Abstract
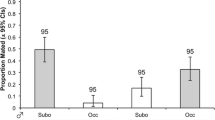
The origin of sexual isolation is the central event in the evolution of biological species and plays a key role in maintaining biological diversity. Three mass culture stocks of D. ananassae originating from different geographic localities showing no isolation with each other were subjected to different degrees of bottlenecks i.e. one pair, five pairs and ten pairs. These drift lines were passed through flush-crash cycle at every generation with same initial number of founders, and maintained for twenty-seven generations and then the pattern of matings was tested among these nine drift lines involving 36 crosses in total. In 23 of 36 crosses, the difference between homogamic and heterogamic matings was significant and isolation indices were significantly more than zero in one direction only providing evidence for asymmetrical sexual isolation. Further, when Bonferroni test for pair-wise analysis was employed, significant differences between homogamic and heterogamic matings were found in 25 crosses. These findings provide evidence for origin of sexual isolation by founder effects in D. ananassae.

Similar content being viewed by others
References
Barker JSF, Frydenberg J, Gonzalez J, Davies HI, Ruiz A, Sorensen JG et al (2009) Bottlenecks, population differentiation and apparent selection at microsatellite loci in Australian Drosophila buzzatii. Heredity 102:389–401
Boughman JW (2002) How sensory drive can promote speciation. Tr Ecol Evol 17:571–577
Butlin RK (1989) Reinforcement: an idea evolving. Tr Ecol Evol 10:432–434
Butlin RK (2010) Population genomics and speciation. Genetica 138:409–418
Carson HL (1975) Genetics of speciation. Am Nat 109:83–92
Castrezana SJ, Markow TA (2008) Sexual isolation and mating propensity among allopatric Drosophila mettleri populations. Behav Genet 38:437–445
Coyne JA, Orr HA (1997) ‘Patterns of speciation in Drosophila’ revisited. Evolution 5:295–303
Coyne JA, Orr HA (2004) Speciation. Sinauer and Associates, Sunderland
Dobzhansky T (1937) Genetics and the origin of species, 1st edn. Columbia University Press, New York
Dobzhansky T (1940) Speciation as a stage in evolutionary divergence. Am Nat 74:312–321
Dodd DMB, Powell JR (1985) Founder-flush speciation: an update of experimental results with Drosophila. Evolution 39:1388–1392
Ehrman L (1965) Direct observations of sexual isolation between allopatric and sympatric strains of different Drosophila paulistorum races. Evolution 19:459–464
Ehrman L, Parsons PA (1980) Sexual isolation among widely distributed populations of Drosophila immigrans. Behav Genet 10:401–407
Elens AA, Wattiaux JM (1964) Direct observation of sexual isolation. Dros Inf Serv 39:118–119
Freire-Maia N (1961) Peculiar gene arrangements in Brazilian natural populations of Drosophila ananassae. Evolution 15:486–495
Galiana A, Moya A, Ayala FJ (1993) Founder-flush speciation in Drosophila pseudoobscura: a large scale experiment. Evolution 47:432–444
Gerhardt HC, Huber F (2002) Acoustic communications in insects and Anurans. University of Chicago Press, Chicago
Haerty W, Lesbats M, Capy P (2005) Pre-reproductive isolation as a consequence of allopatric differentiation between populations of Drosophila melanogaster. Mol Ecol 14:3801–3807
Henderson NR, Lambert DM (1982) No significant deviation from random mating of worldwide populations of Drosophila melanogaster. Nature 300:437–440
Hollocher H, Ting CT, Wu ML, Wu CI (1997) Incipient speciation by sexual isolation in Drosophila melanogaster: the genetics of the Zimbabwe race. Genetics 147:1121–1191
Howard DJ, Berlocher SH (1998) Endless forms: species and speciation. Oxford University Press, New York
Kaneshiro KY (1976) Ethological isolation and phylogeny in the planitibia subgroup of Hawaiian Drosophila. Evolution 30:740–745
Kirkpatrick M (1982) Sexual selection and the evolution of female choice. Evolution 36:1–12
Koepfer HR (1991) Asymmetrical mating patterns between geographic strains of Drosophila mercatorum: a test of the Kaneshiro hypothesis. Evolution 45:455–458
Kopp A, Frank AK (2005) Speciation in progress? A continuum of reproductive isolation in Drosophila bipectinata. Genetica 125:55–68
Markow TA (1981) Mating preferences are not predictive of the direction of evolution in Drosophila. Science 213:1405–1407
Markow TA, Smith LD (1979) Genetics of phototactic behavior in Drosophila ananassae, a member of Drosophila melanogaster species group. Behav Genet 9:61–68
Matute DR (2010) Reinforcement of gametic isolation in Drosophila. PLoS Biol 8:e1000341
Mayr E (1942) Systematics and the origin of species. Columbia University Press, New York
Mayr E (1963) Animal species and evolution. Belknap Press, Massachusetts
Meffert LM (1995) Bottleneck effects on genetic variance for courtship repertoire. Genetics 129:365–374
Meffert LM, Regan JL, Brown BW (1999) Convergent evolution of the mating behaviour of founder-flush populations of the housefly. J Evol Biol 12:859–868
Moodie GEE (1982) Why asymmetric mating preference may not show the direction of evolution. Evolution 36:1096–1097
Moya A, Galiana A, Ayala FJ (1995) Founder-flush speciation theory: failure of experimental corroboration. Proc Natl Acad Sci USA 92:3983–3986
Muller HJ (1942) Isolating mechanisms, evolution and temperature. Biol Symp 6:71–125
Nanda P, Singh BN (2008) No effect of marking procedures and choice situations on the pattern of matings in Drosophila ananassae. Dros Inf Serv 91:10–13
Nanda P, Singh BN (2011a) Evidence for incipient sexual isolation within Drosophila ananassae. Zool Stud (accepted)
Nanda P, Singh BN (2011b) Effect of chromosome arrangement on mate recognition system leading to behavioral isolation in Drosophila ananassae. Genetica 139:273–279
Noor MAF (1995) Speciation driven by natural selection in Drosophila. Nature 375:674–675
Noor MAF (1997) How often does sympatry affect sexual isolation in Drosophila? Am Nat 149:1156–1163
Noor MAF, Grams KL, Bertucci LA, Reiland J (2001) Chromosomal inversions and the reproductive isolation of species. Proc Natl Acad Sci USA 98:12084–12088
Paterson HE (1993) Evolution and the recognition concept of species: collected writings. John Hopkins University, Baltimore
Powell JR (1978) The founder-flush speciation theory: an experimental approach. Evolution 32:465–474
Rice WR, Hostert EE (1993) Laboratory experiments on speciation: what have we learned in forty years. Evolution 47:1637–1653
Ringo J, Barton K, Dowse H (1986) The effect of genetic drift on mating propensity, courtship behaviour, and postmating fitness in Drosophila simulans. Behaviour 97:226–233
Rundle HD, Mooers AO, Whitlock MC (1998) Single founder-flush events and the evolution of reproductive isolation. Evolution 52:1850–1855
Ryan MJ (1998) Sexual selection, receiver biases, and the evolution of sex differences. Science 281:1999–2003
Sawamura K, Zhi H, Setoguchi K, Yamada H, Miyo T, Matsuda M, Oguma Y (2008) Genetic analysis of female mating recognition between Drosophila ananassae and Drosophila pallidosa: application of interspecific mosaic genome lines. Genetica 133:179–185
Schluter D (2001) Ecology and the origin of species. Tr Ecol Evol 16:372–380
Schug MD, Baines JF, Amada K, Mohanty S, Das A, Grath S et al (2008) Evolution of mating isolation between populations of Drosophila ananassae. Mol Ecol 17:2706–2721
Servedio MR (2001) Beyond reinforcement: the evolution of premating isolation by direct selection on preferences and postmating, prezygotic incompatibility. Evolution 55:1909–1920
Servedio MR, Noor MAF (2003) The role of reinforcement in speciation: theory and data. Ann Rev Ecol Evol Syst 34:339–364
Singh BN (1996) Population and behavior genetics of Drosophila ananassae. Genetica 97:321–332
Singh BN (2010a) The origin of reproductive isolating mechanisms is an important event in the process of speciation: evidences from Drosophila. In: Sharma VP (ed) Nature at work: ongoing saga of evolution. Springer Private Limited, India, pp 159–173
Singh BN (2010b) Drosophila ananassae: a good model species for genetical, behavioural and evolutionary studies. Ind J Exp Biol 48:333–345
Singh BN, Chatterjee S (1985) Symmetrical and asymmetrical sexual isolation among laboratory strains of Drosophila ananassae. Can J Genet Cytol 27:405–409
Singh BN, Chatterjee S (1988) Selection for high and low mating propensity in Drosophila ananassae. Behav Genet 18:357–369
Singh BN, Chatterjee S (1989) Rare male mating advantage in Drosophila ananassae. Genet Sel Evol 21:447–455
Singh BN, Pandey M (1993) Selection for high and low pupation height in Drosophila ananassae. Behav Genet 23:239–243
Singh SR, Singh BN (2001) Female remating in Drosophila ananassae: bidirectional selection for remating speed. Behav Genet 31:361–370
Singh SR, Singh BN (2003) Behavioral genetics of Drosophila ananassae. Genet Mol Res 2:394–409
Singh P, Singh BN (2008) Population genetics of Drosophila ananassae. Genet Res 90:409–419
Singh P, Singh BN (2010) Population genetics of Drosophila ananassae: evidence for population sub-structuring at the level of inversion polymorphism in Indian natural populations. Int J Biol 2:19–28
Sobel JM, Chen GF, Watt LR, Schemske DW (2009) The biology of speciation. Evolution 64:295–315
Som A, Singh BN (1998) No effect of marking the flies either by nail polish on scutellum or by wing clipping on mating success in Drosophila ananassae. Dros Inf Serv 81:202–203
Som A, Singh BN (2004) Rare male mating advantage for inversion karyotype in Drosophila ananassae. Behav Genet 34:335–342
Speith HT, Ringo JM (1983) Mating Behavior and sexual isolation in Drosophila. In: Ashburner MA, Carson HL, Thompson JN Jr (eds) The genetics and biology of Drosophila, vol 3c. Academic Press, New York, pp 223–284
Spiess EB (1968) Low frequency advantage in mating of Drosophila pseudoobscura karyotypes. Am Nat 102:363–379
Srivastava T, Singh BN (1996) Bidirectional selection for choice of oviposition site in Drosophila ananassae. Kor J Genet 18:295–300
Stalker HD (1942) Sexual isolation in the species complex Drosophila virilis. Genetics 27:238–257
Templeton AR (1981) The theory of speciation via the founder principle. Genetics 94:1038–1101
Ting CT, Takahashi A, Wu CI (2001) Incipient speciation by sexual isolation in Drosophila: concurrent evolution at multiple loci. Proc Natl Acad Sci USA 98:6709–6713
Turelli M, Barton NH, Coyne JA (2001) Theory and speciation. Tr Ecol Evol 16:330–343
Wassermen M, Koepfer HR (1980) Does asymmetrical mating preference show the direction of evolution? Evolution 34:1116–1126
Watanabe TK, Kawanishi M (1979) Mating preference and the direction of evolution in Drosophila. Science 205:906–907
Yukilevich R, True JR (2008) Incipient sexual isolation among cosmopolitan Drosophila melanogaster populations. Evolution 62:2112–2121
Zouros E, d’Entremont CJ (1980) Sexual isolation among populations of Drosophila mojavensis: response to pressure from a related species. Evolution 34:421–430
Acknowledgments
The financial support from the UGC, New Delhi in the form of a Major Research Project to BNS and Senior Research Fellowship from UGC (NET) to PN is gratefully acknowledged. We also thank the Prof. A. Fontdevila, Associate Editor and reviewers for their helpful suggestions on the original draft of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Nanda, P., Singh, B.N. Origin of sexual isolation in Drosophila ananassae due to founder effects. Genetica 139, 779–787 (2011). https://doi.org/10.1007/s10709-011-9582-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10709-011-9582-0