Abstract
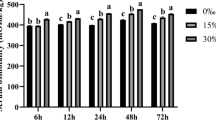
The present work aims to evaluate the tolerance, osmoregulation, metabolism, and antioxidant ability of saline water immersed grass goldfish (Carassius auratus) during the recovery in freshwater. Grass goldfish (38.15 ± 5.48g) acclimated in freshwater were immersed by salinities (0‰, 20‰ and 30‰) for different time durations (10, 20, 30 and 60 min); and the physiological responses were measured during freshwater recovery. The blood osmolalities were not significantly different at any group fish, while whereas the decline of Na+ concentration and the ratio of Na+/Cl- as well as the rise of Cl- concentration was observed in saline treated fish. Soon after freshwater recovery, the transcription of NKA-α and NKA-β mRNA in gills of salinity 20 immersed fish elevated significantly and then decreased, whereas no obvious changes were detected in salinity 30 treated fish. Till 24h post freshwater recovery, gill Na+/K+-ATPase activities in saline treated fish were lower than control group except for the fish immersed by salinity 20 for 10–30 min. At 24h of recovery, cortisol levels in salinity 20 immersed fish were lower than salinity 30 treated fish, but remained higher than control. As for serum lactic acid, fish treated by salinity 20 for 10 or 20 min did not show any fluctuation. However, higher lactic acid contents were detected in all other five salinity treated groups during recovery. Generally, at 24 h of recovery, salinity 20 treated fish exhibited higher SOD and CAT activities than fish immersed by salinity 30. In summary, grass goldfish could survive by immersion in salinity 20 less than 60 min or salinity 30 less than 30min, even though immersion by salinity 20 could minimize the negative effects.












Similar content being viewed by others
Data availability
The data used to support the findings of this study are available from the first author upon request.
Code availability
Not applicable.
References
Árnason T, Magnadóttir B, Björnsson B, Steinarsson A, Björnsson BT (2013) Effects of salinity and temperature on growth, plasma ions, cortisol and immune parameters of juvenile Atlantic cod (Gadus morhua). Aquaculture 380:70–79. https://doi.org/10.1016/j.aquaculture.2012.11.036
Azarias G, Perreten H, Lengacher S, Poburko D, Demaurex N, Magistretti PJ, Chatton J-Y (2011) Glutamate transport decreases mitochondrial pH and modulates oxidative metabolism in Astrocytes. J Neurosci 31:3550–3559. https://doi.org/10.1523/JNEUROSCI.4378-10.2011
Bal A, Panda F, Pati SG, Das K, Agrawal PK, Paital B (2021) Modulation of physiological oxidative stress and antioxidant status by abiotic factors especially salinity in aquatic organisms. Comp Biochem Phys C 241:108971. https://doi.org/10.1016/j.cbpc.2020.108971
Barton BA, Iwama GK (1991) Physiological changes in fish from stress in aquaculture with emphasis on the response and effects of corticosteroids. Annu Rev Fish Dis 1:3–26. https://doi.org/10.1016/0959-8030(91)90019-G
Barton BA, Zitzow RE (1995) Physiological responses of juvenile walleyes to handling stress with recovery in saline water. Prog Fish Cult 57:267–276
Bath RN, Eddy FB (1979) Salt and water balance in rainbow trout (Salmo Gairdneri) rapidly transferred from fresh water to sea water. J Exp Biol 83:193–202. https://doi.org/10.1242/jeb.83.1.193
Blondeau-Bidet E, Bossus M, Maugars G, Farcy E, Lignot J-H, Lorin-Nebel C (2016) Molecular characterization and expression of Na+/K+-ATPase α1 isoforms in the European sea bass Dicentrarchus labrax osmoregulatory tissues following salinity transfer. Fish Physiol Biochem 42:1647–1664. https://doi.org/10.1007/s10695-016-0247-x
Bonga SEW (1997) The stress response in fish. Physiol Rev 77:591–625. https://doi.org/10.1152/physrev.1997.77.3.591
Burg MB, Ferraris JD, Dmitrieva NI (2007) Cellular Response to Hyperosmotic Stresses. Physiol Rev 87:1441–1474. https://doi.org/10.1152/physrev.00056.2006
Bystriansky JS, Richards JG, Schulte PM, Ballantyne JS (2006) Reciprocal expression of gill Na+/K+-ATPase alpha-subunit isoforms alpha1a and alpha1b during seawater acclimation of three salmonid fishes that vary in their salinity tolerance. J Exp Biol 209:1848–1858. https://doi.org/10.1242/jeb.02188
Carneiro PCF, Urbinati EC (2002) Salt as a stress response mitigator of matrinxa, Brycon cephalus (Gunther), during transport. Aquac Res 32:297–304. https://doi.org/10.1046/j.1365-2109.2001.00558.x
Dang Z, Balm PH, Flik G, Wendelaar Bonga SE, Lock RA (2000) Cortisol increases Na(+)/K(+)-ATPase density in plasma membranes of gill chloride cells in the freshwater tilapia Oreochromis mossambicus. J Exp Biol 203:2349–2355. https://doi.org/10.1242/jeb.203.15.2349
Dawood MAO, Alkafafy M, Sewilam H (2022) The antioxidant responses of gills, intestines and livers and blood immunity of common carp (Cyprinus carpio) exposed to salinity and temperature stressors. Fish Physiol Biochem 48:397–408. https://doi.org/10.1007/s10695-022-01052-w
Dytham C (2011) Choosing and using statistics: a biologist's guide. John Wiley&Sons
Evans DH, Piermarini PM, Choe KP (2005) The Multifunctional fish gill: dominant site of gas exchange, osmoregulation, acid-base regulation, and excretion of nitrogenous waste. Physiol Rev 85:97–177. https://doi.org/10.1152/physrev.00050.2003
Fielder DS, Allan GL, Pepperall D, Pankhurst PM (2007) The effects of changes in salinity on osmoregulation and chloride cell morphology of juvenile Australian snapper, Pagrus auratus. Aquaculture 272:656–666. https://doi.org/10.1016/j.aquaculture.2007.08.043
Franzon R, Lamers ML, Stefanello FM, Wannmacher CMD, Wajner M, Wyse ATS (2003) Evidence that oxidative stress is involved in the inhibitory effect of proline on Na+,K+-ATPase activity in synaptic plasma membrane of rat hippocampus. Int J Dev Neurosci 21:303–307. https://doi.org/10.1016/S0736-5748(03)00076-5
Goswami SV, Parwez I, Sundararaj BI (1983) Some aspects of osmoregulation in a stenohaline freshwater catfish, Heteropneustes fossilis (Bloch), in different salinities. J Fish Biol 23:475–487. https://doi.org/10.1111/j.1095-8649.1983.tb02928.x
Hadiroseyani Y, Harti LS, Nuryati S (2009) Control of Ectoparasitic Monogenean Infestation on GIFT Tilapia (Oreochromis sp.) using Salt Addition. J Akuak Indones 8:135. https://doi.org/10.19027/jai.8.135-145
Hwang P-P, Lee T-H (2007) New insights into fish ion regulation and mitochondrion-rich cells. Comp Biochem Phys A 148:479–497. https://doi.org/10.1016/j.cbpa.2007.06.416
Imanpoor MR, Najafi E, Kabir M (2012) Effects of different salinity and temperatures on the growth, survival, haematocrit and blood biochemistry of Goldfish (Carassius auratus). Aquac Res 43:332–338. https://doi.org/10.1111/j.1365-2109.2011.02832.x
Imsland AK, Gunnarsson S, Foss A, Stefansson SO (2003) Gill Na+, K+-ATPase activity, plasma chloride and osmolality in juvenile turbot (Scophthalmus maximus) reared at different temperatures and salinities. Aquaculture 218:671–683. https://doi.org/10.1016/S0044-8486(02)00423-4
Jamme I, Petit E, Divoux D, Alain G, Maixent JM, Nouvelot A (1995) Modulation of mouse cerebral Na+, K+-ATPase activity by oxygen free radicals. NeuroReport 7:333–337. https://doi.org/10.1097/00001756-199512290-00080
Kim J-H, Jeong E-H, Jeon Y-H, Kim SK, Hur YB (2021) Salinity-mediated changes in hematological parameters, stress, antioxidant responses, and acetylcholinesterase of juvenile olive flounders (Paralichthys olivaceus). Environ Toxicol Phar 83:103597. https://doi.org/10.1016/j.etap.2021.103597
Kim J-H et al (2017) Growth performance, oxidative stress, and non-specific immune responses in juvenile sablefish, Anoplopoma fimbria, by changes of water temperature and salinity. Fish Physiol Biochem 43:1421–1431. https://doi.org/10.1007/s10695-017-0382-z
Küçük S (2013) The effects of salinity on growth of goldfish, Carassius auratus and crucian carp, Carassius carassius. Afr J Biotechnol 12:2082–2087. https://doi.org/10.5897/AJB12.430
Kültz D, Bastrop R, Jürss K, Siebers D (1992) Mitochondria-rich (MR) cells and the activities of the Na+K+-ATPase and carbonic anhydrase in the gill and opercular epithelium of Oreochromis mossambicus adapted to various salinities. Comp Biochem Phys B 102:293–301. https://doi.org/10.1016/0305-0491(92)90125-B
Lawson E, Alake S (2011) Salinity adaptability and tolerance of hatchery reared comet goldfish Carassius auratus (Linnaeus 1758). Int J Zoo Res 7:68–76. https://doi.org/10.3923/ijzr.2011.68.76
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2–ΔΔCT method. Methods 25:402–408. https://doi.org/10.1006/meth.2001.1262
Livingstone DR (2001) Contaminant-stimulated reactive oxygen species production and oxidative damage in aquatic organisms. Mar Pollut Bull 42:656–666. https://doi.org/10.1016/S0025-326X(01)00060-1
Luz RK, Martínez-Álvarez RM, De Pedro N, Delgado MJ (2008) Growth, food intake regulation and metabolic adaptations in goldfish (Carassius auratus) exposed to different salinities. Aquaculture 276:171–178. https://doi.org/10.1016/j.aquaculture.2008.01.042
Ma Z, Zheng P, Guo H, Jiang S, Qin JG, Zhang D, Liu X (2016) Salinity regulates antioxidant enzyme and Na+K+-ATPase activities of juvenile golden pompano Trachinotus ovatus (Linnaeus 1758). Aquac Res 47:1481–1487. https://doi.org/10.1111/are.12606
Martínez-Alvarez RM, Hidalgo MC, Domezain A, Morales AE, García-Gallego M, Sanz A (2002) Physiological changes of sturgeon Acipenser naccarii caused by increasing environmental salinity. J Exp Biol 205:3699–3706. https://doi.org/10.1242/jeb.205.23.3699
Mirghaed AT, Ghelichpour M (2019) Effects of anesthesia and salt treatment on stress responses, and immunological and hydromineral characteristics of common carp (Cyprinus carpio, Linnaeus, 1758) subjected to transportation. Aquaculture 501:1–6. https://doi.org/10.1016/j.aquaculture.2018.11.008
Mozanzadeh MT et al (2021) The effect of salinity on growth performance, digestive and antioxidant enzymes, humoral immunity and stress indices in two euryhaline fish species: Yellowfin seabream (Acanthopagrus latus) and Asian seabass (Lates calcarifer). Aquaculture 534:736329. https://doi.org/10.1016/j.aquaculture.2020.736329
Noga EJ (1996) Fish Disease: Diagnosis and Treatment. Mosby, St Louis, MO, USA
Ostrowski AD, Watanabe WO, Montgomery FP, Rezek TC, Shafer TH, Morris JA (2011) Effects of salinity and temperature on the growth, survival, whole body osmolality, and expression of Na+/K+ ATPase mRNA in red porgy (Pagrus pagrus) larvae. Aquaculture 314:193–201. https://doi.org/10.1016/j.aquaculture.2011.02.010
Piermarini PM, Evans DH (2000) Effects of environmental salinity on Na(+)/K(+)-ATPase in the gills and rectal gland of a euryhaline elasmobranch (Dasyatis sabina). J Exp Biol 203:2957–2966. https://doi.org/10.1242/jeb.203.19.2957
Porthé-Nibelle J, Lahlou B (1974) Plasma concentrations of cortisol in hypophysectomized and sodium chloride-adapted goldfish (Carassius auratus L.). J Endocrinol 63:377–387. https://doi.org/10.1677/joe.0.0630377
Qiang J, Ren H-T, Xu P, He J, Li R-W (2012) Synergistic effects of water temperature and salinity on the growth and liver antioxidant enzyme activities of juvenile GIFT Oreochromis niloticus. Chin J Appl Ecol 23:255–263
Richards JG, Semple JW, Bystriansky JS, Schulte PM (2003) Na+/K+-ATPase α-isoform switching in gills of rainbow trout (Oncorhynchus mykiss) during salinity transfer. J Exp Biol 206:4475–4486. https://doi.org/10.1242/jeb.00701
Robergs RA, Ghiasvand F, Parker D (2004) Biochemistry of exercise-induced metabolic acidosis. Am J Physiol Reg I 287:R502–R516. https://doi.org/10.1152/ajpregu.00114.2004
Robertson L, Thomas P, Arnold CR, Trant JM (1987) Plasma cortisol and secondary stress responses of red drum to handling, transport, rearing density, and a disease outbreak. Prog Fish Cult 49:1–12
Sarma K, Prabakaran K, Krishnan P, George G, Kumar A (2013) Response of a freshwater air-breathing fish, Clarias batrachus to salinity stress: An experimental case for their farming in brackishwater areas in Andaman. India Aquacult Int 21. https://doi.org/10.1007/s10499-012-9544-2
Schofield PJ, Brown ME, Fuller PF (2006) Salinity tolerance of goldfish Carassius auratus L., a non-native fish in the United States. Fla Sci 69:258–268
Sinha AK et al (2015) Nutritional status as the key modulator of antioxidant responses induced by high environmental ammonia and salinity stress in European sea bass (Dicentrarchus labrax). PloS One 10:e0135091. https://doi.org/10.1371/journal.pone.0135091
Soltani M, Burke CM (1994) Responses of fish-pathogenic Cytophaga/Flexibacter-like bacteria (CFLB) to environmental conditions B. Eur Assoc Fish Pat 14:185–187
Stewart HA, Noakes DLG, Cogliati KM, Peterson JT, Iversen MH, Schreck CB (2016) Salinity effects on plasma ion levels, cortisol, and osmolality in Chinook salmon following lethal sampling. Comp Biochem Phys A 192:38–43. https://doi.org/10.1016/j.cbpa.2015.11.011
Takahashi H, Takahashi A, Sakamoto T (2006) In vivo effects of thyroid hormone, corticosteroids and prolactin on cell proliferation and apoptosis in the anterior intestine of the euryhaline mudskipper (Periophthalmus modestus). Life Sci 79:1873–1880. https://doi.org/10.1016/j.lfs.2006.06.021
Tsuzuki MY (2001) Physiological responses during stress and subsequent recovery at different salinities in adult pejerrey Odontesthes bonariensis. Aquaculture 200:349–362. https://doi.org/10.1016/S0044-8486(00)00573-1
Tweedley J, Beatty S, Lymbery A, Ash A, Keleher J (2014) Salty Goldfish? Goldfish can use wetlands as ‘bridges’ to invade new rivers. In: Paper presented at the 10th Annual Wetland Management Conference, Perth, Western Australia
Urbina MA, Glover CN (2015) Effect of salinity on osmoregulation, metabolism and nitrogen excretion in the amphidromous fish, inanga (Galaxias maculatus). J Exp Mar Biol Ecol 473:7–15. https://doi.org/10.1016/j.jembe.2015.07.014
Varsamos S, Nebel C, Charmantier G (2005) Ontogeny of osmoregulation in postembryonic fish: A review. Comp Biochem Phys A 141:401–429. https://doi.org/10.1016/j.cbpb.2005.01.013
Vazzana M, Vizzini A, Sanfratello MA, Celi M, Salerno G, Parrinello N (2010) Differential expression of two glucocorticoid receptors in seabass (teleost fish) head kidney after exogeneous cortisol inoculation. Comp Biochem Phys A 157:49–54. https://doi.org/10.1016/j.cbpa.2010.05.003
Verdegem MCJ, Hilbrands AD, Boon JH (1997) Influence of salinity and dietary composition on blood parameter values of hybrid red tilapia, Oreochromis niloticus (Linnaeus) ×O. mossambicus (Peters). Aquac Res 28:453–459. https://doi.org/10.1046/j.1365-2109.1997.00880.x
Vizzini A, Vazzana M, Cammarata M, Parrinello N (2007) Peritoneal cavity phagocytes from the teleost sea bass express a glucocorticoid receptor (cloned and sequenced) involved in genomic modulation of the in vitro chemiluminescence response to zymosan. Gen Comp Endocr 150:114–123. https://doi.org/10.1016/j.ygcen.2006.07.016
Wen X, Chu P, Xu J, Wei X, Fu D, Wang T, Yin S (2021) Combined effects of low temperature and salinity on the immune response, antioxidant capacity and lipid metabolism in the pufferfish (Takifugu fasciatus). Aquaculture 531:735866. https://doi.org/10.1016/j.aquaculture.2020.735866
Wilson JL (1988) Biochemistry (Stryer, Lubert). J Chem Educ 65(12)
Zhang G et al (2016) Modulated expression and enzymatic activities of Darkbarbel catfish, Pelteobagrus vachelli for oxidative stress induced by acute hypoxia and reoxygenation. Chemosphere 151:271–279. https://doi.org/10.1016/j.chemosphere.2016.02.072
Zhang KF, Zhang Z, Chen Y, Lin P, Wang Y (2007) Antioxidant defense system in animals. Chin J Zool 42:153–160
Zhu H et al (2018) Characterization and expression of Na+/K+-ATPase in gills and kidneys of the Teleost fish Oreochromis mossambicus, Oreochromis urolepis hornorum and their hybrids in response to salinity challenge. Comp Biochem Phys A 224:1–10. https://doi.org/10.1016/j.cbpa.2018.05.017
Funding
This work was supported by Beijing Fishery Innovation Team and China Agriculture Research System of MOF and MARA (CARS-45).
Author information
Authors and Affiliations
Contributions
Xiao-wen Wang: Conceptualization, Project administration, Formal analysis, Data curation, Writing. Hui-juan Li: Formal analysis. Rong Zhang: Formal analysis. Li-li Liu: Formal analysis. Hua Zhu: Resources, Supervision.
Corresponding author
Ethics declarations
Conflicts of interest
The authors declare no conflicts of interests.
Ethical approval
All experiments in the present study were performed in accordance with the standards of the China Council on Animal Care.
Consent to participate
Not applicable.
Consent for publication
All authors provided consent for publication.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wang, X., Li, H., Zhang, R. et al. Effects of saline immersion on the physiological alterations of grass goldfish (Carassius auratus) during subsequent recovery in freshwater. Fish Physiol Biochem 49, 455–470 (2023). https://doi.org/10.1007/s10695-023-01187-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10695-023-01187-4