Abstract
Gamete production is a fundamental process for reproduction; however, exposure to stress, such as increased environmental temperature, can decrease or even interrupt this process, affecting fertility. Thus, the survival of spermatogonial stem cells (SSCs) is crucial for the recovery of spermatogenesis upon stressful situations. Here, we show that the Notch pathway is implicated in such survival, by protecting the SSCs against thermal stress. First, we corroborated the impairment of spermatogenesis under heat stress in medaka, observing an arrest in metaphase I at 10 days of heat treatment, an increase in the number of spermatocytes, and downregulation of ndrg1b and sycp3. In addition, at 30 days of treatment, an interruption of spermatogenesis was observed with a strong loss of spermatocytes and spermatids. Then, the exposure of adult males to thermal stress condition induced apoptosis mainly in spermatogenic and supporting somatic cells, with the exception of the germinal region, where SSCs are located. Concomitantly, the Notch pathway–related genes were upregulated, including the ligands (dll4, jag1-2) and receptors (notch1a-3). Moreover, during thermal stress presenilin enhancer-2 (pen-2), the catalytic subunit of γ-secretase complex of the Notch pathway was restricted to the germinal region of the medaka testis, observed in somatic cells surrounding type A spermatogonia (SGa). The importance of Notch pathway was further supported by an ex vivo approach, in which the inhibition of this pathway activity induced a loss of SSCs. Overall, this study supports the importance of Notch pathways for the protection of SSCs under chronic thermal stress.





Similar content being viewed by others
Data availability
All data generated or analyzed during this study are included in this article and its supplementary information files. Raw data are available from the authors upon reasonable request.
References
Alix M, Kjesbu OS, Anderson KC (2020) From gametogenesis to spawning: how climate-driven warming affects teleost reproductive biology. J Fish Biol. https://doi.org/10.1111/jfb.14439
Arias Padilla LF, Castañeda-Cortés DC, Rosa IF, Moreno Acosta OD, Hattori RS, Nóbrega RH, Fernandino JI (2021) Cystic proliferation of germline stem cells is necessary to reproductive success and normal mating behavior in medaka. Elife 10:e62757. https://doi.org/10.7554/eLife.62757
Blackshaw A, Massey P (1978) The effect of cryptorchidism on the quantitative histology, histochemistry and hydrolytic enzyme activity of the rat testis. Aust J Biol Sci 31:53. https://doi.org/10.1071/BI9780053
Blanpain C, Lowry WE, Pasolli HA, Fuchs E (2006) Canonical notch signaling functions as a commitment switch in the epidermal lineage. Genes Dev 20:3022–3035. https://doi.org/10.1101/gad.1477606
Bray SJ (2006) Notch signalling: a simple pathway becomes complex. Nat Rev Mol Cell Biol 7:678–689. https://doi.org/10.1038/nrm2009
Butzge AJ, Yoshinaga TT, Acosta ODM, Fernandino JI, Sanches EA, Tabata YA, de Oliveira C, Takahashi NS, Hattori RS (2021) Early warming stress on rainbow trout juveniles impairs male reproduction but contrastingly elicits intergenerational thermotolerance. Sci Rep 11:17053. https://doi.org/10.1038/s41598-021-96514-1
Campbell WA, Yang H, Zetterberg H, Baulac S, Sears JA, Liu T, Wong STC, Zhong TP, Xia W (2006) Zebrafish lacking Alzheimer presenilin enhancer 2 (Pen-2) demonstrate excessive p53-dependent apoptosis and neuronal loss. J Neurochem 96:1423–1440. https://doi.org/10.1111/j.1471-4159.2006.03648.x
Carlsen E (2003) History of febrile illness and variation in semen quality. Hum Reprod 18:2089–2092. https://doi.org/10.1093/humrep/deg412
Castañeda Cortés DC, Arias Padilla LF, Langlois VS, Somoza GM, Fernandino JI (2019) The central nervous system acts as a transducer of stress-induced masculinization through corticotropin-releasing hormone B. Dev 146:1–10. https://doi.org/10.1242/dev.172866
Chadwick N, Zeef L, Portillo V, Boros J, Hoyle S, van Doesburg JCL, Buckle A-M (2010) Notch protection against apoptosis in T-ALL cells mediated by GIMAP5. Blood Cells. Mol Dis 45:201–209. https://doi.org/10.1016/j.bcmd.2010.07.006
Chen C, Ouyang W, Grigura V, Zhou Q, Carnes K, Lim H, Zhao G-Q, Arber S, Kurpios N, Murphy TL, Cheng AM, Hassell JA, Chandrashekar V, Hofmann M-C, Hess RA, Murphy KM (2005) ERM is required for transcriptional control of the spermatogonial stem cell niche. Nature 436:1030–1034. https://doi.org/10.1038/nature03894
Chen J, Cai T, Zheng C, Lin X, Wang G, Liao S, Wang X, Gan H, Zhang D, Hu X, Wang S, Li Z, Feng Y, Yang F, Han C (2017) MicroRNA-202 maintains spermatogonial stem cells by inhibiting cell cycle regulators and RNA binding proteins. Nucleic Acids Res 45:4142–4157. https://doi.org/10.1093/nar/gkw1287
Chiarini-Garcia H, Hornick JR, Griswold MD, Russell LD (2001) Distribution of type A spermatogonia in the mouse is not random. Biol Reprod 65:1179–1185. https://doi.org/10.1095/biolreprod65.4.1179
Conboy IM, Rando TA (2002) The regulation of notch signaling controls satellite cell activation and cell fate determination in postnatal myogenesis. Dev Cell 3:397–409. https://doi.org/10.1016/S1534-5807(02)00254-X
de Rooij DG (2017) The nature and dynamics of spermatogonial stem cells. Development 144:3022–3030. https://doi.org/10.1242/dev.146571
de Alvarenga ÉR, de França LR (2009) Effects of different temperatures on testis structure and function, with emphasis on somatic cells, in sexually mature Nile tilapias (Oreochromis niloticus)1. Biol Reprod 80:537–544. https://doi.org/10.1095/biolreprod.108.072827
dos Santos N, Silva M, Vale A (2008) Fish and apoptosis: studies in disease and pharmaceutical design. Curr Pharm Des 14:170–183. https://doi.org/10.2174/138161208783378734
Fernandino JI, Popesku JT, Paul-Prasanth B, Xiong H, Hattori RS, Oura M, Strüssmann CA, Somoza GM, Matsuda M, Nagahama Y, Trudeau VL (2011) Analysis of sexually dimorphic expression of genes at early gonadogenesis of pejerrey odontesthes bonariensis using a heterologous microarray. Sex Dev 5:89–101. https://doi.org/10.1159/000324423
Fernandino JI, Hattori RS, Kishii A, Strüssmann CA, Somoza GM (2012) The cortisol and androgen pathways cross talk in high temperature-induced masculinization: The 11β-hydroxysteroid dehydrogenase as a key enzyme. Endocrinology 153:6003–6011. https://doi.org/10.1210/en.2012-1517
Fischer A, Gessler M (2007) Delta–Notch—and then? Protein interactions and proposed modes of repression by Hes and Hey bHLH factors. Nucleic Acids Res 35:4583–4596. https://doi.org/10.1093/nar/gkm477
Fortini ME (2002) γ-Secretase-mediated proteolysis in cell-surface-receptor signalling. Nat Rev Mol Cell Biol 3:673–684. https://doi.org/10.1038/nrm910
Froschauer A, Khatun MSTM, Sprott D, Franz A, Rieger C, Pfennig F, Gutzeit HO (2013) oct4-EGFP reporter gene expression marks the stem cells in embryonic development and in adult gonads of transgenic medaka. 58:48–58. https://doi.org/10.1002/mrd.22135
Garcia TX, Hofmann M-C (2013) NOTCH signaling in Sertoli cells regulates gonocyte fate. Cell Cycle 12:2538–2545. https://doi.org/10.4161/cc.25627
Garcia TX, DeFalco T, Capel B, Hofmann M-C (2013) Constitutive activation of NOTCH1 signaling in Sertoli cells causes gonocyte exit from quiescence. Dev Biol 377:188–201. https://doi.org/10.1016/j.ydbio.2013.01.031
Garcia TX, Farmaha JK, Kow S, Hofmann M-C (2014) RBPJ in mouse Sertoli cells is required for proper regulation of the testis stem cell niche. Development 141:4468 LP – 4478. https://doi.org/10.1242/dev.113969
Garcia TX, Parekh P, Gandhi P, Sinha K, Hofmann M-C (2017) The NOTCH Ligand JAG1 Regulates GDNF Expression in Sertoli cells. Stem Cells Dev 26:585–598. https://doi.org/10.1089/scd.2016.0318
Hansen PJ (2009) Effects of heat stress on mammalian reproduction. Philos Trans r Soc B Biol Sci 364:3341–3350. https://doi.org/10.1098/rstb.2009.0131
Hayashi Y, Kobira H, Yamaguchi T, Shiraishi E, Yazawa T, Hirai T, Kamei Y, Kitano T (2010) High temperature causes masculinization of genetically female medaka by elevation of cortisol. Mol Reprod Dev 77:679–686. https://doi.org/10.1002/mrd.21203
Henrique D, Schweisguth F (2019) Mechanisms of Notch signaling: a simple logic deployed in time and space. Development 146:dev172148. https://doi.org/10.1242/dev.172148
Hutson JM, Li R, Southwell BR, Petersen BL, Thorup J, Cortes D (2013) Germ celldevelopment in the postnatal testis: the key to prevent malignancy in cryptorchidism? Front Endocrinol (Lausanne) 3:176. https://doi.org/10.3389/fendo.2012.00176
Ito LS, Takahashi C, Yamashita M, Strüssmann CA (2008b) Warm water induces apoptosis, gonadal degeneration, and germ cell loss in subadult pejerrey Odontesthes bonariensis (pisces, atheriniformes). Physiol Biochem Zool 81:762–774. https://doi.org/10.1086/590219
Ito LS, Cornejo AM, Yamashita M (2008a) Thermal threshold and histological process of heat-induced sterility in adult Pejerrey (Odontesthes bonariensis ): a comparative analysis of laboratory and wild specimens. 81:775–784. https://doi.org/10.1086/591035
Iwai T, Yoshii A, Yokota T, Sakai C, Hori H, Kanamori A, Yamashita M (2006) Structural components of the synaptonemal complex, SYCP1 and SYCP3, in the medaka fish Oryzias latipes. Exp Cell Res 312:2528–2537. https://doi.org/10.1016/j.yexcr.2006.04.015
Iwasaki Y, Ohkawa K, Sadakata H, Kashiwadate A, Takayama-Watanabe E, Onitake K, Watanabe A (2009) Two states of active spermatogenesis switch between reproductive and non-reproductive seasons in the testes of the medaka, Oryzias latipes. Dev Growth Differ 51:521–532. https://doi.org/10.1111/j.1440-169X.2009.01114.x
Kadekar P, Roy R (2019) AMPK regulates germline stem cell quiescence and integrity through an endogenous small RNA pathway. PLOS Biol 17:e3000309
Kageyama R, Ohtsuka T, Kobayashi T (2007) The Hes gene family: repressors and oscillators that orchestrate embryogenesis. Development 134:1243–1251
Kimble J, Crittenden SL (2007) Controls of germline stem cells, entry into meiosis, and the sperm/oocyte decision in Caenorhabditis elegans. Annu Rev Cell Dev Biol 23:405–433. https://doi.org/10.1146/annurev.cellbio.23.090506.123326
Kinoshita M, Murata K, Naruse K, Tanaka M (2012) Medaka: biology, management, and experimental protocols. John Wiley & Sons Ltd, Ames. https://doi.org/10.1002/9780813818849
Kitadate Y, Kobayashi S (2010) Notch and Egfr signaling act antagonistically to regulate germ-line stem cell niche formation in Drosophila male embryonic gonads. Proc Natl Acad Sci 107:14241–14246. https://doi.org/10.1073/pnas.1003462107
La HM, Hobbs RM (2019) Mechanisms regulating mammalian spermatogenesis and fertility recovery following germ cell depletion. Cell Mol Life Sci 76:4071–4102. https://doi.org/10.1007/s00018-019-03201-6
Liao H-F, Chen WSC, Chen Y-H, Kao T-H, Tseng Y-T, Lee C-Y, Chiu Y-C, Lee P-L, Lin Q-J, Ching Y-H, Hata K, Cheng WTK, Tsai M-H, Sasaki H, Ho H-N, Wu S-C, Huang Y-H, Yen P, Lin S-P (2014) DNMT3L promotes quiescence in postnatal spermatogonial progenitor cells. Development 141:2402 LP – 2413. https://doi.org/10.1242/dev.105130
Losick VP, Morris LX, Fox DT, Spradling A (2011) Drosophila stem cell niches: a decade of discovery suggests a unified view of stem cell regulation. Dev Cell 21:159–171. https://doi.org/10.1016/j.devcel.2011.06.018
Lue Y-H, Sinha Hikim AP, Swerdloff RS, Im P, Taing KS, Bui T, Leung A, Wang C (1999) Single exposure to heat induces stage-specific germ cell apoptosis in rats: role of intratesticular testosterone on stage specificity. Endocrinology 140:1709–1717
Majhi SK, Hattori RS, Rahman SM, Suzuki T, Strüssmann CA (2009) Experimentally induced depletion of germ cells in sub-adult Patagonian pejerrey (Odontesthes hatcheri). Theriogenology 71:1162–1172. https://doi.org/10.1016/j.theriogenology.2008.12.008
Mäkelä J-A, Hobbs RM (2019) Molecular regulation of spermatogonial stem cell renewal and differentiation. Reproduction 158:R169–R187. https://doi.org/10.1530/REP-18-0476
McIntyre DC, Nance J (2020) Niche cell wrapping ensures primordial germ cell quiescence and protection from intercellular cannibalism. Curr Biol 30:708-714.e4. https://doi.org/10.1016/j.cub.2019.12.021
Mieusset R, Bujan L (1995) Testicular heating and its possible contributions to male infertility: a review. Int J Androl 18:169–184. https://doi.org/10.1111/j.1365-2605.1995.tb00408.x
Mommsen TP, Vijayan MM, Moon TW (1999) Cortisol in teleosts: dynamics, mechanisms of action, and metabolic regulation. Rev Fish Biol Fish. https://doi.org/10.1023/A:1008924418720
Morinaga C, Saito D, Nakamura S, Sasaki T, Asakawa S, Shimizu N, Mitani H, Furutani-Seiki M, Tanaka M, Kondoh H (2007) The hotei mutation of medaka in the anti-Müllerian hormone receptor causes the dysregulation of germ cell and sexual development. Proc Natl Acad Sci USA 104:9691–9696. https://doi.org/10.1073/pnas.0611379104
Murta D, Batista M, Silva E, Trindade A, Henrique D, Duarte A, Lopes-da-Costa L (2013) Dynamics of notch pathway expression during mouse testis post-natal development and along the spermatogenic cycle. PLoS One 8:e72767
Nakamura S, Watakabe I, Nishimura T, Toyoda A, Taniguchi Y, Tanaka M (2012) Analysis of medaka sox9 orthologue reveals a conserved role in germ cell maintenance. PLoS One 7. https://doi.org/10.1371/journal.pone.0029982
Nishimura T, Nakamura S, Tanaka M (2016) A structurally and functionally common unit in testes and ovaries of medaka (Oryzias latipes), a teleost fish. Sex Dev 10:159–165. https://doi.org/10.1159/000447313
Nóbrega RH, Greebe CD, van de Kant H, Bogerd J, de França LR, Schulz RW (2010) Spermatogonial stem cell niche and spermatogonial stem cell transplantation in zebrafish. PLoS One 5:1–16. https://doi.org/10.1371/journal.pone.0012808
Nowell CS, Radtke F (2017) Notch as a tumour suppressor. Nat Rev Cancer 17:145–159. https://doi.org/10.1038/nrc.2016.145
Oatley JM, Avarbock MR, Telaranta AI, Fearon DT, Brinster RL (2006) Identifying genes important for spermatogonial stem cell self-renewal and survival. Proc Natl Acad Sci 103:9524 LP – 9529. https://doi.org/10.1073/pnas.0603332103
Ohta H, Aizawa S, Nishimune Y (2003) Functional analysis of the p53 gene in apoptosis induced by heat stress or loss of stem cell factor signaling in mouse male germ cells. 2254:2249–2254. https://doi.org/10.1095/biolreprod.102.014779
Ozaki Y, Higuchi M, Miura C, Yamaguchi S, Tozawa Y, Miura T (2006) Roles of 11beta-hydroxysteroid dehydrogenase in fish spermatogenesis. Endocrinology 147:5139–5146. https://doi.org/10.1210/en.2006-0391
Pfaffl MW (2001) A new mathematical model for relative quantification in real-time RT-PCR. Nucleic Acids Res 29:e45. https://doi.org/10.1093/nar/29.9.e45
Rockett JC, Mapp FL, Garges JB, Luft JC, Mori C, Dix DJ (2001) Effects of hyperthermia on spermatogenesis, apoptosis, gene expression, and fertility in adult male mice. Biol Reprod 65:229–239. https://doi.org/10.1095/biolreprod65.1.229
Rodríguez-Marí A, Cañestro C, BreMiller RA, Nguyen-Johnson A, Asakawa K, Kawakami K, Postlethwait JH (2010) Sex reversal in zebrafish fancl mutants is caused by Tp53-mediated germ cell apoptosis. PLoS Genet 6:1–14. https://doi.org/10.1371/journal.pgen.1001034
Sada A, Hasegawa K, Pin PH, Saga Y (2012) NANOS2 acts downstream of glial cell line-derived neurotrophic factor signaling to suppress differentiation of spermatogonial stem cells. Stem Cells 30:280–291. https://doi.org/10.1002/stem.790
Sarida M, Hattori RS, Zhang Y, Yamamoto Y, Strüssmann CA (2019) Spatiotemporal correlations between amh and cyp19a1a transcript expression and apoptosis during gonadal sex differentiation of Pejerrey, Odontesthes bonariensis. Sex Dev 13:99–108. https://doi.org/10.1159/000498997
Schulz RW, de França LR, Lareyre JJ, LeGac F, Chiarini-Garcia H, Nobrega RH, Miura T (2010) Spermatogenesis in fish. Gen Comp Endocrinol 165:390–411. https://doi.org/10.1016/j.ygcen.2009.02.013
Selim KM, Shinomiya A, Otake H, Hamaguchi S, Sakaizumi M (2009) Effects of high temperature on sex differentiation and germ cell population in medaka, Oryzias latipes. Aquaculture 289:340–349. https://doi.org/10.1016/j.aquaculture.2008.12.019
Selkoe D, Kopan R (2003) Notch and presenilin: regulated intramembrane proteolysis links development and degeneration. Annu Rev Neurosci 26:565–597. https://doi.org/10.1146/annurev.neuro.26.041002.131334
Setchell BP (1998) The Parkes Lecture Heat and the testis. Reproduction 114:179–194. https://doi.org/10.1530/jrf.0.1140179
Wabik A, Jones PH (2015) Switching roles: the functional plasticity of adult tissue stem cells. EMBO J 34:1164–1179. https://doi.org/10.15252/embj.201490386
Wang D, Manali D, Wang T, Bhat N, Hong N, Li Z, Wang L, Yan Y, Liu R, Hong Y (2011) Identification of pluripotency genes in the fish medaka. Int J Biol Sci 7:440
Xavier T, Defalco T, Capel B, Hofmann M (2013) Constitutive activation of NOTCH1 signaling in Sertoli cells causes gonocyte exit from quiescence. Dev Biol 377:188–201
Xia W (2019) γ-Secretase and its modulators: twenty years and beyond. Neurosci Lett 701:162–169. https://doi.org/10.1016/j.neulet.2019.02.011
Yuan L, Liu J-G, Zhao J, Brundell E, Daneholt B, Höög C (2000) The murine SCP3 gene is required for synaptonemal complex assembly, chromosome synapsis, and male fertility. Mol Cell 5:73–83. https://doi.org/10.1016/S1097-2765(00)80404-9
Zetterberg H, Campbell WA, Yang HW, Xia W (2006) The cytosolic loop of the γ-secretase component presenilin enhancer 2 protects zebrafish embryos from apoptosis. J Biol Chem 281:11933–11939. https://doi.org/10.1074/jbc.M512521200
Zhang Z, Hu J (2007) Development and validation of endogenous reference genes for expression profiling of medaka (Oryzias latipes) exposed to endocrine disrupting chemicals by quantitative real-time RT-PCR. Toxicol Sci 95:356–368. https://doi.org/10.1093/toxsci/kfl161
Zhang Z, Nadeau P, Song W, Donoviel D, Yuan M, Bernstein A, Yankner BA (2000) Presenilins are required for γ-secretase cleavage of β-APP and transmembrane cleavage of Notch-1. Nat Cell Biol 2:463–465. https://doi.org/10.1038/35017108
Zhou Z, Kawabe H, Suzuki A, Shinmyozu K, Saga Y (2017) NEDD4 controls spermatogonial stem cell homeostasis and stress response by regulating messenger ribonucleoprotein complexes. Nat Commun 8:15662. https://doi.org/10.1038/ncomms15662
Acknowledgements
We thank Tech. Gabriela C. López for helping with histological preparations. We also thank Dr. Tania Rodriguez for technical support and Tech. Javier Herdman (INTECH) for fish handling. We are grateful to NBRP Medaka (https://shigen.nig.ac.jp/medaka/) for providing HNI (Strain ID: MT835).
Funding
This work was supported by the Agencia Nacional de Promoción Científica y Tecnológica Grant 2501/15 and 1875/18 (to JIF) and CONICET-FAPESP International Cooperation Grant D 2979/16 and 14/50790–0 (to JIF and RSH). ODMA was supported by a PhD scholarship from the National Research Council (CONICET). AFB was supported by an undergraduate scholarship from Universidad Nacional de San Martín (UNSAM). JIF is a member of the Research Scientist Career at the CONICET.
Author information
Authors and Affiliations
Contributions
ODMA: conceptualization, methodology, analysis; AFB: methodology and visualization; RSH: funding acquisition, conceptualization, writing — reviewing and editing, JIF: funding acquisition, conceptualization, investigation, supervision; writing — original draft preparation, writing — reviewing and editing.
Corresponding author
Ethics declarations
Ethics approval
This animal experiment was reviewed and approved by the Institutional Committee for the Care and Use of Animals for Experimental Purposes—Universidad Nacional de San Martín, Argentina (approval number: CICUAE-UNSAM 17/21).
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Table Supplement 1
Primers sequences, ENSEMBL and NCBI accession numbers and the respective references of each gene. (DOCX 26 kb)

Supplementary Fig. S1
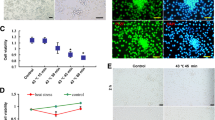
Cycle threshold (Ct) of reference genes rpl7 and ef-1a. Cycle threshold of rpl7 and ef-1a of testis tissue from in vivo (A) and explant incubated at NT and HT (B). Cts quantifications were compared by one-way analysis of variance (ANOVA), followed by Tukey´s multiple comparisons test, for comparing the mean of each column with the mean of every other column. p-values are indicated when differences between treatment at the same sampling day differ significantly (P<0.05). NS, not statistically significant. (PNG 348 kb)

Supplementary Fig. S2
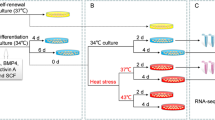
Time and dose optimization for the ex vivo experiment. Time exposure curve of p53 transcript abundance in testis explants incubated at control (NT – 25º C, light grey) and high (HT – 33º C, dark grey) temperature, with sampling at 0, 3, 12 and 24 hours (A). DAPT doses response: Transcript abundance levels of hes1 (Notch pathway cis target genes) in adult testis explants incubated during 24 hours at 0, 6.25, 12.5, 25, 50 and 100 μM of DAPT (γ-secretase inhibitor) (B). Transcript abundance quantification was performed using the geometric mean method and p53 and hes1 values were normalized against geometric mean of reference genes rpl7 and ef-1a. p-values are indicated when transcript abundance between treatment at the same sampling day differs significantly (P<0.05). (PNG 342 kb)

Supplementary Fig. S3
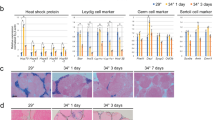
Graphical abstract. The activation of Notch pathway in somatic cell of the testis induces the inhibition of spermatogenesis under heat temperature exposure. SGa: type A spermatogonia; SGb: type b spermatogonia; Sc: spermatocyte; Sd: spermatid; Sz: spermatozoa. (PNG 647 kb)
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Moreno Acosta, O.D., Boan, A.F., Hattori, R.S. et al. Notch pathway is required for protection against heat stress in spermatogonial stem cells in medaka. Fish Physiol Biochem 49, 487–500 (2023). https://doi.org/10.1007/s10695-023-01200-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10695-023-01200-w