Abstract
Next-generation sequencing has allowed us to explore new methods, where comparative and population genomics can be used simultaneously. Keeping this in mind, we surveyed and analyzed the frequency and distribution of microsatellites in the Indian gharial (Gavialis gangeticus) and compared it with American alligator (Alligator mississippiensis) and saltwater crocodile (Crocodylus porosus) to enrich them with genomic resources. The Indian gharial has a low frequency, relative abundance (RA), and relative density (RD) of microsatellites as compared to other crocodilians. RA and RD were positively correlated with the GC content of genomic and transcriptomic sequences. The genomic sequences were dominated by dinucleotide repeats, whereas the transcriptomic sequences had an excess of trinucleotide repeats. Motif conservation studies among the three crocodilians revealed conservation of 69.2% of motifs. Species-specific unique motifs identified in this study could be used as molecular probes for species identification. A total of 67,311 primers were designed in all three species to enrich the crocodilians with genomic resources. The genomic resources developed in this study could accelerate diversity analysis within its individuals to design a proper mating plan to reduce inbreeding stress and further improve the species.

Similar content being viewed by others
Data availability
List of unique motifs identified in the present study (Table S1, Supporting Information).
List of microsatellite primers designed for all the three crocodilians (Table S2, Supporting Information).
List of loci amplified using virtual PCR (Table S3, Supporting Information).
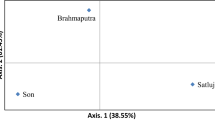
Figure showing PCA plot (Fig. S1, Supporting Information).
References
Bachtrog D, Agis M et al (2000) Microsatellite variability differs between dinucleotide repeat motifs-evidence from Drosophila melanogaster. Mol Biol Evol 17(9):1277–1285. https://doi.org/10.1093/oxfordjournals.molbev.a026411
Choudhary S, Choudhury BC et al (2017) Differential response to disturbance factors for the population of sympatric crocodilians (Gavialis gangeticus and Crocodylus palustris) in Katarniaghat Wildlife Sanctuary, India. Aquat Conserv Mar Freshw Ecosyst 27(5):946–952. https://doi.org/10.1002/aqc.2815
da Maia LC, Palmieri DA et al (2008) SSR Locator: tool for simple sequence repeat discovery integrated with primer design and PCR simulation. Int J Plant Genomics 2008:412696. https://doi.org/10.1155/2008/412696
Datta S, Mahfooz S et al (2010) Cross-genera amplification of informative microsatellite markers from common bean and lentil for the assessment of genetic diversity in pigeonpea. Physiol Mol Biol Plants 16(2):123–134. https://doi.org/10.1007/s12298-010-0014-x
Djian P (1998) Evolution of simple repeats in DNA and their relation to human disease. Cell 94(2):155–160. https://doi.org/10.1016/s0092-8674(00)81415-4
Gans C (1989) Crocodilians in perspective! Am Zool 29(3):1051–1054
Gayral P, Melo-Ferreira J et al (2013) Reference-free population genomics from next-generation transcriptome data and the vertebrate–invertebrate gap. PLoS Genet 9(4):e1003457. https://doi.org/10.1371/journal.pgen.1003457
Ghosh A, Johnson MG et al (2019) A high-quality reference genome assembly of the saltwater crocodile, Crocodylus porosus, reveals patterns of selection in Crocodylidae. Genome Biol Evol 12(1):3635–3646. https://doi.org/10.1093/gbe/evz269
Glenn TC, Dessauer HC et al (1998) Characterization of microsatellite DNA loci in American alligators. Copeia 1998(3):591–601. https://doi.org/10.2307/1447789
Green RE, Braun EL et al (2014) Three crocodilian genomes reveal ancestral patterns of evolution among archosaurs. Science 346(6215):1254449. https://doi.org/10.1126/science.1254449
Isberg SR, Chen Y et al (2004) Analysis of microsatellites and parentage testing in saltwater crocodiles. J Hered 95(5):445–449. https://doi.org/10.1093/jhered/esh067
Janke A, Gullberg A et al (2005) Mitogenomic analyses place the gharial (Gavialis gangeticus) on the crocodile tree and provide pre-K/T divergence times for most crocodilians. J Mol Evol 61(5):620–626. https://doi.org/10.1007/s00239-004-0336-9
Jarne P, David P et al (1998) Microsatellites, transposable elements and the X chromosome. Mol Biol Evol 15(1):28–34. https://doi.org/10.1093/oxfordjournals.molbev.a025844
Jogayya KN, Meganathan PR et al (2013) Novel microsatellite DNA markers for Indian Gharial (Gavialis gangeticus). Conserv Genet Resour 5(3):787–790. https://doi.org/10.1007/s12686-013-9908-6
Katdare S, Srivathsa A et al (2011) Gharial (Gavialis gangeticus) populations and human influences on habitat on the River Chambal, India. Aquat Conserv Mar Freshw Ecosyst 21(4):364–371. https://doi.org/10.1002/aqc.1195
Lang J, Chowfin S et al (2019) Gavialis gangeticus. The IUCN Red List of Threatened Species 2019
Lopez-Flores I, Garrido-Ramos MA (2012) The repetitive DNA content of eukaryotic genomes. Genome Dyn 7:1–28. https://doi.org/10.1159/000337118
Mahfooz S, Srivastava A et al (2015) A comparative analysis of distribution and conservation of microsatellites in the transcripts of sequenced Fusarium species and development of genic-SSR markers for polymorphism analysis. FEMS Microbiol Lett. https://doi.org/10.1093/femsle/fnv131
Mahfooz S, Singh SP et al (2016) A comprehensive characterization of simple sequence repeats in the sequenced Trichoderma genomes provides valuable resources for marker development. Front Microbiol 7:575. https://doi.org/10.3389/fmicb.2016.00575
Mahfooz S, Singh SP et al (2017) A comparison of microsatellites in phyto-pathogenic Aspergillus species in order to develop markers for the assessment of genetic diversity among its isolates. Front Microbiol 8:1774
McMahon BJ, Teeling EC et al (2014) How and why should we implement genomics into conservation? Evol Appl 7(9):999–1007. https://doi.org/10.1111/eva.12193
Metzgar D, Bytof J et al (2000) Selection against frameshift mutations limits microsatellite expansion in coding DNA. Genome Res 10(1):72–80
Miles LG, Isberg SR et al (2009) 253 Novel polymorphic microsatellites for the saltwater crocodile (Crocodylus porosus). Conserv Genet 10(4):963–980. https://doi.org/10.1007/s10592-008-9600-7
Mitra I, Roy S et al (2018) Application of molecular markers in wildlife DNA forensic investigations. J Forensic Sci Med 4(3):156–160
Mortlock DP, Sateesh P et al (2000) Evolution of N-terminal sequences of the vertebrate HOXA13 protein. Mamm Genome 11(2):151–158. https://doi.org/10.1007/s003350010029
Sharma SP, Ghazi MG et al (2021) Microsatellite analysis reveals low genetic diversity in managed populations of the critically endangered gharial (Gavialis gangeticus) in India. Sci Rep 11(1):5627. https://doi.org/10.1038/s41598-021-85201-w
St John JA, Braun EL et al (2012) Sequencing three crocodilian genomes to illuminate the evolution of archosaurs and amniotes. Genome Biol 13(1):415. https://doi.org/10.1186/gb-2012-13-1-415
Tahoor A, Khan JA et al (2019) A comparative survey of microsatellites among wild and domestic cat provides valuable resources for marker development. Mol Biol Rep 46(3):3025–3033. https://doi.org/10.1007/s11033-019-04739-1
Tian X, Strassmann JE et al (2011) Genome nucleotide composition shapes variation in simple sequence repeats. Mol Biol Evol 28(2):899–909. https://doi.org/10.1093/molbev/msq266
Toth G, Gaspari Z et al (2000) Microsatellites in different eukaryotic genomes: survey and analysis. Genome Res 10(7):967–981
Wang X, Wang L (2016) GMATA: an integrated software package for genome-scale SSR mining, marker development and viewing. Front Plant Sci 7:1350. https://doi.org/10.3389/fpls.2016.01350
Acknowledgements
Research in YA Lab was supported by the Extramural Grant from the Indian Council of Medical Research (MoFHW, Government of India) and the Department of Biotechnology, (MoS&T, Government of India).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
None declared.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic Supplementary Material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Mahfooz, S., Singh, P. & Akhter, Y. A comparative study of microsatellites among crocodiles and development of genomic resources for the critically endangered Indian gharial. Genetica 150, 67–75 (2022). https://doi.org/10.1007/s10709-021-00148-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10709-021-00148-0