Abstract
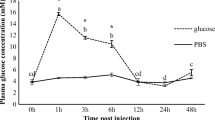
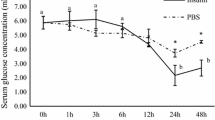
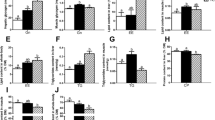
The present study elucidated hepatic molecular and physiological responses of giant gourami to high dietary carbohydrates. Two levels of dietary carbohydrate, normal carbohydrate (34%, NC) and high carbohydrate (53%, HC), were offered to the fish for 60 days. We evaluated the expression of genes that are related to carbohydrate metabolism, lipogenic capacities, amino acid catabolism, Krebs cycle, and energy sensing. In addition, we also observed the digestive enzyme activities, plasma glucose, glycogen content, whole-body composition, and growth performance of the fish. On day 30 after treatment, fish fed with high dietary carbohydrate level has significantly higher expression of gck, pk, hk, and ldh than the NC group (P < 0.05). In contrast, fish in the HC group had lower expression of irs1, igf-1, sdh, fbp, g6pc, gyp, and ampk compared to the NC fish (P < 0.05). On day 60 of the feeding trial, gck and hk expressions were still higher in the HC group (P < 0.05), and gyp, gdh, and ampk became increasingly expressed in the HC group. The increase of dietary carbohydrates resulted in significant increases in amylase and protease activity, plasma glucose, liver glycogen, crude protein, and lipid contents of the fish whole-body (P < 0.05). The high carbohydrate feeding reduced the fish growth rate but increased feed efficiency and did not affect mortality. In conclusion, giant gourami could utilize high carbohydrates due to a high amylase secretion, high modulation of carbohydrate metabolism, and large glucose storage capacity.

Similar content being viewed by others
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Code availability
Not applicable.
References
AOAC (1990) Official methods of analysis of the Association of Official Analytical Chemists, 15th edn. Association of Official Analytical Chemists Inc, Virginia
Belsare S, Dhaker HS, Pawase A et al (2017) Effect of dietary carbohydrate - lipid ratio on growth, body composition and digestive enzyme activities of juvenile goldfish (Carassius auratus). Anim Nutr Feed Technol 17:43–53. https://doi.org/10.5958/0974-181X.2017.00005.1
Bernfeld P (1955) Amylases, alpha and beta. In: Coowick S, Kaplan N (eds) Methods in enzymology I. Academic Press, New York, pp 149–158
Boonanuntanasarn S, Jangprai A, Kumkhong S et al (2018) Adaptation of Nile tilapia (Oreochromis niloticus) to different levels of dietary carbohydrates: new insights from a long term nutritional study. Aquaculture 496:58–65. https://doi.org/10.1016/j.aquaculture.2018.07.011
Boonanuntanasarn S, Kumkhong S, Yoohat K et al (2018) Molecular responses of Nile tilapia Oreochromis niloticus to different levels of dietary carbohydrates. Aquaculture 482:117–123
Borlongan I (1990) Studies on the digestive proteases of the milkfish Chanos chanos. Aquaculture 89:315–325. https://doi.org/10.1016/0044-8486(90)90135-a
Caruso D, Arifin Z, Subagja J et al (2013) Cultured Aquatic Species Information Programme. Osphronemus goramy (Lacepede, 1801). In: FAO Fish. Div. [online]. http://www.fao.org/fishery/culturedspecies/Osphronemus_goramy/en. Accessed 29 Jul 2021
Fountoulaki E, Alexis MN, Nengas I, Venou B (2005) Effect of diet composition on nutrient digestibility and digestive enzyme levels of gilthead sea bream (Sparus aurata L.). Aquac Res 36:1243–1251. https://doi.org/10.1111/j.1365-2109.2005.01232.x
García-Meilán I, Ordóñez-Grande B, Valentín JM et al (2020) High dietary carbohydrate inclusion by both protein and lipid replacement in gilthead sea bream. Changes in digestive and absorptive processes. Aquaculture 520:1–9. https://doi.org/10.1016/j.aquaculture.2020.734977
Halver J, Hardy R (2002) Fish nutrition, 3rd edn. Academic Press, California
Hazen G (1974) Proteinase: method for automatic analysers. In: Bergmeyer HU (ed) Methods of enzymatic analysis, 2nd edn. Academic Press, New York, pp 1000–1005
Kamalam B, Françoise M, Panserat S (2017) Utilisation of dietary carbohydrates in farmed fishes: new insights on influencing factors, biological limitations and future strategies. Aquaculture 467:3–27. https://doi.org/10.1016/j.aquaculture.2016.02.007
Kristanto AH, Slembrouck J, Subagja J et al (2019) Survey on egg and fry production of giant gourami (Osphronemus goramy): current rearing practices and recommendations for future research. J World Aquac Soc 51:119–138. https://doi.org/10.1111/jwas.12647
Li X, Zhu X, Han D et al (2016) Carbohydrate utilization by herbivorous and omnivorous freshwater fish species: a comparative study on gibel carp (Carassius auratus Gibelio. var CAS III) and grass carp (Ctenopharyngodon idellus). Aquac Res 47:128–139. https://doi.org/10.1111/are.12476
Li XF, Xu C, Zhang DD et al (2016) Molecular characterization and expression analysis of glucokinase from herbivorous fish Megalobrama amblycephala subjected to a glucose load after the adaption to dietary carbohydrate levels. Aquaculture 459:89–98. https://doi.org/10.1016/j.aquaculture.2016.03.035
Li X, Zheng S, Wu G (2020) Nutrition and metabolism of glutamate and glutamine in fish. Amino Acids 52:671–691. https://doi.org/10.1007/s00726-020-02851-2
Liu HY, Chen Q, Tan BP et al (2018) Effects of dietary carbohydrate levels on growth, glucose tolerance, glucose homeostasis and GLUT4 gene expression in Tilapia nilotica. Aquac Res 2018:1–11. https://doi.org/10.1111/are.13841
Liu K, Tan B, Zhang W et al (2019) Transcriptome, enzyme activity and histopathology analysis reveal the effects of high level of dietary carbohydrate on glycometabolism in juvenile golden pompano, Trachinotus ovatus. Aquac Res 50:2155–2169. https://doi.org/10.1111/are.14096
Livak K, Schmittgen T (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2-ΔΔCT method. Methods 24:402–408. https://doi.org/10.1006/meth.2001.1262
Lyons CL, Roche HM (2018) Nutritional modulation of AMPK-impact upon metabolic-inflammation. Int J Mol Sci 19:1–17. https://doi.org/10.3390/ijms19103092
Ma HJ, Mou MM, Pu DC et al (2019) Effect of dietary starch level on growth, metabolism enzyme and oxidative status of juvenile largemouth bass, Micropterus salmoides. Aquaculture 498:482–487. https://doi.org/10.1016/j.aquaculture.2018.07.039
Mokoginta I, Takeuchi T, Hadadi A, Dedi J (2004) Different capabilities in utilizing dietary carbohydrate by fingerling and subadult giant gourami Osphronemus goramy. Fish Sci 70:996–1002
Nelson DL, Cox MM (2004) Lehninger principles of biochemistry, 4th edn. W.H. Freeman and Company, New York
Panserat S, Capilla E, Gutierrez J et al (2001) Glucokinase is highly induced and glucose-6-phosphatase poorly repressed in liver of rainbow trout (Oncorhynchus mykiss) by a single meal with glucose. Comp Biochem Physiol - B Biochem Mol Biol 128:275–283. https://doi.org/10.1016/S1096-4959(00)00322-5
Polakof S, Panserat S, Soengas JL, Moon TW (2012) Glucose metabolism in fish: a review. J Comp Physiol B 182:1015–1045. https://doi.org/10.1007/s00360-012-0658-7
Slembrouck J, Arifin OZ, Pouil S et al (2020) Seasonal variation of giant gourami (Osphronemus goramy) spawning activity and egg production in aquaculture ponds. Aquaculture 527:https://doi.org/10.1016/j.aquaculture.2020.735450
Su J, Mei L, Xi L et al (2021) Responses of glycolysis, glycogen accumulation and glucose-induced lipogenesis in grass carp and Chinese longsnout catfish fed high-carbohydrate diet. Aquaculture 533:736146. https://doi.org/10.1016/j.aquaculture.2020.736146
Takahashi LS, Ha N, Pereira MM et al (2018) Carbohydrate tolerance in the fruit-eating fish Piaractus mesopotamicus (Holmberg, 1887). Aquac Res 49:1182–1188. https://doi.org/10.1111/are.13571
Takeuchi T (1988) Laboratory work-chemical evaluation of dietary nutrients. In: Watanabe T (ed) Fish nutrition and mariculture. Department of Aquatic Bioscience. Tokyo University of Fisheries, Kanagawa International Fisheries Training Center, Japan International Cooperation Agency, Tokyo, pp 179–226
Tran-duy A, Smit B, Van DAA, Schrama JW (2008) Effects of dietary starch and energy levels on maximum feed intake, growth and metabolism of Nile tilapia, Oreochromis niloticus. Aquaculture 277:213–219. https://doi.org/10.1016/j.aquaculture.2008.03.004
Wang J, Li X, Han T et al (2016) Effects of different dietary carbohydrate levels on growth, feed utilization and body composition of juvenile grouper Epinephelus akaara. Aquaculture 459:143–147
Wedemeyer G, Yasutake W (1977) Clinical methods for the assessment of the effects of environmental stress on fish health [Technical paper]. US Fish Wildlife Service, Washington
Zhang W, Liu K, Tan B et al (2019) Transcriptome, enzyme activity and histopathology analysis reveal the effects of dietary carbohydrate on glycometabolism in juvenile largemouth bass, Micropterus salmoides. Aquaculture 504:39–51. https://doi.org/10.1016/j.aquaculture.2019.01.030
Zhou C, Ge X, Niu J et al (2015) Effect of dietary carbohydrate levels on growth performance, body composition, intestinal and hepatic enzyme activities, and growth hormone gene expression of juvenile golden pompano, Trachinotus ovatus. Aquaculture 437:390–397. https://doi.org/10.1016/j.aquaculture.2014.12.016
Zhou P, Wang M, Xie F et al (2016) Effects of dietary carbohydrate to lipid ratios on growth performance, digestive enzyme and hepatic carbohydrate metabolic enzyme activities of large yellow croaker (Larimichthys crocea). Aquaculture 452:45–51. https://doi.org/10.1016/j.aquaculture.2015.10.010
Acknowledgements
The authors thank Dr. Wira Wisnu Wardani; Reza Samsudin, MSi; Mr. Dedi Supriadi; Dodi Hermawan, MSi; and Mrs. Lina Mulyani for their excellent technical assistance with this study.
Funding
This study was funded by the Ministry of Research, Technology, and Higher Education of the Republic of Indonesia for the research grant No. 4099/IT3.L1/PN/2020.
Author information
Authors and Affiliations
Contributions
D.N.S.: conceptualization, methodology, formal analysis, investigation, writing—original draft preparation, editing, and validation. H.N.: formal analysis, writing—review, editing, and validation. J.E., M.A.S., and A.A.: conceptualization, data curation, writing—review, editing, and validation.
Corresponding author
Ethics declarations
Ethics approval
All fish were handled in accordance with the Animal Care and Use Committee of IPB University, Indonesia (Grant No. 192–2021 IPB) and the national standard for fish experimentation guidelines (01–6489-2000) of the Republic of Indonesia. All surgery was performed under MS-222 anesthesia, and all efforts were made to minimize suffering.
Consent to participate
All authors consent to participate.
Consent for publication
All authors approved the submitted version of this manuscript.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Sari, D.N., Ekasari, J., Nasrullah, H. et al. High carbohydrate increases amylase, plasma glucose, and gene expression related to glycolysis in giant gourami Osphronemus goramy. Fish Physiol Biochem 48, 1495–1505 (2022). https://doi.org/10.1007/s10695-022-01155-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10695-022-01155-4