., “Crystal chemical studies of several samples of palygorskite of various genesis,” Kristallografiya, 54(5), 930 – 947 (2009).
G. I. Gorbunov and A. D. Zhukov, Scientific Foundations of the Formation of the Structure and Properties of Building Materials [in Russian], Moscow State University of Civil Engineering, IPR Media – EBS ASV, Moscow (2016).
L. A. Novikova, L. I. Belchinskaya, and F. Ressner, “Influence of acid treatment on the surface properties of clay minerals,” Sorbts. Khromatograf. Prots., 5(6), 798 – 805 (2005).
E. B. Inzhinova, Catalytic Cracking of Paraffin on Modified Superacid Native Zeolite [in Russian], KazNRTU im. K. I. Satpaeva, Almaty (2019).
N. N. Matyushenko, “Adsorption refining of plant oils,” Nauch. Tr. Vseros. Nauch.-Issled. Inst. Ovtsevodstva i Kozovodstva [Research Institute of Sheep and Goat Breeding], 1(9), 123 – 126 (2016).
V. V. Ponomarev, Technology of Adsorbents for Purification of Plant Oils Based on Diatomite and Bentonite of the Rostov Oblast’, Author’s Abstract of Candidate’s Thesis [in Russian], Novocherkassk (2010).
E. M. Kotova, Development of Effective Methods of Adsorption Refining of Plant Oils, Author’s Abstract of Candidate’s Thesis [in Russian], Moscow (2008).
Yu. A. Ubas’kina, E. G. Fetyukhina, and Yu. A. Korosteleva, “Study of the adsorption capacity of diatomite,” Vest. Belgorod. Gos. Tekhnol. Univ. im. V. G. Shukhova, No. 5, 140 – 143 (2016).
S. V. Pugacheva, Adsorbents Based on Diatomite and Bentonite of the Rostov Obllast’ for Regeneration of Petroleum Oils, Author’s Abstract of Candidate’s Thesis [in Russian], Novocherkassk (2002).
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Steklo i Keramika, No. 10, pp. 42 – 50, October, 2021.
Rights and permissions
About this article
Cite this article
Ubas’kina, Y.A., Alekhina, M.B. Application of Granulometry for Rapid Assessment of the Performance Properties of Acid-Activated Sedimentary Rocks. Glass Ceram 78, 416–422 (2022). https://doi.org/10.1007/s10717-022-00423-2
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10717-022-00423-2