Abstract
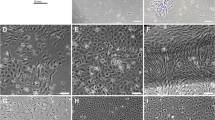
Skeletal muscle myoblastic cell lines can provide a valuable new in vitro model for the exploration of the mechanisms that control skeletal muscle development and its associated molecular regulation. In this study, the skeletal muscle tissues of grass carp were digested with trypsin and collagenase I to obtain the primary myoblast cell culture. Myoblast cells were obtained by differential adherence purification and further analyzed by cryopreservation and resuscitation, chromosome analysis, immunohistochemistry, and immunofluorescence. A continuous grass carp myoblast cell line (named CIM) was established from grass carp (Ctenopharyngodon idellus) muscle and has been subcultured > 100 passages in a year and more. The CIM cells revived at 79.78–95.06% viability after 1–6 months of cryopreservation, and shared a population doubling time of 27.24 h. The number of modal chromosomes of CIM cells was 48, and the mitochondrial 12S rRNA sequence of the CIM cell line shared 99% identity with those of grass carp registered in GenBank. No microorganisms (bacteria, fungi, or mycoplasma) were detected during the whole study. The cell type of CIM cells was proven to be myoblast by immunohistochemistry of specific myogenic protein markers, including CD34, desmin, MyoD, and MyHC, as well as relative expression of key genes. And the myogenic rate and fusion index of this cell line after 10 days of induced differentiation were 8.96 ~ 9.42% and 3–24%, respectively. The telomerase activity and transfection efficiency of CIM cell line were 0.027 IU/mgprot and 23 ~ 24%, respectively. These results suggest that a myoblast cell line named CIM with normal biological function has been successfully established, which may provide a valuable tool for related in vitro studies.









Similar content being viewed by others
Data availability
Not applicable.
References
Alfaro LAS, Dick SA, Siegel AL, Anonuevo AS, McNagny KM, Megeney LA, Cornelison DD, Rossi FM (2011) CD34 promotes satellite cell motility and entry into proliferation to facilitate efficient skeletal muscle regeneration. Stem Cells 29(12):2030–2041. https://doi.org/10.1002/stem.759
Ali S, Mahmood A (2017) Microbial and viral contamination of animal and stem cell cultures: common contaminants. Detect Elimination. https://doi.org/10.15406/jsrt.2017.02.00078
Buckingham M (2001) Skeletal muscle formation in vertebrates. Curr Opin Genet Dev 11(4):440–448. https://doi.org/10.1016/S0959-437X(00)00215-X
Cao Y, Kumar RM, Penn BH, Berkes CA, Kooperberg C, Boyer LA, Young RA, Tapscott SJ (2006) Global and gene-specific analyses show distinct roles for Myod and Myog at a common set of promoters. EMBO J 25(3):502–511. https://doi.org/10.1038/sj.emboj.7600958
Collura RV, Stewart CB (1995) Insertions and duplications of mtDNA in the nuclear genomes of Old World monkeys and hominoids. Nature 378(6556):485–489
Csapo R, Gumpenberger M, Wessner B (2020) Skeletal muscle extracellular matrix—what do we know about its composition, regulation, and physiological roles? A Narrative review. Front Physiol 11:253. https://doi.org/10.3389/fphys.2020.00253
Cuénod A, Foucault F, Pflüger V, Egli A (2021) Factors associated with MALDI-TOF mass spectral quality of species identification in clinical routine diagnostics. Front Cell Infect Microbiol 11:104. https://doi.org/10.3389/fcimb.2021.646648
Doyle A, Hay R, Kirsop BE (1990) Animal cells, living resources for biotechnology. Cambridge University Press, Cambridge, UK
Fauconneau B, Paboeuf G (2000) Effect of fasting and refeeding on in vitro muscle cell proliferation in rainbow trout (Oncorhynchus mykiss). Cell Tissue Res 301(3):459–463. https://doi.org/10.1007/s004419900168
Froehlich JM, Seiliez I, Gabillard J-C, Biga PR (2014) Preparation of primary myogenic precursor cell/myoblast cultures from basal vertebrate lineages. Jove-J vis Exp 86:e51354. https://doi.org/10.3791/51354
Gabillard JC, Sabin N, Paboeuf G (2010) In vitro characterization of proliferation and differentiation of trout satellite cells. Cell Tissue Res 342(3):471–477. https://doi.org/10.1007/s00441-010-1071-8
Guan WJ, Liu CQ, Li CY, Liu D, Zhang WX, Ma YH (2010) Establishment and cryopreservation of a fibroblast cell line derived from Bengal tiger (Panthera Tigris Tigris). CryoLetters 31(2):130–138. https://doi.org/10.1016/j.cbpa.2009.12.013
Hiyama E, Hiyama K (2007) Telomere and telomerase in stem cells. Brit J Cancer 96(7):1020–1024. https://doi.org/10.1038/sj.bjc.6603671
Hossain MM, Ali ML, Khan S, Haque MM, Shahjahan M (2020) Use of Asian watergrass as feed of grass carp. Aquac Rep 18:100434. https://doi.org/10.1016/j.aqrep.2020.100434
Huang X, Huang Y, Ouyang Z, Xu L, Yan Y, Cui H, Han X, Qin Q (2011) Singapore grouper iridovirus, a large DNA virus, induces nonapoptotic cell death by a cell type dependent fashion and evokes ERK signaling. Apoptosis 16(8):831–845. https://doi.org/10.1007/s10495-011-0616-y
Hubbard BD, Lazarides E (1979) Copurification of actin and desmin from chicken smooth muscle and their copolymerization in vitro to intermediate filaments. J Cell Biol 80(1):166–182. https://doi.org/10.1083/jcb.80.1.166
Kaufman SJ, Foster RF (1988) Replicating myoblasts express a muscle-specific phenotype. PNAS 85(24):9606. https://doi.org/10.1073/pnas.85.24.9606
Kong X, Wang X, Li M, Song W, Huang K, Zhang F, Zhang Q, Qi J, He Y (2021) Establishment of myoblast cell line and identification of key genes regulating myoblast differentiation in a marine teleost. Sebastes Schlegelii Gene 802:145869. https://doi.org/10.1016/j.gene.2021.145869
Koumans JTM, Akster HA, Dulos GJ, Osse JWM (1990) Myosatellite cells of Cyprinus carpio (Teleostei) in vitro: isolation, recognition and differentiation. Cell Tissue Res 261(1):173–181. https://doi.org/10.1007/BF00329450
Kumar A, Singh N, Goswami M, Srivastava J, Lakra W (2016) Establishment and characterization of a new muscle cell line of zebrafish (Danio rerio) as an in vitro model for gene expression studies. J Anim Sci Biotechno 27(3):166–173. https://doi.org/10.1080/10495398.2016.1147455
Lazarides E, Hubbard BD (1977) Immunological characterization of the subunit of the 100 filaments from muscle cells. PNAS 73(12):4344–4348. https://doi.org/10.1073/pnas.73.12.4344
Li H, Yang J, Wei X, Song C, Dong D, Huang Y, Lan X, Plath M, Lei C, Ma Y (2018) CircFUT10 reduces proliferation and facilitates differentiation of myoblasts by sponging miR-133a. J Cell Physiol 233(6):4643–4651. https://doi.org/10.1002/jcp.26230
Lina KD, Ellen G, Barbara GM, Aurélie J, Danielle G, Shahragim T (2018) Pax3/Pax7 mark a novel population of primitive myogenic cells during development. Genes Dev 19(12):1426–1431. https://doi.org/10.1101/gad.345505
Liu L (1980) On karyotype of the grass-carp, ctenopharyngodon idellus. Acta Zoologica Sinica 26(2):126–131 (In Chinese with English abstract)
Mazzaro G, Bossi G, Coen S, Sacchi A, Soddu S (1999) The role of wild-type p53 in the differentiation of primary hemopoietic and muscle cells. Oncogene 18(42):5831–5835. https://doi.org/10.1038/sj.onc.1202962
Mitchell KJ, Pannérec A, Cadot B, Parlakian A, Besson V, Gomes ER, Marazzi G, Sassoon DA (2010) Identification and characterization of a non-satellite cell muscle resident progenitor during postnatal development. Nat Cell Biol 12(3):257–266. https://doi.org/10.1038/ncb2025
Montarras D, Morgan J, Collins C, Relaix F, Zaffran S, Cumano A, Partridge T, Buckingham M (2005) Direct isolation of satellite cells for skeletal muscle regeneration. Science 309(5743):2064–2067
Montserrat N, Sánchez-Gurmaches J, García de la Serrana D, Navarro MI, Gutiérrez J (2007) IGF-I binding and receptor signal transduction in primary cell culture of muscle cells of gilthead sea bream: changes throughout in vitro development. Cell Tissue Res 330(3):503–513. https://doi.org/10.1007/s00441-007-0507-2
Morgan JE, Partridge TA (2003) Muscle satellite cells. Int J Biochem Cell Biol 35(8):1151–1156. https://doi.org/10.1016/S1357-2725(03)00042-6
Nurmik M, Ullmann P, Rodriguez F, Haan S, Letellier E (2020) In search of definitions: Cancer-associated fibroblasts and their markers. Int J Cancer 146(4):895–905. https://doi.org/10.1002/ijc.32193
Pfaffl MW (2001) A new mathematical model for relative quantification in real-time RT–PCR. Nucleic Acids Res 29(9):e45–e45. https://doi.org/10.1093/nar/29.9.e45
Quinn ME, Goh Q, Kurosaka M, Gamage DG, Petrany MJ, Prasad V, Millay DP (2017) Myomerger induces fusion of non-fusogenic cells and is required for skeletal muscle development. Nat Commun 8(1):15665. https://doi.org/10.1038/ncomms15665
Rudnicki MA, Schnegelsberg PNJ, Stead RH, Braun T, Arnold H-H, Jaenisch R (1993) MyoD or Myf-5 is required for the formation of skeletal muscle. Cell 75(7):1351–1359. https://doi.org/10.1016/0092-8674(93)90621-V
Schiaffino S, Reggiani C (2011) Fiber types in mammalian skeletal muscles. Physiol Rev 91(4):1447–1531. https://doi.org/10.1152/physrev.00031.2010
Seale P, Sabourin LA, Girgis-Gabardo A, Mansouri A, Gruss P, Rudnicki MA (2000) Pax7 is Required for the specification of myogenic satellite cells. Cell 102(6):777–786. https://doi.org/10.1016/S0092-8674(00)00066-0
Sinha S, Elbaz-Alon Y, Avinoam O (2022) Ca2+ as a coordinator of skeletal muscle differentiation, fusion and contraction. FEBS J. https://doi.org/10.1111/febs.16552
Tone Y, Mamchaoui K, Tsoumpra MK, Hashimoto Y, Terada R, Maruyama R, Gait MJ, Arzumanov AA, McClorey G, Imamura M, Si T, Yokota T, Wood MJA, Mouly V, Aoki Y (2021) Immortalized canine dystrophic myoblast cell lines for development of peptide-conjugated splice-switching oligonucleotides. Nucleic Acid Ther 31(2):172–181. https://doi.org/10.1089/nat.2020.0907
Uaesoontrachoon K, Yoo H-J, Tudor EM, Pike RN, Mackie EJ, Pagel CN (2008) Osteopontin and skeletal muscle myoblasts: association with muscle regeneration and regulation of myoblast function in vitro. Int J Biochen Cell B 40(10):2303–2314. https://doi.org/10.1016/j.biocel.2008.03.020
Uno Y, Nozu R, Kiyatake I, Higashiguchi N, Sodeyama S, Murakumo K, Sato K, Kuraku S (2020) Cell culture-based karyotyping of orectolobiform sharks for chromosome-scale genome analysis. Commun 3(1):652. https://doi.org/10.1038/s42003-020-01373-7
Weintraub H, DaVis R, Tapscott S, Thayer M, Krause M, Benezra R, Blackwell T, Turner D, Rupp R, Hollenberg S (1991) The myoD gene family: nodal point during specification of the muscle cell lineage. Science 251(4995):761–766. https://doi.org/10.1126/science.1846704
Yafee D, Saxel O (1977) Serial passaging and differentiation of myogenic cells isolated from dystrophic mouse muscle. Nature 270(5639):725–727. https://doi.org/10.1038/270725a0
Yang L, Tan Z, Wang D, Xue L, Guan M-x, Huang T, Li R (2014) Species identification through mitochondrial rRNA genetic analysis. Sci Rep 4(1):4089. https://doi.org/10.1038/srep04089
Zammit PS, Partridge TA, Yablonka-Reuveni Z (2006) The skeletal muscle satellite cell: the stem cell that came in from the cold. J Histochem Cytochem 54(11):1177–1191
Zhang W, Tong H, Zhang Z, Shao S, Liu D, Li S, Yan Y (2018) Transcription factor EGR1 promotes differentiation of bovine skeletal muscle satellite cells by regulating MyoG gene expression. J Cell Physiol 233(1):350–362. https://doi.org/10.1002/jcp.25883
Zhi XC, Yeap SK, Wan YH (2021) Transfection types, methods and strategies: a technical review. PeerJ 9(1):e11165. https://doi.org/10.7717/peerj.11165
Zhu DM, Yang K, Wang WM, Song W (2013) Establishment and characterization of a fin cell line from blunt snout bream, Megalobrama amblycephala. Fish Physiol Biochem 39(6):1399–1410. https://doi.org/10.1007/s10695-013-9794-6
Acknowledgements
The authors thank the College of Fisheries of Huazhong Agricultural University for the infrastructure provided. Finally, thanks to everyone who contributed to this experiment.
Funding
This work was supported by the National Natural Science Foundation of China (Grant No.: 32072950).
Author information
Authors and Affiliations
Contributions
Conceptualization: Xianmei Long, Wangwang Chen, Qingsong Tan; formal analysis: Xianmei Long, Guoqing Liu, Qingsong Tan; investigation: Xianmei Long, Wangwang Chen, Wenguang Hu; methodology: Xianmei Long, Qingsong Tan; data curation: Xianmei Long, Wangwang Chen; writing — original draft: Xianmei Long; Resources: Guoqing Liu; project administration: Qingsong Tan; writing — review and editing: Xianmei Long, Qingsong Tan. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval
All animal experiments complied with the ARRIVE guidelines and were carried out in accordance with the UK Animals (Scientific Procedures). What is more, the animal sample collection and experimental protocols were approved by the Institutional Ethics Committee of Huazhong Agriculture University (NO. FISH-2020–04-03). All animal handling and methods were performed according to the relevant guidelines.
Consent to participate
All authors have consented to participate to this study and approved the final manuscript.
Consent for publication
All authors have read, reviewed, and revised this article, and all agree to publish this article.
Competing interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Long, X., Chen, W., Liu, G. et al. Establishment and characterization of a skeletal myoblast cell line of grass carp (Ctenopharyngodon idellus). Fish Physiol Biochem 49, 1043–1061 (2023). https://doi.org/10.1007/s10695-023-01246-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10695-023-01246-w