Abstract
Tris (2-chloroethyl) phosphate (TCEP), a typical organophosphate flame retardant, is of increasingly great concern considering their ubiquitous presence in aquatic environments and potential ecotoxicity. The present work was aimed to investigate the potential growth inhibition and hepatic stress induced by whole life-cycle exposure to TCEP (0.8, 4, 20 and 100 μg/L) in zebrafish. The results revealed that the body length, body mass and hepatic-somatic index (HSI) of zebrafish were significantly declined after exposure to TCEP for 120 days. GPx activity and GSH content were increased in the liver of zebrafish treated with low concentrations (0.8 and 4 μg/L) of TCEP, while exposure to high concentrations (20 and 100 μg/L) of TCEP reduced antioxidative capacity and elevated lipid peroxidation (LPO) levels. Gene transcription analysis demonstrated that the mRNA levels of nrf2 were altered in a similar manner to the transcription of the downstream genes nqo1 and hmox1, suggesting that Nrf2-Keap1 pathway mediated TCEP-induced oxidative stress in zebrafish liver. In addition, TCEP exposure might alleviate inflammatory response through down-regulating transcription of inflammatory cytokines (il-1β, il-6 and inos), and induce apoptosis via activating the p53-Bax pathway. Moreover, whole life-cycle exposure to TCEP caused a series of histopathological anomalies in zebrafish liver. Overall, our results revealed that lifetime exposure to environmentally relevant concentrations of TCEP could result in growth retardation and induce significant hepatotoxicity in zebrafish.






Similar content being viewed by others
Data availability
Data and materials will be made available on request.
References
Abdallah MAE, Covaci A (2014) Organophosphate flame retardants in indoor dust from egypt: implications for human exposure. Environ Sci Technol 48:4782–4789
Ali D, Alarifi S, Kumar S, Ahamed M, Siddiqui MA (2012) Oxidative stress and genotoxic effect of zinc oxide nanoparticles in freshwater snail Lymnaea luteola L. Aquat Toxicol 124–125:83–90
Arukwe A, Carteny CC, Eggen T (2016) Lipid peroxidation and oxidative stress responses in juvenile salmon exposed to waterborne levels of the organophosphate compounds tris (2-butoxyethyl)- and tris (2-chloroethyl) phosphates. J Toxicol Environ Health A 79:515–525
Benotti MJ, Trenholm RA, Vanderford BJ, Holady JC, Stanford BD, Snyder SA (2009) Pharmaceuticals and endocrine disrupting compounds in U.S. drinking water. Environ Sci Technol 43:597–603
Bernardi P, Petronilli V, Di Lisa F, Forte M (2001) A mitochondrial perspective on cell death. Trends Biochem Sci 26:112–117
Bollmann UE, Möller A, Xie Z, Ebinghaus R, Einax JW (2012) Occurrence and fate of organophosphorus flame retardants and plasticizers in coastal and marine surface waters. Water Res 46:531–538
Calaf GM, Echiburu-Chau C, Roy D (2009) Organophosphorous pesticides and estrogen induce transformation of breast cells affecting p53 and c-Ha-ras genes. Int J Oncol 35(5):1061–1068
Cantrell SM, LuTz LH, Tillitt DE, Hannlnk M (1996) Embryotoxicity of 2,3,7,8-Tetrachlorodibenzo-p-dioxin (TCDD): the embryonic vasculature is a physiological target for TCDD-induced DNA damage and apoptotic cell death in Medaka (Orizias iatipes). Toxicol Appl Pharmacol 141(1):23–34
Chen Q, Sun Y, Liu Z, Li Y (2017) Sex-dependent effects of subacute mercuric chloride exposure on histology, antioxidant status and immune-related gene expression in the liver of adult zebrafish (Danio rerio). Chemosphere 188:1–9
Chen H, Wang P, Du Z, Wang G, Gao S (2018) Oxidative stress, cell cycle arrest, DNA damage and apoptosis in adult zebrafish (Danio rerio) induced by tris (1,3-dichloro-2-propyl) phosphate. Aquat Toxicol 194:37–45
Cory S, Adams JM (2002) The Bcl2 family: regulators of the cellular life-or-death switch. Nat Rev Cancer 2:647–656
D’Arcy MS (2019) Cell death: a review of the major forms of apoptosis, necrosis and autophagy. Cell Biol Int 43:582–592
Deng S, Tian L, Liu F, Jin S, Liang G, Yang H, Du Z, Liu Y (2010) Toxic effects and residue of aflatoxin B1 in tilapia (Oreochromis niloticus×O. aureus) during long-term dietary exposure. Aquaculture 307:233–240
Engelsma MY, Huising MO, Van Muiswinkel WB, Flik G, Kwang J, Savelkoul HF, Verburg-van Kemenade BL (2002) Neuroendocrine–immune interactions in fish: a role for interleukin-1. Vet Immunol Immunopathol 87:467–479
Erkmen B, Karasu Benli AÇ, Ağuş HH, Yıldırım Z, Mert R, Erkoç F (2017) Impact of sublethal di-n-butyl phthalate on the aquaculture fish species Nile tilapia (Oreochromis niloticus): histopathology and oxidative stress assessment. Aquac Res 48:675–685
Fernandes C, Fontaínhas-Fernandes A, Rocha E, Salgado MA (2008) Monitoring pollution in Esmoriz-Paramos lagoon, Portugal: liver histological and biochemical effects in Liza saliens. Environ Monit Assess 145:315–322
Getachew Y, Cusimano FA, Gopal P, Reisman SA, Shay JW (2016) The synthetic triterpenoid RTA 405 (CDDO-EA) halts progression of liver fibrosis and reduces hepatocellular carcinoma size resulting in increased survival in an experimental model of chronic liver injury. Toxicol Sci 149:111–120
Hermann AC, Kim CH (2005) Effects of arsenic on zebrafish innate immune system. Mar Biotechnol 7:494–505
Hinton DE, Segner H, Braunbeck T (2017) Toxic responses of the liver. CRC Press
Hu F, Zhao Y, Yuan Y, Yin L, Dong F, Zhang W, Chen X (2021) Effects of environmentally relevant concentrations of tris (2-chloroethyl) phosphate (TCEP) on early life stages of zebrafish (Danio rerio). Environ Toxicol Pharmacol 83:1–7
Hu F, Zhao Y, Dong F, Wang H, Zheng M, Zhang W, Chen X (2022) Insights into the mechanisms of tris (2-chloroethyl) phosphate-induced growth inhibition in juvenile yellow catfish Pelteobagrus fulvidraco. Aquat Toxicol 247:106170
Jin Y, Zhang X, Shu L, Chen L, Sun L, Qian H, Liu W, Fu Z (2010) Oxidative stress response and gene expression with atrazine exposure in adult female zebrafish (Danio rerio). Chemosphere 78:846–852
Karan S, Dash P, Kaushik H, Sahoo PK, Garg LC, Dixit A (2016) Structural and functional characterization of recombinant interleukin-10 from Indian major carp Labeo rohita. J Immunol Res 2016:1–11
Kawagoshi Y, Fukanaga I, Itoh H (1999) Distribution of organophosphoric acid triesters between water and sediment at a sea-based solid waste disposal site. J Mater Cycles Waste Manage 1:53–61
Kim J, Cha Y, Surh Y (2010) A protective role of nuclear factor-erythroid 2-related factor-2 (Nrf2) in inflammatory disorders. Mutat Res-Fundam Mol Mech Mutagen 690:12–23
Kopecka J, Pempkowiak J (2008) Temporal and spatial variations of selected biomarker activities in flounder (Platichthys flesus) collected in the Baltic proper. Ecotoxicol Ecotoxicol Environ Safety 70:379–391
Kumar S (2007) Caspase function in programmed cell death. Cell Death Diff Integral Equ 14:32–43
Lackner R (1998) “Oxidative stress” in fish by environmental pollutants. Fish Ecotoxicol 203–224
Larsson Å, Haux C, Sjöbeck M-L, Lithner G (1984) Physiological effects of an additional stressor on fish exposed to a simulated heavy-metal-containing effluent from a sulfide ore smeltery. Ecotoxicol Environ Saf 8:118–128
Li H, Yuan S, Su G, Li M, Wang Q, Zhu G, Liu C (2017) Whole-life-stage characterization in the basic biology of Daphnia magna and effects of TDCIPP on growth, reproduction, survival, and transcription of genes. Environ Sci Technol 51:13967–13975
Li R, Wang H, Mi C, Feng C, Zhang L, Yang L, Zhou B (2019) The adverse effect of TCIPP and TCEP on neurodevelopment of zebrafish embryos/larvae. Chemosphere 220:811–817
Liu X, Ji K, Jo A, Moon HB, Choi K (2013) Effects of TDCPP or TPP on gene transcriptions and hormones of HPG axis, and their consequences on reproduction in adult zebrafish (Danio rerio). Aquat Toxicol 134–135:104–111
Liu C, Su G, Giesy JP, Letcher RJ, Li G, Agrawal I, Li J, Yu L, Wang J, Gong Z (2016) Acute exposure to tris (1,3-dichloro-2-propyl) phosphate (TDCIPP) causes hepatic inflammation and leads to hepatotoxicity in zebrafish. Sci Rep 6(1):19045
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2−ΔΔCT method. Methods 25:402–408
Marklund A, Andersson B, Haglund P (2005) Traffic as a source of organophosphorus flame retardants and plasticizers in snow. Environ Sci Technol 39:3555–3562
McIlwain DR, Berger T, Mak TW (2013) Caspase functions in cell death and disease. Cold Spring Harb Perspect Biol 5(4):a008656
Moalem G, Leibowitz-Amit R, Yoles E, Mor F, Cohen IR, Schwartz M (1999) Autoimmune T cells protect neurons from secondary degeneration after central nervous system axotomy. Nat Med 5:49–55
Moser VC, Phillips PM, Hedge JM, McDaniel KL (2015) Neurotoxicological and thyroid evaluations of rats developmentally exposed to tris (1,3-dichloro-2-propyl) phosphate (TDCIPP) and tris (2-chloro-2-ethyl) phosphate (TCEP). Neurotoxicol Teratol 52:236–247
Özdemir S, Altun S, Arslan H (2018) Imidacloprid exposure cause the histopathological changes, activation of TNF-α, iNOS, 8-OHdG biomarkers, and alteration of caspase 3, iNOS, CYP1A, MT1 gene expression levels in common carp (Cyprinus carpio L.). Toxicol Rep 5:125–133
Polekhina G, Board PG, Gali RR, Rossjohn J, Parker MW (1999) Molecular basis of glutathione synthetase deficiency and a rare gene permutation event. EMBO J 18:3204–3213
Ramesh M, Anitha S, Poopal RK, Shobana C (2018) Evaluation of acute and sublethal effects of chloroquine (C18H26CIN3) on certain enzymological and histopathological biomarker responses of a freshwater fish Cyprinus carpio. Toxicol Rep 5:18–27
Ray P, Huang B, Tsuji Y (2012) Reactive oxygen species (ROS) homeostasis and redox regulation in cellular signaling. Cell Signal 24:981–990
Saha RN, Pahan K (2006) Regulation of inducible nitric oxide synthase gene in glial cells. Antioxid Redox Signal 8:929–947
Shaw P, Mondal P, Bandyopadhyay A, Chattopadhyay A (2019) Environmentally relevant concentration of chromium activates Nrf2 and alters transcription of related XME genes in liver of zebrafish. Chemosphere 214:35–46
Sule RO, Condon L, Gomes AV (2022) A common feature of pesticides: oxidative stress-the role of oxidative stress in pesticide-induced toxicity. Oxidative Med Cell Longev 2022
Sun L, Tan H, Peng T, Wang S, Xu W, Qian H, Jin Y, Fu Z (2016) Developmental neurotoxicity of organophosphate flame retardants in early life stages of Japanese medaka (Oryzias latipes). Environ Toxicol Chem 35:2931–2940
Sutha J, Anila PA, Umamaheswari S, Ramesh M, Narayanasamy A, Poopal RK, Ren Z (2020) Biochemical responses of a freshwater fish Cirrhinus mrigala exposed to tris (2-chloroethyl) phosphate (TCEP). Environ Sci Pollut Res 27:34369–34387
Sutha J, Anila PA, Gayathri M, Ramesh M (2022) Long term exposure to tris (2-chloroethyl) phosphate (TCEP) causes alterations in reproductive hormones, vitellogenin, antioxidant enzymes, and histology of gonads in zebrafish (Danio rerio): in vivo and computational analysis. Comp Biochem Physiol C-Toxicol Pharmacol 254:109263
Tian D, Yu Y, Lu L, Tong D, Zhang X, Shi W, Liu G (2023) Tris(2-chloroethyl) Phosphate Exerts Hepatotoxic Impacts on Zebrafish by Disrupting Hypothalamic–Pituitary–Thyroid and Gut-Liver Axes. Environ Sci Technol 57(24):9043–9054
Tsuchiya Y, Nakajima M, Yokoi T (2005) Cytochrome P450-mediated metabolism of estrogens and its regulation in human. Cancer Lett 227:115–124
Van den Eede N, Maho W, Erratico C, Neels H, Covaci A (2013) First insights in the metabolism of phosphate flame retardants and plasticizers using human liver fractions. Toxicol Lett 223:9–15
Vliegenthart ADB, Tucker CS, Del Pozo J, Dear JW (2014) Zebrafish as model organisms for studying drug-induced liver injury. Br J Clin Pharmacol 78:1217–1227
Wang X, Liu J, Yin Y (2011) Development of an ultra-high-performance liquid chromatography–tandem mass spectrometry method for high throughput determination of organophosphorus flame retardants in environmental water. J Chromatogr A 1218:6705–6711
Wang G, Shao J, Wu M, Meng Y, Gul Y, Yang H, Xiong D (2019a) Effect of acute exposure of triazophos on histological structure and apoptosis of the brain and liver of zebrafish (Danio rerio). Ecotoxicol Environ Saf 180:646–655
Wang Y, Zhang Y, Li W, Yang L, Guo B (2019b) Distribution, metabolism and hepatotoxicity of neonicotinoids in small farmland lizard and their effects on GH/IGF axis. Sci Total Environ 662:834–841
Wang C, Chen Z, Lu Y, Wang L, Zhang Y, Zhu X, Song J (2020) Neurotoxicity and related mechanisms of flame retardant TCEP exposure in mice. Toxicol Mech Methods 30:490–496
Wang Y, Tian J, Shi F, Li X, Hu Z, Chu J (2021) Protective effect of surfactin on copper sulfate-induced inflammation, oxidative stress, and hepatic injury in zebrafish. Microbiol Immunol 65:410–421
Wang H, Jing C, Peng H, Liu S, Zhao H, Zhang W, Chen X, Hu F (2022) Parental whole life-cycle exposure to tris (2-chloroethyl) phosphate (TCEP) disrupts embryonic development and thyroid system in zebrafish offspring. Ecotoxicol Environ Saf 248:114313
Wang X, Zhang J, Lu C, Liu Y, Yang X, Hou K, Du Z, Li B, Juhasz A, Zhu L (2023) Development toxicity and cytotoxicity of pyroxsulam on embryos and adults of zebrafish (Danio rerio). Environ Pollut 319:121040
Xu T, Wang Q, Shi Q, Fang Q, Guo Y, Zhou B (2015) Bioconcentration, metabolism and alterations of thyroid hormones of Tris (1,3-dichloro-2-propyl) phosphate (TDCPP) in Zebrafish. Environ Toxicol Pharmacol 40:581–586
Xu Q, Wu D, Dang Y, Yu L, Liu C, Wang J (2017) Reproduction impairment and endocrine disruption in adult zebrafish (Danio rerio) after waterborne exposure to TBOEP. Aquat Toxicol 182:163–171
Yu L, Jia Y, Su G, Sun Y, Letcher RJ, Giesy JP, Yu H, Han Z, Liu C (2017) Parental transfer of tris (1,3-dichloro-2-propyl) phosphate and transgenerational inhibition of growth of zebrafish exposed to environmentally relevant concentrations. Environ Pollut 220:196–203
Zanotti S, Kumar A, Kumar A (2002) Cytokine modulation in sepsis and septic shock. Expert Opin Investig Drugs 11:1061–1075
Zhang J, Shen H, Wang X, Wu J, Xue Y (2004) Effects of chronic exposure of 2,4-dichlorophenol on the antioxidant system in liver of freshwater fish Carassius auratus. Chemosphere 55:167–174
Zhao M, Zhang Y, Wang C, Fu Z, Liu W, Gan J (2009) Induction of macrophage apoptosis by an organochlorine insecticide acetofenate. Chem Res Toxicol 22:504–510
Zhou X, Liang Y, Ren G, Zheng K, Wu Y, Zeng X, Zhong Y, Yu Z, Peng P (2020) Biotransformation of tris (2-chloroethyl) phosphate (TCEP) in sediment microcosms and the adaptation of microbial communities to TCEP. Environ Sci Technol 54:5489–5497
Funding
This work was supported by National College Students Innovation and Entrepreneurship Training Program (China, 202310389025).
Author information
Authors and Affiliations
Contributions
FH: Writing -Writing - Review & Editing, Investigation, Supervision, Project administration. WL: Conceptualization, Methodology, Validation, Investigation, Writing - original draft & Review, Funding acquisition. HW: Conceptualization, Methodology, Formal analysis, Investigation. HP: Validation, Visualization. JH: Investigation. JD: Validation. WZ: Validation. All the authors revised and approved the ms.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethical approval
Our experimental protocols were approved by Laboratory Animals Ethics and Welfare Committee of College of Animal Science, Fujian Agriculture and Forestry University (PZCASFAFU22039).
Conflict of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Highlights
Whole life-cycle exposure to environmental relevant concentrations of TCEP could inhibit the growth of zebrafish.
Exposure to TCEP induced oxidative stress and led to lipid peroxidation in zebrafish liver.
Inflammatory response might be alleviated through the down-regulation of inflammatory cytokines mRNA expression.
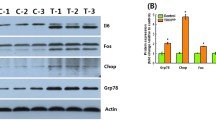
Whole life-cycle exposure to TCEP might induce apoptosis through the activation of p53-Bax pathway.
Whole life-cycle exposure to TCEP resulted in a series of histopathological anomalies in zebrafish liver.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Hu, F., Li, W., Wang, H. et al. Environmentally relevant concentrations of tris (2-chloroethyl) phosphate (TCEP) induce hepatotoxicity in zebrafish (Danio rerio): a whole life-cycle assessment. Fish Physiol Biochem 49, 1421–1433 (2023). https://doi.org/10.1007/s10695-023-01265-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10695-023-01265-7