Abstract
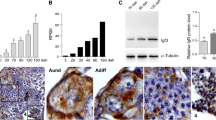
Insulin-like growth factor 3 plays an important role in gonad development in teleost fish. Previous studies found that igf3 was specifically expressed in gonads of silver pomfret (Pampus argenteus). Unlike in other fish, IGF3 is a membrane protein in silver pomfret, and its specific role in gonads is unclear. Herein, we explored the importance of IGF3 in oogenesis and spermatogenesis in silver pomfret by analyzing gene expression and cellular localization. During follicular development, igf3 was detected in ovaries at both mRNA and protein levels during the critical stages of vitellogenesis (IV–VI). Localization analysis detected igf3 mRNA and protein in somatic cells, including theca and granulosa cells around oocytes. Similar to cathepsin L and cathepsin K, igf3 was consistently expressed in ovaries during vitellogenesis, suggesting that it might play a key role in vitellogenesis of oocytes. During spermatogenesis, igf3 mRNA and protein levels were high in stages II, IV, and V, similar to sycp3 and dmc1, and the highest igf3 mRNA and protein levels were reached in stage VI. Furthermore, igf3 mRNA and protein were detected in spermatogonia, spermatocytes, spermatids, and surrounding Sertoli cells, but not in spermatozoon, indicating that IGF3 might be involved in differentiation and meiosis of spermatogonia.





Similar content being viewed by others
Data availability
Available on reasonable request.
Code availability
Not applicable.
References
Andrea B, Manfred A, Ricardo B (2007) Synaptonemal complex protein SYCP3: conserved polymerization properties among vertebrates. Biochim Biophys Acta 1774(5):595–602. https://doi.org/10.1016/j.bbapap.2007.03.008
Chourasia TK, Joy KP (2008) Estrogen-2/4-hydroxylase activity is stimulated during germinal vesicle breakdown induced by hCG, IGF-1, GH and insulin in the catfish Heteropneustes fossilis. Gen Comp Endocrinol 155(2):413–421. https://doi.org/10.1016/j.ygcen.2007.07.005
Diego S, van der Henk JG, Diego C, Bogerd J, Schulz RW (2017) Follicle-stimulating hormone regulates igfbp gene expression directly or via downstream effectors to modulate Igf3 effects on zebrafish spermatogenesis. Front Endocrinol 8:328. https://doi.org/10.3389/fendo.2017.00328
Dilip M, Dola M, Utpal S, Sudipta P, Bhattacharyya SP (2006) In vitro effects of insulin-like growth factors and insulin on oocyte maturation and maturation-inducing steroid production in ovarian follicles of common carp, Cyprinus carpio. Comp Biochem Physiol a: Mol Integr Physiol 144(1):63–77. https://doi.org/10.1016/j.cbpa.2006.01.012
Dinda R, Angka MB, Junhwan, Akihiro T (2020)Molecular cloning of insulin-like growth factor 3 (igf3) and its expression in the tissues of a female damselfish, Chrysiptera cyanea, in relation to seasonal and food-manipulated reproduction.Gen Comp Endocrinol 295. https://doi.org/10.1016/j.ygcen.2020.113479
Duan C, Ren H, Gao S (2010) Insulin-like growth factors (IGFs), IGF receptors, and IGF-binding proteins: roles in skeletal muscle growth and differentiation. Gen Comp Endocrinol 167(3):344–351. https://doi.org/10.1016/j.ygcen.2010.04.009
Eduarda MS, Mariann R-W, Charles RT (2001) Follicle-stimulating hormone and its alpha and beta subunits in rainbow trout (Oncorhynchus mykiss): purification, characterization, development of specific radioimmunoassays, and their seasonal plasma and pituitary concentrations in females. Biol Reprod 65(1):288–294. https://doi.org/10.1095/biolreprod65.1.288
Edward SH, William TF, Adam JL (2021) Molecular and morphological sex differentiation in sablefish (Anoplopoma fimbria), a marine teleost with XX/XY sex determination. Gene 764. https://doi.org/10.1016/j.gene.2020.145093
El-Roeiy A, Chen X, Roberts VJ, LeRoith D, Roberts CT, Yen SS (1993) Expression of insulin-like growth factor-I (IGF-I) and IGF-II and the IGF-I, IGF-II, and insulin receptor genes and localization of the gene products in the human ovary. J Clin Endocrinol Metab 77(5):1411–1418. https://doi.org/10.1210/jcem.77.5.8077342
Funes V, Asensio E, Ponce M, Infante C, Cañavate JP, Manchado M (2006) Insulin-like growth factors I and II in the sole Solea senegalensis: cDNA cloning and quantitation of gene expression in tissues and during larval development. Gen Comp Endocrinol 149(2):166–172. https://doi.org/10.1016/j.ygcen.2006.05.017
Gac FL, Loir M, Bail P-YL, Ollitrault M (1996) Insulin-like growth factor (IGF-I) mRNA and IGF-I receptor in trout testis and in isolated spermatogenic and Sertoli cells. Wiley Subscription Services Inc, A Wiley Company 44(1):23–35. https://doi.org/10.1002/(SICI)1098-2795(199605)44:1%3c23::AID-MRD3%3e3.0.CO;2-V
Giorgi B, Helena DC, Jean-rançois B, Helmut S, Manfred R (2006) Differential expression of IGF-I mRNA and peptide in the male and female gonad during early development of a bony fish, the tilapia Oreochromis niloticus. Gen Comp Endocrinol 146(3):204–210. https://doi.org/10.1016/j.ygcen.2005.11.008
Gregory MW, Craig VS (2000) Effects of insulin-like growth factor-I on in vitro final oocyte maturation and ovarian steroidogenesis in striped bass, Morone saxatilis. Biol Reprod 63(4):1049–1057. https://doi.org/10.1095/biolreprod63.4.1049
Gu W, Yang Y, Ning C, Wang Y, Hu J, Zhang M, Kuang S, Sun Y, Li Y, Zhang Y, Sun J, Ying D, Xu S (2021) Identification and characteristics of insulin-like growth factor system in the brain, liver, and gonad during development of a seasonal breeding teleost, Pampus argenteus. Gen Comp Endocrinol 300:113645–113655. https://doi.org/10.1016/j.ygcen.2020.113645
Hirohiko K, Makito K, Yoshihisa H, Katsumi A (1994) Insulin and insulin-like growth factors I and II induce final maturation of oocytes of red seabream, Pagrus major, in vitro. Gen Comp Endocrinol 95(2):293–300. https://doi.org/10.1006/gcen.1994.1126
Hirohiko K, Shunsuke M, Hiroshi K (1995) Immunocytochemical localization of IGF-I in the ovary of the red seabream, Pagrus major. Gen Compar Endocrinol 99(3):307–315
Hirohiko K, Koichiro G, Koichi O, Hideki T (2003) Effects of luteinizing hormone and follicle-stimulating hormone and insulin-like growth factor-I on aromatase activity and P450 aromatase gene expression in the ovarian follicles of red seabream, Pagrus major. Biol Reprod 68(5):1562–1568. https://doi.org/10.1095/biolreprod.102.008219
Johnl G, Steven DT (1988) Energy allocation in mammalian reproduction. Integr Comp Biol 28(3):863–875. https://doi.org/10.1006/gcen.1995.1114
Jordi V, Francesc P (2008) Stage-specific gene expression during fish spermatogenesis as determined by laser-capture microdissection and quantitative-PCR in sea bass (Dicentrarchus labrax) gonads. Biol Reprod 79(4):738–747. https://doi.org/10.1095/biolreprod.108.069708
Kestemont P, Cooremans J, Abi-Ayad A, Mélard C (1999) Cathepsin L in eggs and larvae of perch Perca fluviatilis: variations with developmental stage and spawning period. Fish Physiol Biochem 21(1):59–64. https://doi.org/10.1023/A:1007714314072
Kwon J, Francisco P, Clive R, Tyler CR (2001) Molecular characterization of putative yolk processing enzymes and their expression during oogenesis and embryogenesis in rainbow trout (Oncorhynchus mykiss). Biol Reprod 65(6):1701–1709. https://doi.org/10.1095/biolreprod65.6.1701
Laurence AMD, André A, Alexandra IF, Adelino VMC (2012) Reference genes to quantify gene expression during oogenesis in a teleost fish. Gene 506(1):69–75. https://doi.org/10.1016/j.gene.2012.06.047
Li W, Ma H (2006) Double-stranded DNA breaks and gene functions in recombination and meiosis. Cell Res 16(5):402–412. https://doi.org/10.1016/j.synthmet.2009.08.054
Li J, Liu Z, Wang D, Cheng C (2011) Insulin-like growth factor 3 is involved in oocyte maturation in zebrafish1. Biol Reprod 84(3):476–486. https://doi.org/10.1095/biolreprod.110.086363
Li M, Wu F, Gu Y, Wang T, Wang H, Yang S, Sun Y, Zhou L, Huang X, Jiao B, Cheng CHK, Wang D (2012) Insulin-like growth factor 3 regulates expression of genes encoding steroidogenic enzymes and key transcription factors in the Nile tilapia gonad1. Biol Reprod 86(5):163–173. https://doi.org/10.1095/biolreprod.111.096248
Li J, Niu C, Cheng CHK (2018) Igf3 serves as a mediator of luteinizing hormone in zebrafish ovulation. Biol Reprod 99(6):1235–1243. https://doi.org/10.1093/biolre/ioy143
Li M, Liu X, Dai S, Xiao H, Qi S, Li Y, Zheng Q, Jie M, Cheng CHK, Wang D (2020) Regulation of spermatogenesis and reproductive capacity by Igf3 in tilapia. Cell Mol Life Sci 77(23):4921–4938. https://doi.org/10.1007/s00018-019-03439-0
Lin Q, Chen Y, Hu J, Wang S, Yang X, Shen Z, Zhao H (2016) Cloning and expression pattern analysis of igf3 gene from Pseudosciaena crocea. South Fisher Sci (1):50–58. https://doi.org/10.3969/j.issn.2095-0780.2016.01.008
Lokman PM, Kataraina ANG, Sean LD, Michael A, Graham Y (2007) 11-Ketotestosterone and IGF-I increase the size of previtellogenic oocytes from shortfinned eel, Anguilla australis, in vitro. Reproduction 133(5):955–967. https://doi.org/10.1530/REP-06-0229
Manal RN, Takeshi M, Nobutaka A, Chiemi M, Kohei Y (1999) Recombinant human insulin-like growth factor I stimulates all stages of 11-ketotestosterone-induced spermatogenesis in the Japanese eel, Anguilla japonica, in vitro. Biol Reprod 61(4):944–947. https://doi.org/10.1095/biolreprod61.4.944
Manfred R, Annette S, Rebekka E, Dominique L-C (1997) Insulin-like growth factor I in the teleost Oreochromis mossambicus, the tilapia: gene sequence, tissue expression, and cellular localization. Endocrinology 138(9):3613–3619. https://doi.org/10.1210/endo.138.9.5375
Mercedes F, Joan C (2004) Ovarian cysteine proteinases in the teleost Fundulus heteroclitus: molecular cloning and gene expression during vitellogenesis and oocyte maturation. Mol Reprod Dev 67(3):282–294. https://doi.org/10.1002/mrd.20018
Migaud H, Davie A, Taylor JF (2010) Current knowledge on the photoneuroendocrine regulation of reproduction in temperate fish species. J Fish Biol 76(1):27–68. https://doi.org/10.1111/j.1095-8649.2009.02500.x
Nagahama Y (1994) Endocrine regulation of gametogenesis in fish. Int J Dev Biol 38(2):217–229. https://doi.org/10.1016/0141-8130(94)90045-0
Negatu Z, Hsiao SM, Wallace RA (1998) Effects of insulin-like growth factor-I on final oocyte maturation and steroid production in Fundulus heteroclitus. Fish Physiol Biochem 19(1):13–21. https://doi.org/10.1023/A:1007777926238
Peggy RB, Gerald TS, Ronald WH, Kenneth DCK, Overturf A, Troy LO (2004) The effects of recombinant bovine somatotropin (rbST) on tissue IGF-I, IGF-I receptor, and GH mRNA levels in rainbow trout, Oncorhynchus mykiss. Gen Compar Endocrinol 135(3):324–333. https://doi.org/10.1016/j.ygcen.2003.10.014
Perks CM, Denning-Kendall PA, Gilmour RS, Wathes DC (1995) Localization of messenger ribonucleic acids for insulin-like growth factor I (IGF-I), IGF-II, and the type 1 IGF receptor in the ovine ovary throughout the estrous cycle. Endocrinology 136(12):5266–5273. https://doi.org/10.1210/endo.136.12.7588270
Perks CM, Peters AR, Wathes DC (1999) Follicular and luteal expression of insulin-like growth factors I and II and the type 1 IGF receptor in the bovine ovary. J Reprod Fertil 116(1):157–165. https://doi.org/10.1530/jrf.0.1160157
Radaelli G, Marco P, Lisa M, Bruria F (2003) Expression and cellular localization of insulin-like growth factor-II protein and mRNA in Sparus aurata during development. J Endocrinol 178(2):285–299. https://doi.org/10.1677/joe.0.1780285
Reinecke M, Björnsson BT, Dickhoff WW, McCormick SD (2005) Growth hormone and insulin-like growth factors in fish: where we are and where to go. Gen Comp Endocrinol 142(1–2):20–24. https://doi.org/10.1016/j.ygcen.2005.01.016
Rüdiger WS, Luiz RDF, Jean-Jacques L, Florence L, Helio C-G (2010) Spermatogenesis in fish. Gen Compar Endocrinol 165(3):390–411. https://doi.org/10.1016/j.ygcen.2009.02.013
Safian D, Bogerd J, Schulz RDW (2018) Igf3 activates β-catenin signaling to stimulate spermatogonial differentiation in zebrafish. J Endocrinol 238(3):245–257. https://doi.org/10.1530/JOE-18-0124
Schmid AC, Näf E, Kloas W, Reinecke M (1999) Insulin-like growth factor-I and -II in the ovary of a bony fish, Oreochromis mossambicus, the tilapia: in situ hybridisation, immunohistochemical localisation, northern blot and cDNA sequences. Mol Cell Endocrinol 156(1–2):141–149. https://doi.org/10.1016/S0303-7207(99)00131-8
Sharon NN, GlenVan DK (2010) The role of the insulin-like growth factor (IGF) system in zebrafish (Danio rerio) ovarian development. Gen Comp Endocrinol 168(1):103–110. https://doi.org/10.1016/j.ygcen.2010.04.021
Shunsuke M, Kiyoko Y, Tomomi T, Hiroaki C, Hiroshi K (2008) Identification of two insulin-like growth factor IIs in the Japanese eel, Anguilla japonica: cloning, tissue distribution, and expression after growth hormone treatment and seawater acclimation. Comp Biochem Physiol b: Biochem Mol Biol 149(1):47–57. https://doi.org/10.1016/j.cbpb.2007.08.005
Song F, Wang L, Zhu W, Fu J, Dong J, Dong Z (2016) A novel igf3 gene in common carp (Cyprinus carpio): evidence for its role in regulating gonadal development. Plos One 11(12). https://doi.org/10.1371/journal.pone.0168874
Teresa MM, Duane HK (1991) Effects of dietary energy on ovarian function, estrogen suppression of luteinizing hormone and follicle-stimulating hormone, and competency of the gonadotropin surge. Biol Reprod 45(3):486–492. https://doi.org/10.1095/biolreprod45.3.486
Tokalov SV, Gutzeit HO (2010) Spermatogenesis in testis primary cell cultures of the tilapia (Oreochromis niloticus). Dev Dyn 233(4):1238–1247. https://doi.org/10.1002/dvdy.20379
Valerie P, Elizabetha BM, Yehoshua G, Shu JC, Bruria F (2000) Insulin-like growth factor receptors and their ligands in gonads of a hermaphroditic species, the gilthead seabream (Sparus aurata): expression and cellular localization. Biol Reprod 63(1):229–241. https://doi.org/10.1095/biolreprod63.1.229
Wang D, Jiao B, Hu C, Liu Z, Christopher HK (2008) Discovery of a gonad-specific IGF subtype in teleost. Biochem Biophys Res Commun 367(2):336–341. https://doi.org/10.1016/j.bbrc.2007.12.136
Weber GM, Sullivan CV (2005) Insulin-like growth factor-I induces oocyte maturational competence but not meiotic resumption in white bass (Morone chrysops) follicles in vitro: evidence for rapid evolution of insulin-like growth factor action. Biol Reprod 72(5):1177–1186. https://doi.org/10.1095/biolreprod.104.036251
Weber GM, Sullivan CV (2007) In vitro actions of insulin-like growth factor-I on ovarian follicle maturation in white perch (Morone americana). Gen Comp Endocrinol 151(2):180–187. https://doi.org/10.1016/j.ygcen.2007.01.007
Xie J (2019) Cloning and expression pattern analysis of medaka (Oryzias latipes) insulin-like growth factor igf3. Shanghai Ocean Univ. https://doi.org/10.27314/d.cnki.gsscu.2019.000574
Xue Y (2018) Study on the mechanism of insulin-like growth factor 3 of oocyte maturation in Epinephelus coioides. South China Agricultural University, Guangzhou
Yang H, Chen H, Zhao H, Liu L, Xie Z (2015) Molecular cloning of the insulin-like growth factor 3 and difference in the expression of igf genes in orange-spotted grouper (Epinephelus coioides). Comp Biochem Physiol b: Biochem Mol Biol 186:68–75. https://doi.org/10.1016/j.cbpb.2015.04.005
Yonathan Z, José Antonio M-C, Abigail E, Olivier K (2010) Neuroendocrinology of reproduction in teleost fish. Gen Comp Endocrinol 165(3):438–455. https://doi.org/10.1016/j.ygcen.2009.04.017
Zhou J, Zhang L, Zhang C, Zhang T, Peng S (2018) Study on karyotype of silver pomfret. Marine Fishery 40(1):97–101. https://doi.org/10.13233/j.cnki.mar.fish.2018.01.011
Funding
This work was supported by the Ningbo Natural Science Foundation (grant number 2019A610423); the Ningbo public welfare science and technology planning project (grant number 202002N3035); the Scientific research project of Zhejiang Provincial Department of Education (grant number Y201940867); the Ningbo 2025 Major Project of Science Technology and Innovation (grant number 2021Z003); the Zhejiang Major Science Project (grant number 2019C02059); the Natural Science Foundation of Zhejiang (grant number LY18C190008 and LY18C1900013); the Agriculture Key Special Project of Ningbo (grant number 2015C110003); the Zhejiang University Student Science and Technology Innovation Activity Plan (grant number 2021R405085); and the Natural Science Foundation of China (grant number 31772869, 31872586, and 31872195).
Author information
Authors and Affiliations
Contributions
All authors contributed to the study; YL and YY wrote the first draft; YZ and JH finalized the article; MZ, JS, and XT prepared the materials; YJ and DZ handled the data; and YW, SX, and XY were responsible for funding and project management.
Corresponding authors
Ethics declarations
Ethics approval
All experiments were conducted in accordance with the principles and procedures approved by the Institutional Animal Care and Use Committee of Ningbo University.
Consent to participate
All authors have consented to participate in the project.
Consent for publication
All authors have consented to publish the data in a scientific journal.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Li, Y., Yang, Y., Zhang, Y. et al. Expression and cellular localization of insulin-like growth factor 3 in gonads of the seasonal breeding teleost silver pomfret (Pampus argenteus). Fish Physiol Biochem 48, 1377–1387 (2022). https://doi.org/10.1007/s10695-022-01122-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10695-022-01122-z