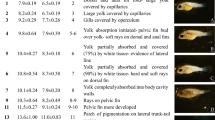
Abstract
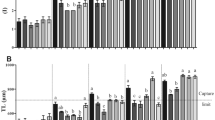
Artemia nauplii are widely used as fish larvae feed due to its beneficial nutritional characteristics for larval development; however, efficient feeding strategies are needed to balance its high costs. Therefore, we evaluated the effects of different densities of Artemia nauplii (100, 250, 500, 750, and 1000 nauplii/post-larvae) on the growth, survival, water quality, and myogenic gene expression of tambaqui (Colossoma macropomum) post-larvae cultivated in a recirculating aquaculture system. After 2 weeks of trial, there was a significant decrease in dissolved oxygen concentration with the increase in nauplii density, but it did not interfere with larval performance and survival. In the first week, larvae fed with fewer than 500 nauplii/post-larvae presented slower growth, while in the second week, larvae fed with 1000 nauplii/post-larvae had the highest final weight and length. Regression analysis suggests that the optimum feeding density of Artemia nauplii during the first week is 411 nauplii/post-larvae, while for the second week, the growth increased proportionally to the feeding densities. The relative expression of the myod, myog, and mstn genes was higher in larvae fed with fewer than 500 nauplii/post-larvae. Although low-growing larvae showed increased expression of myod and myog genes, responsible for muscle hyperplasia and hypertrophy, respectively, mstn expression may have played a significant inhibitory role in larval development. Further research is needed to better determine the effects of the live food on the zootechnical performance and the expression of the myogenic genes in the initial phase of the life cycle of the tambaqui post-larvae.


Similar content being viewed by others
Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
References
Abe HA, Dias JAR, Reis RGA et al (2016) Manejo alimentar e densidade de estocagem na larvicultura do peixe ornamental amazônico Heros severus. Bol Inst Pesca 42:514–522. https://doi.org/10.20950/1678-2305.2016v42n3p514
Alami-Durante H, Cluzeaud M, Duval C et al (2014) Early decrease in dietary protein:energy ratio by fat addition and ontogenetic changes in muscle growth mechanisms of rainbow trout: short-and long-term effects. Br J Nutr 112:674–687. https://doi.org/10.1017/S0007114514001391
Alfei L, Onali A, Spanò L et al (1994) PCNA/cyclin expression and BrdU uptake define proliferating myosatellite cells during hyperplastic muscle growth of fish (Cyprinus carpio L.). Eur J Histochem 38:151–162
Amali AA, Lin CJF, Chen YH et al (2004) Up-regulation of muscle-specific transcription factors during embryonic somitogenesis of zebrafish (Danio rerio) by knock-down of myostatin-1. Dev Dyn 229:847–856. https://doi.org/10.1002/dvdy.10454
Araújo-Lima CARM, Gomes LC (2005) Tambaqui (Colossoma macropomum). In Baldisserotto B, Gomes LC (eds) Espécies nativas para piscicultura no Brasil, 1st edn. Editora UFSM, Santa Maria, pp 175–202
Aride PHR, Roubach R, Val AL (2007) Tolerance response of tambaqui Colossoma macropomum (Cuvier) to water pH. Aquac Res 38:588–594. https://doi.org/10.1111/j.1365-2109.2007.01693.x
Balon EK (1984) Reflections on some decisive events in the early life of fishes. Trans Am Fish Soc 113:178–185. https://doi.org/10.1577/1548-8659(1984)113%3c178:ROSDEI%3e2.0.CO;2
Beux LF, Zaniboni-Filho E (2006) Influência da baixa salinidade na sobrevivência de náuplios de Artemia sp. Bol Inst Pesca 32:73–77
Canada P, Engrola S, Mira S et al (2016) The supplementation of a microdiet with crystalline indispensable amino-acids affects muscle growth and the expression pattern of related genes in Senegalese sole (Solea senegalensis) larvae. Aquaculture 458:158–169. https://doi.org/10.1016/j.aquaculture.2016.03.010
Chen JY, Zeng C, Jerry DR, Cobcroft JM (2020) Recent advances of marine ornamental fish larviculture: broodstock reproduction, live prey and feeding regimes, and comparison between demersal and pelagic spawners. Rev Aquac 12:1518–1541. https://doi.org/10.1111/raq.12394
Conceição LEC, Yúfera M, Makridis P et al (2010) Live feeds for early stages of fish rearing. Aquac Res 41:613–640. https://doi.org/10.1111/j.1365-2109.2009.02242.x
Diemer O, Neu DH, Sary C et al (2012) Artemia sp. na alimentação de larvas de de jundiá (Rhamdia quelen). Ciênc Anim Bras 13:175–179. https://doi.org/10.5216/cab.v13i2.9011
Fabregat TEHP, Damian J, Fialho NS et al (2015) Toxicidade aguda ao sal comum e larvicultura intensiva do jundiá Rhamdia quelen em água salobra. Arq Bras Med Vet Zootec 67:547–554. https://doi.org/10.1590/1678-7660
Fuentes EN, Pino K, Navarro C et al (2013) Transient inactivation of myostatin induces muscle hypertrophy and overcompensatory growth in zebrafish via inactivation of the SMAD signaling pathway. J Biotechnol 168:295–302. https://doi.org/10.1016/j.jbiotec.2013.10.028
Fuentes EN, Valdés JA, Molina A, Björnsson BT (2013) Regulation of skeletal muscle growth in fish by the growth hormone – Insulin-like growth factor system. Gen Comp Endocrinol 192:136–148. https://doi.org/10.1016/J.YGCEN.2013.06.009
Hayashi C (2014) Desenvolvimento inicial das larvas de dourado Salminus brasiliensis (Cuvier, 1816); suplementados com plâncton, artêmia e rações, em cultivos experimentais. Periódico Eletrônico Fórum Ambiental da Alta Paulista 10:137–150. https://doi.org/10.17271/198008271032014847
Instituto Brasileiro de Geografia e Estatística – IBGE (2021) Pesquisa da pecuária municipal: Tabela 3940 Produção da aquicultura, por tipo de produto. SIDRA Banco de Tabelas Estatísticas. https://sidra.ibge.gov.br/tabela/3940. Accessed 18 July 2022
Jha P, Sarkar K, Barat S (2004) Effect of different application rates of cowdung and poultry excreta on water quality and growth of ornamental carp, Cyprinus carpio vr. koi, in concrete tanks. Turk J Fish Aquat Sci 4:17–22
Jiménez-Amilburu V, Salmerón C, Codina M et al (2013) Insulin-like growth factors effects on the expression of myogenic regulatory factors in gilthead sea bream muscle cells. Gen Comp Endocrinol 188:151–158. https://doi.org/10.1016/J.YGCEN.2013.02.033
Jobling M (1981) The influences of feeding on the metabolic rate of fishes: a short review. J Fish Biol 18:385–400. https://doi.org/10.1111/j.1095-8649.1981.tb03780.x
Jobling M (1993) Bioenergetics: feed intake and energy partitioning. In: Rankin JC, Jensen FB (eds) Fish Ecophysiol. Chapman & Hall Fish and Fisheries Series, 9. Springer, Dordrecht, pp 1– 44. https://doi.org/10.1007/978-94-011-2304-4_1
Johnston IA (2006) Environment and plasticity of myogenesis in teleost fish. J Exp Biol 209:2249–2264. https://doi.org/10.1242/jeb.02153
Jomori RK, Luz RK, Portella MC (2012) Effect of salinity on larval rearing of Pacu, Piaractus mesopotamicus, a freshwater species. J World Aquac Soc 43:423–432. https://doi.org/10.1111/j.1749-7345.2012.00570.x
Jomori RK, Luz RK, Takata R et al (2013) Água levemente salinizada aumenta a eficiência da larvicultura de peixes neotropicais. Pesqui Agropecu Bras 48:809–815. https://doi.org/10.1590/S0100-204X2013000800001
Kerdchuen N, Legendre M (1994) Larval rearing of an African catfish, Heterobranchus longifilis, (Teleostei, Clariidae): a comparison between natural and artificial diet. Aquat Living Resour 7:247–253. https://doi.org/10.1051/alr:1994027
Kuradomi RY, Figueiredo MA, Lanes CFC et al (2011) GH overexpression causes muscle hypertrophy independent from local IGF-I in a zebrafish transgenic model. Transgenic Res 20:513–521. https://doi.org/10.1007/S11248-010-9429-Y/FIGURES/7
Lanes CFC, Bolla S, Fernandes JMO et al (2012) Nucleotide enrichment of live feed: a promising protocol for rearing of Atlantic Cod Gadus morhua larvae. Mar Biotechnol 14:544–558. https://doi.org/10.1007/s10126-012-9458-z
Leitão NJ, Pai-Silva MD, Almeida FLA, Portella MC (2011) The influence of initial feeding on muscle development and growth in pacu Piaractus mesopotamicus larvae. Aquaculture 315:78–85. https://doi.org/10.1016/j.aquaculture.2011.01.006
Lima JC, Lansac-Tôha FA, Mantovano T et al (2021) Post-larval Colossoma macropomum (Characiformes, Serrasalmidae) show better performance in excavated than concrete tanks under different feeding strategies. Acta Sci Biol Sci 43:e52054. https://doi.org/10.4025/actascibiolsci.v43i1.52054
Luz RK, Portella MC (2005) Freqüência alimentar na larvicultura do trairão (Hoplias lacerdae). Rev Bras Zootec 34:1442–1448. https://doi.org/10.1590/S1516-35982005000500003
Luz RK, Portella MC (2015) Effect of prey concentrations and feed training on production of Hoplias lacerdae juvenile. An Acad Bras Cienc 87:1125–1132. https://doi.org/10.1590/0001-3765201520140412
Luz RK, Zaniboni-Filho E (2001) Utilização de diferentes dietas na primeira alimentação do mandi-amarelo (Pimelodus maculatus, Lacépéde). Acta Sci Biol Sci 23:483–489
Luz RK, Santos JCE, Pedreira MM, Teixeira EA (2011) Effect of water flow rate and feed training on “pacamã” (Siluriforme: Pseudopimelodidae) juvenile production. Arq Bras Med Vet Zootec 63:973–979. https://doi.org/10.1590/S0102-09352011000400024
Ma Z, Qin JG, Hutchinson W, Chen BN (2013) Food consumption and selectivity by larval yellowtail kingfish Seriola lalandi cultured at different live feed densities. Aquac Nutr 19:523–534. https://doi.org/10.1111/anu.12004
Megeney LA, Rudnicki MA (1995) Determination versus differentiation and the MyoD family of transcription factors. Biochem Cell Biol 73:723–732. https://doi.org/10.1139/o95-080
Mommsen TP (2001) Paradigms of growth in fish. Comp Biochem Physiol B Biochem Mol Biol 129:207–219. https://doi.org/10.1016/S1096-4959(01)00312-8
Mun SH, You JH, Oh HJ et al (2019) Expression patterns of growth related genes in juvenile red spotted grouper (Epinephelus akaara) with different growth performance after size grading. Dev Reprod 23:35. https://doi.org/10.12717/DR.2019.23.1.035
Patruno M, Radaelli G, Mascarello F, Candia Carnevali MD (1998) Muscle growth in response to changing demands of functions in the teleost Sparus aurata (L.) during development from hatching to juvenile. Anat Embryol (Berl) 198:487–504. https://doi.org/10.1007/s004290050199
Pedreira MM, Sipaúba-Tavares LH (2001) Effect of light green and dark brown colored tanks on survival rates and development of tambaqui larvae, Colossoma macropomum (Osteichthyes, Serrasalmidae). Acta Sci 23:521–525
Puvanendran V, Brown JA (1999) Foraging, growth and survival of Atlantic cod larvae reared in different prey concentrations. Aquaculture 175:77–92. https://doi.org/10.1016/S0044-8486(99)00023-X
Rabe J, Brown JA (2000) A pulse feeding strategy for rearing larval fish: an experiment with yellowtail flounder. Aquaculture 191:289–302. https://doi.org/10.1016/S0044-8486(00)00434-8
Reis RGA, Alves PCJ, Abe HA et al (2021) Feed management and stocking density for larviculture of the Amazon ornamental fish L333 king tiger pleco Hypancistrus sp. (Siluriformes: Loricariidae). Aquac Res 52:1995–2003. https://doi.org/10.1111/are.15047
Rodrigues RV, Freitas LS, Robaldo RB, Sampaio LA (2012) Crescimento e sobrevivência de juvenis do linguado Paralichthys orbignyanus: efeitos do enriquecimento da Artemia sp. com N-3 HUFA. Atlântica 34:121–127. https://doi.org/10.5088/atl.2012.34.2.121
Rønnestad I, Thorsen A, Finn RN (1999) Fish larval nutrition: a review of recent advances in the roles of amino acids. Aquaculture 177:201–216. https://doi.org/10.1016/S0044-8486(99)00082-4
Rowlerson A, Veggetti A (2001) Cellular mechanisms of post-embryonic muscle growth in aquaculture species. In: Johnston IA (ed) Muscle Development and Growth. Fish Physiology, 18. Academic Press, San Diego, pp103–140. https://doi.org/10.1016/s1546-5098(01)18006-4
Rudnicki MA, Jaenisch R (1995) The MyoD family of transcription factors and skeletal myogenesis. BioEssays 17:203–209. https://doi.org/10.1002/bies.950170306
Sáez-Arteaga A, Wu Y, Silva-Marrero JI et al (2022) Gene markers of dietary macronutrient composition and growth in the skeletal muscle of gilthead sea bream (Sparus aurata). Aquaculture 555:738221. https://doi.org/10.1016/j.aquaculture.2022.738221
Santos JCE, Luz RK (2009) Effect of salinity and prey concentrations on Pseudoplatystoma corruscans, Prochilodus costatus and Lophiosilurus alexandri larviculture. Aquaculture 287:324–328. https://doi.org/10.1016/j.aquaculture.2008.10.014
Santos SS, Lopes JP, Santos-Neto MA, Santos LS (2007) Larvicultura do tambaqui em diferentes densidades de estocagem. Rev Bras Eng Pesca 2:18–25
Santos JCE, Correia ES, Luz RK (2015) Effect of daily Artemia nauplii concentrations during juvenile production of Lophiosilurus alexandri. Bolet Inst Pesca 41:771–776. https://doi.org/10.20950/1678-2305.2015v41nep771
Santos FAC, Costa Julio GS, Luz RK (2021) Stocking density in Colossoma macropomum larviculture, a freshwater fish, in recirculating aquaculture system. Aquac Res 52:1185–1191. https://doi.org/10.1111/are.14976
Santos FAC, Julio GSC, Batista FS et al (2022) High stocking densities in the larviculture of Colossoma macropomum in a recirculating aquaculture system: performance, survival and economic viability. Aquaculture 552:738016. https://doi.org/10.1016/j.aquaculture.2022.738016
Santos JCE, Pedreira MM, Luz RK (2012) The effects of stocking density, prey concentration and feeding on Rhinelepis aspera (Spix e Agassiz, 1829) (Pisces: Loricariidae) larviculture. Acta Sci Biol Sci 34:133–139. https://doi.org/10.4025/actascibiolsci.v34i2.8541
Sharma M, Kambadur R, Matthews KG et al (1999) Myostatin, a transforming growth factor-β superfamily member, is expressed in heart muscle and is upregulated in cardiomyocytes after infarct. J Cell Physiol 180:1–9. https://doi.org/10.1002/(SICI)1097-4652(199907)180:1%3c1::AID-JCP1%3e3.0.CO;2-V
Silva P, Valente LMP, Olmedo M et al (2009) Hyperplastic and hypertrophic growth of lateral muscle in blackspot seabream Pagellus bogaraveo from hatching to juvenile. J Fish Biol 74:37–53. https://doi.org/10.1111/j.1095-8649.2008.02122.x
Silva ADR, Santos RB, Bruno AMSS, Soares EC (2013) Cultivo de tambaqui em canais de abastecimento sob diferentes densidades de peixes. Acta Amazon 43:517–523. https://doi.org/10.1590/S0044-59672013000400014
Song F, Ye H, Shi L et al (2022) Characterization and functional analysis of myostatin and myogenin genes involved in temperature variation and starvation stress in Golden pompano, Trachinotus blochii. Comp Biochem Physiol A Mol Integr Physiol 267:111183. https://doi.org/10.1016/j.cbpa.2022.111183
Sorgeloos P, Dhert P, Candreva P (2001) Use of the brine shrimp, Artemia spp., in marine fish larviculture. Aquaculture 200:147–159. https://doi.org/10.1016/S0044-8486(01)00698-6
Sterzelecki FC, Jesus AM, Jorge JLC et al (2022) Açai palm, Euterpe oleracea, seed for aquaponic media and seedling production. Aquac Eng 98:102270. https://doi.org/10.1016/j.aquaeng.2022.102270
Tavares-Dias M, Araújo CSO, Porto SMA, Viana GM, Monteiro PC (2013) Sanidade do tambaqui Colossoma macropomum nas fases de larvicultura e alevinagem. Universidade Nilton Lins, Instituto de Pesquisas da Amazônia, Embrapa Amapá, Manaus
Thomas M, Langley B, Berry C et al (2000) Myostatin, a negative regulator of muscle growth, functions by inhibiting myoblast proliferation. J Biol Chem 275:40235–40243. https://doi.org/10.1074/jbc.M004356200
Timmons MB, Guerdat T, Vinci BJ (2018) Recirculating aquaculture. Ithaca Publishing Company LLC, Ithaca, New York
Treece GD (2000) Artemia production for marine larval fish culture, Publication No. 702. Southern Regional Aquaculture Center, Stoneville.
Van Stappen G (1996) Introduction, biology and ecology of Artemia. In: Lavens P, Sorgeloos P (eds) Manual on the production and use of live food for aquaculture, FAO Fisheries Technical Paper, 361. FAO, Rome, pp 79 – 106
Veggetti A, Mascarello F, Scapolo PA et al (1993) Muscle growth and myosin isoform transitions during development of a small teleost fish, Poecilia reticulata (Peters) (Atheriniformes, Poeciliidae): a histochemical, immunohistochemical, ultrastructural and morphometric study. Anat Embryol (Berl) 187:353–361. https://doi.org/10.1007/BF00185893
Vélez EJ, Lutfi E, Azizi S et al (2017) Understanding fish muscle growth regulation to optimize aquaculture production. Aquaculture 467:28–40. https://doi.org/10.1016/J.AQUACULTURE.2016.07.004
Watabe S (1999) Myogenic regulatory factors and muscle differentiation during ontogeny in fish. J Fish Biol 55:1–18. https://doi.org/10.1111/j.1095-8649.1999.tb01042.x
Zhao H, Xia J, Zhang X et al (2018) Diet affects muscle quality and growth traits of grass carp (Ctenopharyngodon idellus): a comparison between grass and artificial feed. Front Physiol 9:283. https://doi.org/10.3389/fphys.2018.00283
Zhao Y, Jiang Q, Zhou XQ et al (2020) Effect of dietary threonine on growth performance and muscle growth, protein synthesis and antioxidant-related signalling pathways of hybrid catfish Pelteobagrus vachelli♀ × Leiocassis longirostris♂. Br J Nutr 123:121–134. https://doi.org/10.1017/S0007114519002599
Zheng GD, Sun CF, Pu JW et al (2015) Two myostatin genes exhibit divergent and conserved functions in grass carp (Ctenopharyngodon idellus). Gen Comp Endocrinol 214:68–76. https://doi.org/10.1016/j.ygcen.2015.03.008
Zhu X, Li YL, Liu L et al (2016) Molecular characterization of Myf5 and comparative expression patterns of myogenic regulatory factors in Siniperca chuatsi. Gene Expr Patterns 20:1–10. https://doi.org/10.1016/j.gep.2015.10.003
Acknowledgements
We are especially grateful to the professors and technical staff at the LGA, BIOAQUAM, and LSBM for their technical support during the present study.
Funding
This work was supported by the Brazilian Coordination for Higher Education Personnel Training (CAPES) with grant number 88881.200563/2018–01 (PROCAD-AM), grant number 88881.510299/2020–01 (PDPG-Amazônia Legal) and National Council for Scientific and Technological Development (CNPq) with grant number 432209/2018–2 (CNPq/Universal), grant number 314102/2021–3 (CNPq/Productivity in Research).
Author information
Authors and Affiliations
Contributions
Debora Sayumi Doami Melo: Conceptualization, investigation, writing—original draft, visualization. André Luiz Alves de Sá: Writing—review and editing and supervision. Sávio Lucas de Matos Guerreiro: Investigation. Joane Natividade: Investigation. Paola Fabiana Fazzi Gomes: Investigation. Rodrigo Takata: Supervision. Ednaldo da Silva Filho: Resources, writing—review and editing. Glauber David Almeida Palheta: Conceptualization, methodology, and supervision. Nuno Filipe Alves Correia de Melo: Resources. Fabio Carneiro Sterzelecki: Conceptualization, methodology, formal analysis, writing—review and editing, visualization, and project administration. Igor Guerreiro Hamoy: Resources, writing—review and editing, supervision, project administration, funding acquisition.
Corresponding author
Ethics declarations
Ethics approval
The project described here was approved by the Ethics Committee for the Experimental Use of Animals of the Federal Rural University of Amazonia, under protocol number 7509070720.
Consent to participate
All authors agree to participate in this study.
Consent for publication
All authors agree to participate in the publication of this article.
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Melo, D.S.D., de Sá, A.L.A., de Matos Guerreiro, S.L. et al. Growth, survival, and myogenic gene expression in the post-larvae of Colossoma macropomum provisioned with Artemia nauplii. Fish Physiol Biochem 50, 145–155 (2024). https://doi.org/10.1007/s10695-023-01182-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10695-023-01182-9