Abstract
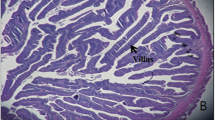
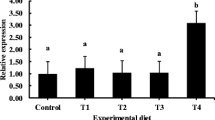
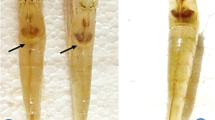
This study examined the effects of feeding fermented tuna by-product (FTBP) on the growth, non-specific immune response, liver and intestinal morphology, and disease resistance of olive flounder Paralichthys olivaceus. Olive flounders (n = 20; 2 g) were randomly assigned into four dietary groups in triplicates. Fish were fed four test diets (50% crude protein; 10% crude lipid) for 10 weeks in which unprocessed tuna by-product (TBP) meal was replaced (on a protein basis) with 0% (FTBP0 as the control diet), 15% (FTBP15), 30% (FTBP30), and 60% (FTBP60) of FTBP protein. Results showed that growth performance, blood parameters, and proximate composition were not influenced by FTBP inclusion. Non-specific immune parameters such as superoxide dismutase activity in the FTBP30 and FTBP60 groups were significantly higher than in the FTBP15 and the control groups, whereas lysozyme and myeloperoxidase activities were not different. Liver histopathology revealed normal architecture in groups fed FTBP0 and FTBP15 diets although mild alterations were noted in the FTBP30- and FTBP60-fed groups. Intestinal villi height and muscular thickness were not significantly altered with FTBP inclusion. Moreover, higher cumulative survival rate was observed in the FTBP60-fed group than fish fed with the other diets following the 13-day challenge with E. tarda. Together, these results demonstrate that fermented tuna by-product meal could be included at 60% in olive flounder diets.



Similar content being viewed by others
Data availability
Data are available from the corresponding author upon reasonable request and with permission of the funding agency.
Code availability
Not applicable.
References
Adams SM, Brown AM, Goede RW (1993) A quantitative health assessment index for rapid evaluation of fish condition in the field. Trans Am Fish Soc 122:63–73. https://doi.org/10.1577/1548-8659(1993)122%3c0063:AQHAIF%3e2.3.CO;2
AOAC (Association of Official Analytical Chemists) (2005) Official methods of analysis, 18th edn. Gaithersburg, MD, Association of Official Analytical Chemists
Ashida T, Okimasu E (2005) Immunostimulatory effects of fermented vegetable product on the non-specific immunity of Japanese flounder Paralicthys olivaceus. Fish Sci 71:257–262. https://doi.org/10.1111/j.1444-2906.2005.00958.x
Batista S, Ramos MA, Cunha S, Barros R, Cristóvão B, Rema P, Pires MA, Valente LMP, Ozório ROA (2015) Immune responses and gut morphology of Senegalese sole (Solea senegalensis, kaup 1858) fed monospecies and multispecies probiotics. Aquac Nutr 21:625–634. org/https://doi.org/10.1111/anu.12191
Bae J, Hamidoghli A, Won S, Choi W, Lim SG, Kim KW, Lee BJ, Hun SW, Bai SC (2020) Evaluation of seven different functional feed additives in a low fish meal diet for olive flounder, Paralichthys olivaceus. Aquaculture 525:735333. https://doi.org/10.1016/j.aquaculture.2020.735333
Choi W, Hamidoghli A, Rivero CJ, Bae J, Kim K-W, Lee B-J, Hur S-W, Han H, Choi YH, Bai SC (2022) Animal and plant proteins as alternative feed ingredients in diets for sub-adult olive flounder Paralichthys olivaceus at farm conditions. Aquac Res 00:1–11. https://doi.org/10.1111/are.15789
Garling DL, Wilson RP (1976) Optimum dietary protein to energy ratio for channel catfish fingerling, Ictalurus punctatus. J Nutr 106:1368–1375. https://doi.org/10.1093/jn/106.9.1368
Güllü K, Acar U, Tezel R, Yozukmaz A (2014) Replacement of fish meal with fish processing by-product silage in diets for the rainbow trout, Oncorhynchus mykiss. Pakistan J Zool 46:1697–1703
Halver JE, Hardy RW (2002) Fish Nutrition. In: Sargent JR, Tocher DR, Bell G (eds) The Lipids, 3rd edn. Academic Press, California, pp 182–246
Hamidoghli A, Won S, Lee S, Lee S, Farris NW, Bai SC (2020) Nutrition and feeding of olive flounder Paralichthys olivaceus: A review. Rev Fish Sci Aquac 28:340–357. https://doi.org/10.1080/23308249.2020.1740166
Han Z, Zhou Y, Zhang X, Yan J, Xiao J, Luo Y, Zheng H, Zhong H (2020) Ghrelin modulates the immune response and increases resistance to Aeromonas hydrophila infection in hybrid tilapia. Fish Shellfish Immunol 98:100–108. https://doi.org/10.1016/j.fsi.2020.01.006
Hultmark D, Steiner H, Rasmuson T, Boman HG (1980) Insect immunity. Purification and properties of three inducible bactericidal proteins from hemolymph of immunized pupae of Hyalophora cecropia. Eur J Biochem 106:7–16. https://doi.org/10.1111/j.1432-1033.1980.tb05991.x
Jeon GH, Kim HS, Myung SH, Cho SH (2014) The effect of the dietary substitution of fishmeal with tuna by-product meal on growth, body composition, plasma chemistry, and amino acid profiles of juvenile Korean rockfish (Sebastes schlegeli). Aquac Nutr 20:753–761. https://doi.org/10.1111/anu.12153
Jung HY, Cho JY, Han KR, Jung HY, Kim IS, Lee G, Kim JK (2017) Biodegradation of the mixture of wild rice (Zizania latifolia) powder and tuna processing waste for use as an additive in fish feed. Korean Fisheries and Aquaculture Society (KOFSAS) Fisheries Conference, Busan, Republic of Korea. Book of Abstracts no. PA-48
Kader MA, Koshio S, Ishikawa M, Yokoyama S, Bulbul M, Honda Y, Mamauag RE, Laining A (2011) Growth, nutrient utilization, oxidative condition, and element composition of juvenile red sea bream Pagrus major fed with fermented soybean meal and scallop by-product blend as fishmeal replacement. Fish Sci 77:119–128. https://doi.org/10.1007/s12562-010-0312-9
Kader MA, Koshio S, Ishikawa M, Yokoyama S, Bulbul M, Nguyen BT, Gao J, Laining A (2012) Can fermented soybean meal and squid by-product blend be used as fishmeal replacements for Japanese flounder (Paralichthys olivaceus)? Aquac Res 43:1427–1438. https://doi.org/10.1111/j.1365-2109.2011.02945.x
Kibenge FSB, Godoy MG, Fast M, Workenhe S, Kibenge MJT (2012) Countermeasures against viral diseases of farmed fish. Antivir Res 95:257–281. https://doi.org/10.1016/j.antiviral.2012.06.003
Kim SS, Galaz GB, Pham MA, Jang JW, Oh DH, Yeo IK, Lee KJ (2009) Effects of dietary supplementation of a meju, fermented soybean meal, and Aspergillus oryzae for juvenile parrot fish (Oplegnathus fasciatus). Asian-Aust J Anim Sci 22:849–856. https://doi.org/10.5713/ajas.2009.80648
Kim SS, Pham MA, Kim KW, Son MH, Lee KJ (2010) Effects of microbial fermentation of soybean on growth performances, phosphorus availability, and antioxidant activity in diets for juvenile olive flounder (Paralichthys olivaceus). Food Sci Biotechnol 19:1605–1610. https://doi.org/10.1007/s10068-010-0227-3
Kim HS, Jung WG, Myung SH, Cho SH, Kim DS (2014a) Substitution effects of fishmeal with tuna byproduct meal in the diet on growth, body composition, plasma chemistry and amino acid profiles of juvenile olive flounder (Paralichthys olivaceus). Aquaculture 431:92–98. https://doi.org/10.1016/j.aquaculture.2014.03.025
Kim HS, Jung WG, Myung SH, Cho SH, Kim DS (2014b) Substitution effects of fishmeal with tuna by-product meal in the diet on growth, body composition, plasma chemistry and amino acid profiles of juvenile olive flounder (Paralichthys olivaceus). Aquaculture 431:92–98
Kim KD, Jang JW, Kim KW, Lee BJ, Hur SW, Han HS (2018) Tuna by-product meal as a dietary protein source replacing fishmeal in juvenile Korean rockfish Sebastes schlegeli. Fish Aquatic Sci 21:29. https://doi.org/10.1186/s41240-018-0107-y
Martínez-Llorens S, Baeza-Ariño R, Nogales-Mérida S, Jover-Cerdá M, Tomás-Vidal A (2012) Carob seed germ meal as a partial substitute in gilthead sea bream (Sparus aurata) diets: Amino acid retention, digestibility, gut and liver histology. Aquaculture 338–341:124–133. https://doi.org/10.1016/j.aquaculture.2012.01.029
Moniruzzaman M, Bae JH, Won SH, Cho SJ, Chang KH, Bai SC (2018) Evaluation of solid-state fermented protein concentrates as a fish meal replacer in the diets of juvenile rainbow trout, Oncorhynchus mykiss. Aquacult Nutr 24:1198–1212. https://doi.org/10.1111/anu.12658
Moniruzzaman M, Damusaru JH, Won SH, Cho SJ, Chang KH, Bai SC (2020) Effects of partial replacement of dietary fish meal by bioprocessed plant protein concentrates on growth performance, hematology, nutrient digestibility and digestive enzyme activities in juvenile Pacific white shrimp, Litopenaeus vannamei. J Sci Food Agric 100:1285–1293. https://doi.org/10.1002/jsfa.10141
Oncul FO, Aya FA, Hamidoghli A, Won S, Lee G, Han KR, Bai SC (2019) Effects of the dietary fermented tuna by-product meal on growth, blood parameters, nonspecific immune response, and disease resistance in juvenile olive flounder, Paralichthys olivaceus. J World Aquac Soc 50:65–77. https://doi.org/10.1111/jwas.12535
Pham HD, Siddik MAB, Ngo MV, Le HM, Rahman MA (2021) Effects of tuna hydrolysate supplementation on growth performance, nutrient utilization and physiological response of snubnose pompano (Trachinotus blochii) fed high poultry by-product meal diets. Aquaculture Reports 21:100875
Quade MJ, Roth JA (1997) A rapid, direct assay to measure degranulation of bovine neutrophil primary granules. Vet Immunol Immunopathol 58:239–248. https://doi.org/10.1016/s0165-2427(97)00048-2
Sakai M (1999) Current research status of fish immunostimulants. Aquaculture 172:63–92. https://doi.org/10.1016/S0044-8486(98)00436-0
Seong M, Lee S, Lee S, Song Y, Bae J, Chang K, Bai SC (2018) The effects of different levels of dietary fermented plant-based protein concentrate on growth, hematology, and non-specific immune responses in juvenile olive flounder, Paralicthys olivaceus. Aquaculture 483:196–202. https://doi.org/10.1016/j.aquaculture.2017.10.023
Siddik MAB, Howieson J, Ilham I, Fotedar R (2018a) Growth, biochemical response and liver health of barramundi, Lates calcarifer fed fermented and nonfermented tuna hydrolysate as fishmeal protein replacement ingredients. PeerJ 6:e4870. https://doi.org/10.7717/peerj.4870
Siddik MAB, Howieson J, Patridge GJ, Fotedar R, Gholipourkanani H (2018b) Dietary tuna hydrolysate modulates growth performance, immune response, intestinal morphology and resistance to Streptococcus iniae in juvenile barramundi. Lates Calcarifer Sci Rep 8:15942. https://doi.org/10.1038/s41598-018-34182-4
Siddik MAB, Howieson J, Fotedar R (2019) Beneficial effects of tuna hydrolysate in poultry by-product meal diets on growth, immune response, intestinal health and disease resistance to Vibrio harvei in barramundi, Lates calcarifer. Fish Shellfish Immunol 89:61–70. https://doi.org/10.1016/j.fsi.2019.03.042
Sogbesan A, Ugwumba A (2008) Nutritional evaluation of termite (Macrotermes subhyalinus) meal as animal protein supplements in the diets of Heterobranchus longifilis (Valenciennes, 1840) fingerlings. Turk J Fish Aquat Sci 8:149–157
Uyan O, Koshio S, Teshima S, Ishikawa M, Thu M, Alam MS, Michael FR (2006) Growth and phosphorus loading by partially replacing fishmeal with tuna muscle by-product powder in the diet of juvenile Japanese flounder, Paralichthys olivaceus. Aquaculture 257:437–445. https://doi.org/10.1016/j.aquaculture.2006.02.060
Won S, Lee S, Hong J, Park JK, Kim S, Bai SC (2017) Evaluation of dietary natural mineral materials as an antibiotic replacer on growth performance, non-specific immune response and disease resistance in rainbow trout, Oncorhynchus mykiss. Aquac Res 48:4735–4747. https://doi.org/10.1111/are.13295
Zeitler MH, Kirchgessner M, Schwarz FJ (1984) Effects of different protein and energy supplies on carcass composition of carp (Cyprinus carpio L). Aquaculture 36:37–48. https://doi.org/10.1016/0044-8486(84)90052-8
Acknowledgements
The assistance of Feeds and Foods Nutrition Research Center (FFNRC) staff is gratefully appreciated. MM would like to acknowledge the National Research Foundation of Korea (NRF) for providing the postdoctoral research fellowship under the Brain Pool Program (Grant No. 2019H1D3A1A01101555) funded by the Ministry of Science, ICT, and Future Planning.
Funding
This study was supported by the National Research Foundation of Korea through the Framework of International Cooperation Program (NRF-2016K2A9A1A09913758). The study was done as part of the Postdoctoral Research Fellowship of F. A. A.
Author information
Authors and Affiliations
Contributions
FAA and SCB designed the study; FAA, SW, and AH collected and analyzed data, and interpreted results; GSP performed the histological examination of liver and intestine; FAA wrote the manuscript; FAA, MM, TM, and SCB reviewed the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval
Handling of fish specimens were conducted in accordance with the approved protocols of the Animal Care and Use Committee of Pukyong National University.
Consent to participate
All authors consent the participation.
Consent for publication
All authors consent the publication.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Aya, F.A., Moniruzzaman, M., Pagador, G.S. et al. Evaluation of dietary fermented tuna by-product meal as partial replacement for unprocessed tuna by-product meal in fishmeal-based diets for juvenile olive flounder Paralichthys olivaceus. Fish Physiol Biochem 48, 1507–1519 (2022). https://doi.org/10.1007/s10695-022-01141-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10695-022-01141-w