Abstract
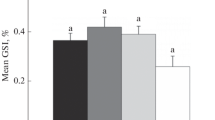
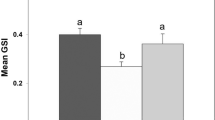
Reproduction in fish is modulated by several factors that include environmental and endocrine components. The aim of this study was to elucidate the effect of aquacultural stressors along the pituitary-testis axis in a continuously breeding cichlid fish Oreochromis mossambicus. The fish (35.05 ± 2.20 g) were divided into three groups (n = 10 in each group, n = 5 in each replicate), namely initial controls (euthanized on the day of initiation of experiment), time-matched controls (kept undisturbed), and stressed fish, which were subjected to different kinds of randomized aquacultural stressors such as handling, chasing, frequent netting, and low water levels, daily for a period of 21 days. Although the gonadosomatic index and the mean numbers of spermatogonia-A and spermatogonia-B did not differ significantly among different experimental groups, significant decrease was observed in the mean numbers of primary spermatocytes, secondary spermatocytes, early spermatids, and late spermatids in fish exposed to stressors compared to those of initial controls and time-matched controls. While the diameter of the seminiferous lobule was significantly lower, the size of the lumen and the serum levels of cortisol were significantly increased in stressed fish compared with initial controls and time-matched controls. Furthermore, weak androgen receptor immunoreactivity was observed in the Sertoli cells of the testis in contrast to the strongly immunoreactive androgen receptors in initial controls and time-matched controls. Concomitant with this, there was a significant decrease in the percent area and the intensity of luteinizing hormone (LH) immunoreactive content in the proximal pars distalis (PPD) region of the pituitary gland in stressed fish compared with initial controls and time-matched controls. Overall, these results suggest that exposure to chronic aquacultural stressors causes suppression of LH synthesis in the pituitary gland concomitant with decreased androgen receptor expression and blockade of recruitment of germline cells at the meiosis stage. This inhibition appears to be mediated through the hypothalamic-pituitary-interrenal axis in the tilapia O. mossambicus.








Similar content being viewed by others
Data availability
Not applicable.
Code availability
Not applicable.
References
Allyn ML, Sheeham RJ, Christopher CK (2001) The effect of capture and transportation stress on white bass semen osmolality and their alleviation via sodium chloride. Trans Am Fish Soc 130:706–711
Balasch JC, Tort L (2019) Netting the stress responses in fish. Front Endocrinol 10:62. https://doi.org/10.3389/fendo.2019.00062
Barata EN, Hubbard PC, Almeida OG, Miranda A, Canário AVM (2007) Male urine signals social rank in the Mozambique tilapia (Oreochromis mossambicus). BMC Biol 5:54. https://doi.org/10.1186/1741-7007-5-54
Barton BA (2002) Stress in fishes: a diversity of responses with particular reference to changes in circulating corticosteroids. Integr Comp Biol 42:517–525
Barton BA, Iwama GK (1991) Physiological changes in fish from stress in aquaculture with emphasis on the response and effects of corticosteroids. Annu Rev Fish Dis 59:13–26
Bayunova L, Barannikova I, Semenkova T (2002) Sturgeon stress reactions in aquaculture. J App Ichthyol 18:397–404
Berlinsky DL, William KV, Hodson RG (1997) Hormone induced spawning of summer flounder Paralichthys dentatus. J World Aquacult Soc 28:79–86
Bhat SK, Ganesh CB (2020) Domperidone treatment attenuates stress-induced suppression of reproduction in viviparous mosquitofish Gambusia affinis. J Fish Biol 96:37–48. https://doi.org/10.1111/jfb.14183
Bobe J, Labbé C (2010) Egg and sperm quality in fish. Gen Comp Endocrinol 165:535–548
Borg B (1994) Androgens in teleost fishes. Comp Biochem Physiol Part C: Pharmacol Toxicol Endocrinol 109:219–245
Butzge AJ, Yoshinaga TT, Acosta ODM, Fernandino JI, Sanches EA, Tabata YA, de Oliveira C, Takahashi NS, Hattori RS (2021) Early warming stress on rainbow trout juveniles impairs male reproduction but contrastingly elicits intergenerational thermotolerance. Sci Rep 11:17053. https://doi.org/10.1038/s41598-021-96514-1
Castranova DA, King WV, Woods LC III (2005) The effects of stress on androgen production, spermiation response and sperm quality in high and low cortisol responsive domesticated male striped bass. Aquaculture 246:413–422
Castro JS, Braz-Mota S, Campos DF, Souza SS, Val AL (2020) High temperature, pH, and hypoxia cause oxidative stress and impair the spermatic performance of the Amazon fish Colossoma macropomum. Front Physiol 11:772. https://doi.org/10.3389/fphys.2020.00772
Campbell PM, Pottinger TG, Sumpter JP (1992) Stress reduces the quality of gametes produced by rainbow trout. Biol Reprod 47:1140–1150
Campbell PM, Pottinger TG, Sumpter JP (1994) Preliminary evidence that chronic confinement stress reduces the quality of gametes produced by brown and rainbow trout. Aquaculture 120:151–169
Carragher JF, Pankhurst NW(1991) Stress and reproduction in a commercially important marine fish Pagrus auratus (Sparidae). In: Scott AP, Sumpter JP, Kime DE, Rolfe MS (eds) Reproductive physiology of fish. Fish symposium, Sheffield, pp 253–255
Chabbi A, Ganesh CB (2012) Stress-induced inhibition of recruitment of ovarian follicles for vitellogenic growth and interruption of spawning cycle in the fish Oreochromis mossambicus. Fish Physiol Biochem 38:1521–1532
Chabbi A, Ganesh CB (2014) Glucocorticoid synthesis inhibitor metyrapone blocks stress-induced suppression along luteinizing hormone secreting cells–ovary axis in the fish Oreochromis mossambicus. J Exp Zool A Ecol Genet Physiol 321:125–134. https://doi.org/10.1002/jez.1842
Chabbi A, Ganesh CB (2015) Evidence for the involvement of dopamine in stress induced suppression of reproduction in the cichlid fish Oreochromis mossambicus. J Neuroendocrinol 27:343–356
Chabbi A, Ganesh CB (2017) Influence of cortisol along the pituitary-ovary axis in the cichlid fish Oreochromis mossambicus. J Appl Ichthyol 33:1146–1152
Clearwater SJ, Crim LW (1998) Gonadotrophin releasing hormone-analogue treatment increase sperm motility, seminal plasma pH and sperm production in yellowtail flounder Pleuronectes ferrugineus. Fish Physiol Biochem 19:349–357
Consten D, Bogerd J, Komen J, Lambert JG, Goos HJ (2001a) Cortisol affects testicular development in male common carp, Cyprinus carpio L., but not via an effect on LH secretion. Comp Biochem Physiol Part B: Biochem Mol Biol 129:671–677. https://doi.org/10.1016/s1096-4959(01)00368-2
Consten D, Lambert JG, Goos HJ (2001b) Cortisol affects testicular development in male common carp, Cyprinus carpio L., but not via an effect on LH secretion. Comp Biochem Physiol Part B: Biochem Mol Biol 129:671–677
Contreras-Sánchez WM, Schreck CB, Fitzpatrick MS, Pereira CB (1998) Effects of stress on the reproductive performance of rainbow trout (Oncorhynchus mykiss). Biol Reprod 58:439–447. https://doi.org/10.1095/biolreprod58.2.439
Fan X, Cui L, Hou T, Xue X, Zhang S, Wang Z (2021) Stress responses of testicular development, inflammatory and apoptotic activities in male zebrafish (Danio rerio) under starvation. Dev Comp Immunol 114:103833. https://doi.org/10.1016/j.dci.2020.103833
Flik G, Klaren PHM, Van den Burg EH, Metz JR, Huising MO (2006) CRF and stress in fish. Gen Comp Endocrinol 146:36–44
García-López A, Bogerd J, Granneman JC, van Dijk W, Trant JM, Taranger GL, Schulz RW (2009) Leydig cells express follicle-stimulating hormone receptors in African catfish. Endocrinology 150:357–365
García-López A, de Jonge H, Nóbrega RH, de Waal PP, van Dijk W, Hemrika W, Taranger GL, Bogerd J, Schulz RW (2010) Studies in zebrafish reveal unusual cellular expression patterns of gonadotropin receptor messenger ribonucleic acids in the testis and unexpected functional differentiation of the gonadotropins. Endocrinology 151:2349–2360
Haddy JA, Pankhurst NW (1999) Stress induced changes in concentrations of plasma sex steroids in black bream. J Fish Biol 55:1304–1316
Iwama GK (2006) Stress in fish. Ann N Y Acad Sci 851:304–310
Jardine JJ, Van Der Kraak GJ, Munkittrick KR (1996) Capture and confinement stress in white sucker exposed to bleached kraft pulp mill effluent. Ecotoxicol Environ Saf 33:287–298
Konkal P, Ganesh CB (2018) Exposure to low or high light intensity affects pituitary-testicular activity in the fish Oreochromis mossambicus. Aquaculture 497:109–116. https://doi.org/10.1016/j.aquaculture.2018.07.043
Konkal P, Ganesh CB (2020) Exposure to hypoxia inhibits pituitary-testicular activity in the fish Oreochromis mossambicus. Aquaculture 515:734552
Lahnsteiner F, Caberlotto S (2012) Motility of gilthead seabream Sparus aurata spermatozoa and its relation to temperature, energy metabolism and oxidative stress. Aquaculture 370–371:76–83. https://doi.org/10.1016/j.aquaculture.2012.09.034
Maruska KP, Fernald RD (2013) Social regulation of male reproductive plasticity in an African cichlid fish. Integr Comp Biol 53:938–950
Mommsen TP, Vijayan MM, Moon TW (1999) Cortisol in teleosts: dynamics, mechanisms of action and metabolic regulation. Rev Fish Biol Fish 9:211–268
Nagahama Y (1994) Endocrine regulation of gametogenesis in fish. Int J Dev Biol 38:217–229
Ogawa S, Soga T, Sakuma Y, Parhar IS (2003) Modulation of GnRH subtypes by social stress and aggressive behavior. Fish Physiol Biochem 28:49–50
Ohta T, Miyake H, Miura C, Kamei H, Aida K, Miura T (2007) Follicle stimulating hormone induces spermatogenesis mediated by androgen production in Japanese eel Anguilla japonica. Biol Reprod 77:970–977
Oliveira RF, Almada VC (1998) Mating tactics and male-male courtship in the lek-breeding cichlid Oreochromis mossambicus. J Fish Biol 52:1115–1129
Pankhurst NW (2001) Stress inhibition of reproductive endocrine processes in a natural population of the spiny damselfish Acanthochromis polyacanthus. Marine Freshw Res 52:753–761
Pankhurst NW (2011) The endocrinology of stress in fish: an environmental perspective. Gen Comp Endocrinol 170:265–275
Pankhurst NW, Van Der Kraak G (1997) Effects of stress on reproduction and growth of fish. In: Iwama GK, Pickering AD, Sumpter JP, Schreck CB (eds) Fish stress and health in aquaculture. Cambridge University Press, Cambridge, pp 73–93
Pickering AD, Pottinger TG, Carragher J, Sumpter JP (1987) The effects of acute and chronic stress on the levels of reproductive hormones in the plasma of mature brown trout, Salmo trutta L. Gen Comp Endocrinol 68:249–259
Pikle RP, Jatiger RM, Ganesh CB (2017) Food-deprivation-induced suppression of pituitary-testicular-axis in the tilapia Oreochromis mossambicus. Int Aquat Res 9:203–213
Rurangwa E, Kime DE, Ollevier F, Nash JP (2004) The measurement of sperm motility and factors affecting sperms and factors affecting sperm quality in cultured fish. Aquaculture 234:1–28
Schreck CB (2010) Stress and fish reproduction: the roles of allostasis and hormesis. Gen Comp Endocrinol 165(3):549–556. https://doi.org/10.1016/j.ygcen.2009.07.004
Schulz RW, de França LR, Lareyre JJ, Le Gac F, Chiarini-Garcia H, Nobrega RH, Miura T (2010) Spermatogenesis in fish. Gen Comp Endocrinol 165:390–411
Stacey NE, Mackenzie DS, Marchant TA, Kyle L, Peter RE (1984) Endocrine changes during natural spawning in the white sucker, Catostomus commersoni: I. Gonadotropin, growth hormone and thyroid hormones. Gen Comp Endocrinol 56:333–348
Suquet M, Omnes MH, Normant Y, Fauvel C (1992) Assessment of sperm concentration and motility in turbot (Scophthalmus maximus). Aquaculture 101:177–185
Tyor AK, Pahwa K (2018) Testicular oxidative stress and cellular deformities in Clarias gariepinus (Burchell) from river Yamuna in Delhi region, India. Bull Environ Contam Toxicol 100:659–664
Vijayalaxmi, Ganesh CB (2017) Distribution of endomorphin-like-immunoreactive neurones in the brain of the cichlid fish Oreochromis mossambicus. J Neuroendocrinol 29:1–17
Wendelaar Bonga SE (1997) The stress response in fish. Physiol Rev 77:591–625
Yu G, Zhang D, Liu W, Wang J, Liu X, Zhou C, Gui J, Xiao W (2018) Zebrafish androgen receptor is required for spermatogenesis and maintenance of ovarian function. Oncotarget 9:24320–24334. https://doi.org/10.18632/oncotarget.24407
Zhu F, Schlupp I, Tiedemann R (2016) Sequence evolution and expression of the androgen receptor and other pathway-related genes in a unisexual fish, the Amazon molly, Poecilia formosa, and its bisexual ancestors. PLoS ONE 11:e0156209
Zohar Y (1980) Dorsal aorta catheterization in rainbow trout (Salmo gairdneri) I. Its validity in the study of blood gonadotropin patterns. Reprod Nutr Dev 20:1811–1823
Zupa R, Fauvel C, Mylonas CC, Pousis C, Santamaria N, Papadaki M, Fakriadis I, Cicirelli V, Mangano S, Passantino L, Lacalandra GM, Corriero A (2017) Rearing in captivity affects spermatogenesis and sperm quality in greater amberjack, Seriola dumerili (Risso, 1810). J Anim Sci 95:4085–4100
Acknowledgements
Infrastructural facility received from Science and Engineering Research Board – Department of Science and Technology (SERB-DST), New Delhi, India is gratefully acknowledged. The first author is grateful to Karnatak University, Dharwad for the award of University Research Scholarship (URS).
Funding
This work was supported by a grant from UGC-SAP (No.18/2015/DSA-I), New Delhi.
Author information
Authors and Affiliations
Contributions
Deepak Shinde conducted the experiment and performed data analysis. Ganesh was responsible for funding acquisition, conceptualization, and review and editing of the manuscript.
Corresponding author
Ethics declarations
Ethics approval
The experimental procedures were approved by an IAEC (No. 639/GO/Re/S/02/CPCSEA).
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Shinde, D., Ganesh, C.B. Chronic exposure to aquacultural stressors affects pituitary-testis axis in the Mozambique tilapia Oreochromis mossambicus. Fish Physiol Biochem 48, 437–448 (2022). https://doi.org/10.1007/s10695-022-01061-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10695-022-01061-9