Abstract
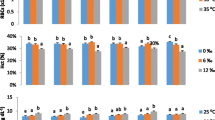
Members of wolf fish family Anarhichadidae have emerged as potential cold-water marine aquaculture species. This study examined growth performance and osmoregulation in juvenile wolf eel (Anarrhichthys ocellatus) held in a series of dilute salinities (30, 14, 9, and 6 ‰) over an 8-week trial. At the conclusion of the growth study, fish were sampled for analysis of gill and intestine enzyme activity, plasma ion content, and muscle moisture. Growth rate remained positive in all salinities throughout the 8-week trial. Specific growth rate was maintained above 3.0% mass day−1 at salinities of 30 and 14 ‰, but was significantly reduced at 9 (2.9% mass day−1) and 6 ‰ (2.0% mass day−1). Muscle water content increased with increasing salinity dilution (77.9% water in 30 ‰; 79.8% water in 6 ‰), and plasma osmolality (~ 320 mOsm kg−1) was maintained in salinities as dilute as 9 ‰ but was significantly lower (~ 280 mOsm kg−1) in the most dilute salinity of 6 ‰. Segmental linear regression analyses revealed that the calculated isosmotic point for wolf eel of ~ 10.6 ‰ was a critical limit for maintaining growth performance and osmoregulatory homeostasis. It is an important finding that fish considered to be a typical marine stenohaline organism could maintain ion and water balance as low as the isosmotic point, and exhibit survival and positive growth rates in salinities as dilute as 6 ‰. This work delivers a fundamental step in the empirical examination of this emerging aquaculture species and provides a model for evaluating osmoregulatory performance of marine stenohaline fishes in low-salinity aquaculture.



Similar content being viewed by others
Availability of data and material
All data is presented in this manuscript.
Code availability
Not applicable.
References
Ahmed N, Turchini GM (2021) Recirculating aquaculture systems (RAS): environmental solution and climate change adaptation. J Clean Prod 297:126604. https://doi.org/10.1016/j.jclepro.2021.126604
Altinok I, Grizzle JM (2001) Effects of brackish water on growth, feed conversion and energy absorption efficiency by juvenile euryhaline and freshwater stenohaline fishes. J Fish Biol 59:1142–1152. https://doi.org/10.1046/j.1365-2761.2001.00306.x
Angel D, Jokumsen A, Lembo G (2019) Aquaculture production systems and environmental interactions. In: Lembo G, Mente E (eds) Organic aquaculture : impacts and future developments. Springer International Publishing, Cham, pp 103–118
Atalah J, Sanchez-Jerez P (2020) Global assessment of ecological risks associated with farmed fish escapes. Glob Ecol Conserv 21:e00842. https://doi.org/10.1016/j.gecco.2019.e00842
Baldisserotto B, Mancera JM, Kapoor BG (2019) Fish osmoregulation. CRC Press, Boca Raton, FL
Beamish RJ, Leask KD, Ivanov OA et al (1999) The ecology, distribution, and abundance of midwater fishes of the Subarctic Pacific gyres. Prog Oceanogr 43:399–442. https://doi.org/10.1016/S0079-6611(99)00017-8
Bœuf G, Payan P (2001) How should salinity influence fish growth? Comp Biochem Physiol C 130:411–423. https://doi.org/10.1016/S1532-0456(01)00268-X
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254. https://doi.org/10.1016/0003-2697(76)90527-3
Brett JR (1979) Environmental factors and growth. In: Hoar WS, Randall DJ, Brett JR (eds) Fish physiology, 8th edn. Academic Press Inc, New York, pp 599–675
Cross SF, Flaherty M, Byrne A (2017) Diversification of aquaculture in North America. In: Harvey B, Soto D, Carolsfeld J, et al. (eds) Planning for aquaculture diversification: the importance of climate change and other drivers, 23rd-25 June edn. FAO Fisheries and Aquaculture Proceedings, Rome
Feeney RF, Lea RN, Dyer S, Gietler S (2007) First record of the wolf-eel, Anarrhichthys ocellatus (Pisces: Anarhichadidae), from Baja California, Mexico. Calif Fish Game 93:52–55
Ferreira FW, Baldisserotto B (2019) Diet and osmoregulation. In: Fish osmoregulation. CRC Press, pp 67–83
Flik G, WendelaarBonga SE, Fenwick JC (1983) Ca2+-dependent phosphatase and ATPase activities in eel gill plasma membranes-I. Identification of Ca2+ activated ATPase activities with non-specific phosphatase-activities. Comp Biochem Physiol - B Biochem Mol Biol 76:745–754
Foss A, Evensen TH, Imsland AK, Oiestad V (2001) Effects of reduced salinities on growth, food conversion efficiency and osmoregulatory status in the spotted wolffish. J Fish Biol 59:416–426. https://doi.org/10.1006/jfbi.2001.1652
Foss A, Imsland AK, Falk-Petersen I-B, Øiestad V (2004) A review of the culture potential of spotted wolffish Anarhichas minor Olafsen. Rev Fish Biol Fish 277–294
Grosell M (2019) Intestinal transport processes in marine fish osmoregulation. In: Fish osmoregulation. CRC Press, pp 333–357
Imsland AK, Foss A, Gunnarsson S et al (2001) The interaction of temperature and salinity on growth and food conversion in juvenile turbot (Scophthalmus maximus). Aquaculture 198:353–367. https://doi.org/10.1016/S0044-8486(01)00507-5
Jobling M (1994) Fish Bioenergetics, 1st edn. Chapman & Hall, London
Kang’ombe J, Brown J (2008) Effect of salinity on growth, feed utilization, and survival of Tilapia rendalli under laboratory conditions. J Appl Aquac 20:256–271. https://doi.org/10.1080/10454430802498229
Langan R (2009) Opportunities and challenges for off-shore farming. Woodhead Publishing Limited
Martin SJ, Mather C, Knott C, Bavington D (2021) ‘Landing’ salmon aquaculture: ecologies, infrastructures and the promise of sustainability. Geoforum 123:47–55. https://doi.org/10.1016/j.geoforum.2021.04.025
Mecklenburg CW, Mecklenburg TA, Thorsteinson LK (2002) Fishes of Alaska. American Fisheries Society, Bethesda
Moksness E, Pavlov DA (2008) Management by life cycle of wolffish, Anarhichas lupus L., a new species for cold-water aquaculture: a technical paper. Aquac Res 27:865–883
Naylor RL, Hardy RW, Bureau DP et al (2009) Feeding aquaculture in an era of finite resources. Proc Natl Acad Sci 106:15103–15110. https://doi.org/10.1073/pnas.0905235106
Riche MA, Pfeiffer TJ, Wills PS et al (2012) Inland marine fish culture in low salinity recirculating aquaculture systems. Bull Fish Res Agen 35:65–75
Sampaio LA, Bianchini A (2002) Salinity effects on osmoregulation and growth of the euryhaline flounder Paralichthys orbignyanus. J Exp Mar Bio Ecol 269:187–196
Schultz E, McCormick SD (2013) Euryhalinity in an evolutionary context. In: McCormick SD, Farrell AP, Brauner CJ (eds) Fish physiology: euryhaline fishes, 1st edn. Academic Press Inc, Amsterdam, pp 477–533
Shaughnessy CA, Breves JP (2021) Molecular mechanisms of Cl- transport in fishes: new insights and their evolutionary context. J Exp Zool 335:207–216. https://doi.org/10.1002/jez.2428
Shaughnessy CA, Baker DW, Brauner CJ et al (2015) Interaction of osmoregulatory and acid–base compensation in white sturgeon (Acipenser transmontanus) during exposure to aquatic hypercarbia and elevated salinity. J Exp Biol 218:2712–2719. https://doi.org/10.1242/jeb.125567
Wiber MG, Mather C, Knott C, Gómez MAL (2021) Regulating the blue economy? Challenges to an effective Canadian aquaculture act. Mar Policy 131:14–17. https://doi.org/10.1016/j.marpol.2021.104700
Zajicek P, Corbin J, Belle S, Rheault R (2021) Refuting marine aquaculture myths, unfounded criticisms, and assumptions. Rev Fish Sci Aquac 0:1–28. https://doi.org/10.1080/23308249.2021.1980767
Zaugg WS (1982) A simplified preparation for adenosine triphosphatase determination in gill tissue. Can J Fish Aquat Sci 39:215–217. https://doi.org/10.1139/f82-027
Zaugg WS, McLain LR (1970) Adenosinetriphosphatase activity in gills of salmonids: seasonal variations and salt water influence in coho salmon, Oncorhynchus kisutch. Comp Biochem Physiol 35:587–596. https://doi.org/10.1016/0010-406X(70)90975-8
Acknowledgements
We would like to acknowledge the technical support of VAMSC research technicians, J. Radloff, L. Garcia, D. Carlson, and B. Hung, as well as DePaul University students K.-M. Dam and B. Sukhera.
Funding
This study received funding support from the Fisheries and Oceans, Aquaculture Collaborative Research and Development Program (S.K.B.) and Island Scallops Ltd, as well as a University Research Council Research Grant and a College of Science and Health Faculty Summer Research Grant from DePaul University (J.S.B.).
Author information
Authors and Affiliations
Contributions
CS performed the experiments, analyzed the data, and wrote the original draft of the manuscript. SB conceived and performed the experiments, facilitated the research, and contributed to the final draft of the manuscript. JB performed the experiments, facilitated the research, and contributed to the final draft of the manuscript. All authors approved the final draft of this manuscript.
Corresponding author
Ethics declarations
Ethics approval
This study was approved by and performed in accordance with the DFO Pacific Region Animal Care Committee (Protocol No: 12–002).
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Shaughnessy, C.A., Balfry, S.K. & Bystriansky, J.S. The isosmotic point as critical salinity limit for growth and osmoregulation, but not survival, in the wolf eel Anarrhichthys ocellatus. Fish Physiol Biochem 48, 471–480 (2022). https://doi.org/10.1007/s10695-022-01064-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10695-022-01064-6