Abstract
Photoperiod plays a role in controlling the initiation and termination of reproduction in fish. Melatonin is an internal transducer of environmental photoperiod and is involved in regulating reproduction. The present study aimed to examine how melatonin impacts the transcript levels of kisspeptin (kiss1 and kiss2), gonadotropin-releasing hormones (gnrh1), and the β-subunit of gonadotropins (fshβ and lhβ) in the brain of the sapphire devil, a tropical damselfish with long photoperiod preference. Feeding mature females with melatonin-containing pellets inhibited increases in the transcript levels of kiss1, gnrh1, and lhβ within 3 h. Continuous melatonin treatment for 1 week resulted in oocyte regression and downregulation of kiss2, gnrh1, fshβ, and lhβ. When the transcript levels of kiss1 and gnrh1 were measured at 4-h intervals in the brain of sapphire devil, a day–high/night–low fluctuation was observed. The hypothalamic–pituitary–gonadal (HPG) axis may be influenced by melatonin, exerting a negative effect at night because the transcript levels of aralkylamine N-acetyltransferase (aanat2) increased during the scotophase. The expression of aanat2 was higher under short-day than long-day conditions, suggesting that there is a seasonal change in melatonin levels at night. It was concluded that change in photoperiod becomes a key factor for controlling the hormone synthesis in the HPG axis through melatonin.




Similar content being viewed by others
Data availability
All data related to this study are included in the manuscript.
References
Amano M, Iigo M, Ikuta K et al (2000) Roles of melatonin in gonadal maturation of underyearling precocious male masu salmon. Gen Comp Endocrinol 120:190–197. https://doi.org/10.1006/gcen.2000.7547
Amano M, Iigo M, Ikuta K et al (2004a) Disturbance of plasma melatonin profile by high dose melatonin administration inhibits testicular maturation of precocious male masu salmon. Zoolog Sci 21:79–85. https://doi.org/10.2108/0289-0003(2004)21[79:DOPMPB]2.0.CO;2
Amano M, Okubo K, Yamanome T et al (2004b) Changes in brain GnRH mRNA and pituitary GnRH peptide during testicular maturation in barfin flounder. Comp Biochem Physiol Part B Biochem Mol Biol 138:435–443. https://doi.org/10.1016/j.cbpc.2004.05.005
Badruzzaman M, Bapary MAJ, Takemura A (2013) Possible roles of photoperiod and melatonin in reproductive activity via changes in dopaminergic activity in the brain of a tropical damselfish, Chrysiptera cyanea. Gen Comp Endocrinol 194:240–247. https://doi.org/10.1016/j.ygcen.2013.09.012
Bapary MAJ, Fainuulelei P, Takemura A (2009) Environmental control of gonadal development in the tropical damselfish Chrysiptera cyanea. Mar Biol Res 5:462–469. https://doi.org/10.1080/17451000802644722
Bapary MAJ, Takemura A (2010) Effect of temperature and photoperiod on the reproductive condition and performance of a tropical damselfish Chrysiptera cyanea during different phases of the reproductive season. Fish Sci 76:769–776. https://doi.org/10.1007/s12562-010-0272-0
Bapary MAJ, Nurul Amin M, Takemura A (2012) Food availability as a possible determinant for initiation and termination of reproductive activity in the tropical damselfish Chrysiptera cyanea. Mar Biol Res 8:154–162. https://doi.org/10.1080/17451000.2011.605146
Bayarri MJ, Rodríguez L, Zanuy S et al (2004) Effect of photoperiod manipulation on the daily rhythms of melatonin and reproductive hormones in caged European sea bass (Dicentrarchus labrax). Gen Comp Endocrinol 136:72–81
Bromage N, Porter M, Randall C (2001) The environmental regulation of maturation in farmed finfish with special reference to the role of photoperiod and melatonin. Aquaculture 197:63–98. https://doi.org/10.1016/B978-0-444-50913-0.50008-4
Carnevali O, Gioacchini G, Maradonna F et al (2011) Melatonin induces follicle maturation in Danio rerio. PLoS One 6:e19978
Chai K, Liu X, Zhang Y, Lin H (2013) Day-night and reproductive cycle profiles of melatonin receptor, kiss and gnrh expression in orange-spotted grouper (Epinephelus coioides). Mol Reprod Dev 80:535–548. https://doi.org/10.1002/mrd.22191
Coon SL, Begay V, Deurloo D et al (1999) Two arylalkylamine N-acetyltransferase genes mediate melatonin synthesis in fish. J Biol Chem 274:9076–9082
Danilova N, Krupnik VE, Sugden D, Zhdanova IV (2004) Melatonin stimulates cell proliferation in zebrafish embryo and accelerates its development. FASEB J 18:751–753. https://doi.org/10.1096/fj.03-0544fje
Dubocovich ML, Delagrange P, Krause DN et al (2010) International Union of Basic and Clinical Pharmacology. LXXV. Nomenclature, classification, and pharmacology of G protein-coupled melatonin receptors. Pharmacol Rev 62:343–380. https://doi.org/10.1124/pr.110.002832
Falcón J, Besseau L, Fazzari D et al (2003) Melatonin modulates secretion of growth hormone and prolactin by trout pituitary glands and cells in culture. Endocrinology 144:4648–4658. https://doi.org/10.1210/en.2003-0707
Falcón J, Besseau L, Sauzet S, Boeuf G (2007) Melatonin effects on the hypothalamo–pituitary axis in fish. Trends Endocrinol Metab 18:81–88. https://doi.org/10.1016/j.tem.2007.01.002
Fukunaga K, Yamashina F, Ohta N et al (2019) Involvement of melatonin in transducing moon-related signals into the reproductive network of the female honeycomb grouper Epinephelus merra. Gen Comp Endocrinol 282:113211. https://doi.org/10.1016/j.ygcen.2019.113211
Gaildrat P, Falcón J (2000) Melatonin receptors in the pituitary of a teleost fish: mRNA Expression, 2-[125I]iodomelatonin binding and cyclic AMP response. Neuroendocrinology 72:57–66. https://doi.org/10.1159/000054571
García-Allegue R, Madrid JA, Sánchez-Vázquez FJ (2001) Melatonin rhythms in European sea bass plasma and eye: influence of seasonal photoperiod and water temperature. J Pineal Res 31:68–75. https://doi.org/10.1034/j.1600-079X.2001.310110.x
Gaildrat P, Becq F, Falcón J (2002) First cloning and functional characterization of a melatonin receptor in fish brain: A novel one? J Pineal Res 32:74–84. https://doi.org/10.1034/j.1600-079x.2002.1817.x
Ghosh J, Nath P (2005) Seasonal effects of melatonin on ovary and plasma gonadotropin and vitellogenin levels in intact and pinealectomized catfish, Clarias batrachus (Linn). Indian J Exp Biol 43:224–232
Herrera-Pérez P, Servili A, Rendón MC et al (2011) The pineal complex of the European sea bass (Dicentrarchus labrax): I. Histological, immunohistochemical and qPCR study. J Chem Neuroanat 41:170–180. https://doi.org/10.1016/j.jchemneu.2011.01.006
Hong LY, Hong WS, Zhu WB et al (2014) Cloning and expression of melatonin receptors in the mudskipper Boleophthalmus pectinirostris: Their role in synchronizing its semilunar spawning rhythm. Gen Comp Endocrinol 195:138–150. https://doi.org/10.1016/j.ygcen.2013.11.004
Hoque M, Takemura A, Takano K (1998) Annual changes in oocyte development and serum vitellogenin level in the rabbitfish Siganus canaliculatus (Park) in Okinawa, Southern Japan. Fish Sci 64:44–51. https://doi.org/10.2331/fishsci.64.44
Ikegami T, Motohashi E, Doi H et al (2009) Synchronized diurnal and circadian expressions of four subtypes of melatonin receptor genes in the diencephalon of a puffer fish with lunar-related spawning cycles. Neurosci Lett 462:58–63. https://doi.org/10.1016/j.neulet.2009.06.076
Imamura S, Hur S-P, Takeuchi Y et al (2020) The mRNA expression patterns of kisspeptins, GnRHs, and gonadotropins in the brain and pituitary gland of a tropical damselfish, Chrysiptera cyanea, during the reproductive cycle. Fish Physiol Biochem 46:277–291. https://doi.org/10.1007/s10695-019-00715-5
Isorna E, Aliaga-Guerrero M, El M’RA et al (2011) Identification of two arylalkylamine N-acetyltranferase 1 genes with different developmental expression profiles in the flatfish Solea senegalensis. J Pineal Res 51:434–444. https://doi.org/10.1111/j.1600-079X.2011.00907.x
Jin YH, Park JW, Kim J, Kwon JY (2013) The expression pattern of melatonin receptor 1a gene during early life stages in the Nile tilapia (Oreochromis niloticus). Dev Reprod 17(45–53):10.12717/DR.2013.17.1.045
McStay E, Migaud H, Vera LM et al (2014) Comparative study of pineal clock gene and AANAT2 expression in relation to melatonin synthesis in Atlantic salmon (Salmo salar) and European seabass (Dicentrarchus labrax). Comp Biochem Physiol - A Mol Integr Physiol 169:77–89. https://doi.org/10.1016/j.cbpa.2013.12.011
Porter M (2000) Environmental influences on melatonin secretion in Atlantic cod (Gadus morhua L.) and their relevance to commercial culture. Fish Physiol Biochem 23:191–200
Porter MJR, Duncan N, Handeland SO (2001) Temperature, light intensity and plasma melatonin levels in juvenile Atlantic salmon. J Fish Biol 58:431–438. https://doi.org/10.1006/jfbi.2000.1455
Renuka K, Joshi BN (2010) Melatonin-induced changes in ovarian function in the freshwater fish Channa punctatus (Bloch) held in long days and continuous light. Gen Comp Endocrinol 165:42–46. https://doi.org/10.1016/j.ygcen.2009.05.020
Sauzet S, Besseau L, Herrera Perez P et al (2008) Cloning and retinal expression of melatonin receptors in the European sea bass, Dicentrarchus labrax. Gen Comp Endocrinol 157:186–195. https://doi.org/10.1016/j.ygcen.2008.04.008
Sébert M-E, Legros C, Weltzien F-A et al (2008) Melatonin activates brain dopaminergic systems in the eel with an inhibitory impact on reproductive function. J Neuroendocrinol 20:917–929. https://doi.org/10.1111/j.1365-2826.2008.01744.x
Sundararaj BI, Keshavanath P (1976) Effects of melatonin and prolactin treatment on the hypophysial-ovarian system in the catfish, Heteropneustes fossilis (Bloch). Gen Comp Endocrinol 29:84–96. https://doi.org/10.1016/0016-6480(76)90010-1
Takemura A, Uchimura M, Shibata Y (2010) Dopaminergic activity in the brain of a tropical wrasse in response to changes in light and hydrostatic pressure. Gen Comp Endocrinol 166:513–519. https://doi.org/10.1016/j.ygcen.2010.01.001
Urasaki H (1972) Effects of restricted photoperiod and melatonin administration on gonadal weight in the Japanese killifish. J Endocrinol 55:619–620. https://doi.org/10.1677/joe.0.0550619
Vera LM, De Oliveira C, López-Olmeda JF et al (2007) Seasonal and daily plasma melatonin rhythms and reproduction in Senegal sole kept under natural photoperiod and natural or controlled water temperature. J Pineal Res 43:50–55. https://doi.org/10.1111/j.1600-079X.2007.00442.x
Acknowledgements
We gratefully thank the staff of Sesoko Station, Tropical Biosphere Research Center, University of the Ryukyus, Okinawa, Japan, for use of facilities.
Code availability
Not applicable
Funding
This study was supported in part by a Grant-in-Aid for Scientific Research (B) (KAKENHI, Grant number 16H05796) from the Japan Society for the Promotion of Science (JSPS) to AT and Heiwa Nakajima Foundation to AT.
Author information
Authors and Affiliations
Contributions
All authors contributed to the design of this research. SI, SPH, and MB collected fish from coral reefs and maintained them under experimental conditions. SI and YT sampled tissues and carried out molecular analyses. SI, AM, and DR analyzed data statistically and discussed about the results under supervision of AT. All authors contributed to the manuscript preparation. All authors revised and commented on all versions of the manuscript, and then approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval
All experiments were conducted in compliance with the guidelines of the Animal Care and Use Committee of the University of the Ryukyus and the regulations for the care and use of laboratory animals in Japan.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary data 1.
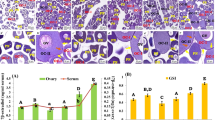
Effect of single treatment with melatonin on ovarian histology of the sapphire devil. (A) Cross section of an ovary (CSO) of fish at the onset of the experiment, (B) CSO of the fish 3 h after feeding with melatonin-free pellets, (C) CSO of the fish 3 h after feeding with melatonin-containing pellets, (D) CSO of the fish 6 h after feeding with melatonin-containing pellets. TYS; tertiary yolk stage. Scale bar = 200 μm. (EPS 2890 kb)
Supplementary data 2.
Effect of daily treatment with melatonin on ovarian histology of the sapphire devil. (A) Cross section of an ovary (CSO) of fish at the onset of the experiment, (B) CSO of the fish fed with melatonin-free pellets for 2 weeks (C) CSO of the fish fed with melatonin-containing pellets for 1 week, (D) CSO of the fish fed with melatonin-containing pellets for 2 weeks. AO, atretic oocyte; PNS, peri-nucleolus stage; TYS, tertiary yolk stage. Scale bar = 200μm. (EPS 2819 kb)
Rights and permissions
About this article
Cite this article
Imamura, S., Hur, SP., Takeuchi, Y. et al. Effect of short- and long-term melatonin treatments on the reproductive activity of the tropical damselfish Chrysiptera cyanea. Fish Physiol Biochem 48, 253–262 (2022). https://doi.org/10.1007/s10695-022-01051-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10695-022-01051-x