Abstract
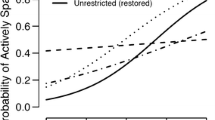
Temperature fluctuations and climate change impacts may substantially affect spawning success of fish, especially migratory species with a limited spawning window. Factors affecting American shad (Alosa sapidissima) spawning success and survival were investigated at different temperatures and periods (peak- and late-spawning periods) during the Connecticut River, USA, spawning migration in 2017. Wild caught American shad were exposed to constant temperatures regimes of 15, 18, 21, 24 and 27 °C for 2 weeks. During the peak-spawning period, an increase in temperature (15–24 °C) was shown to increase spawning success factors, including spawning probability, number of eggs, and fertilization success, but decreased egg size. Temperatures between 18 and 27 °C did not affect these factors during the late-spawning period. Glochidia infection by the alewife floater (Anodonta implicata) was much higher in the late-spawning period and significantly decreased the survival of American shad. Further research should investigate the parasite-host relationship between the alewife floater and American shad to determine annual variability of glochidia infections and how they affect American shad from physiological and passage perspectives. Higher temperatures were shown to increase spawning success of American shad during the peak-spawning period, but temperature had no effect during the late-spawning period. However, any effect during the late-spawning period may have been masked by a high level of glochidia infection.








Similar content being viewed by others
Data availability
Data available upon request.
Code availability
Code available upon request.
References
Bayse SM, McCormick SD, Castro-Santos T (2019) How lipid content and temperature affect American shad (Alosa sapidissima) attempt rate and sprint swimming: implications for overcoming migration barriers. Can J Fish Aquat Sci 76:2235–2244. https://doi.org/10.1139/cjfas-2018-0406
Bayse SM, Regish AM, McCormick SD (2018) Proximate composition, lipid utilization and validation of a non-lethal method to determine lipid content in migrating American shad Alosa sapidissima. J Fish Biol 92:1832–1848. https://doi.org/10.1111/jfb.13624
Bayse SM, Shaughnessy CA, Regish AM, McCormick SD (2020) Upper thermal tolerance and heat shock protein response of juvenile American shad (Alosa sapidissima). Estuar Coasts 43:182–188. https://doi.org/10.1007/s12237-019-00642-x
Bolker B (2017) Package bbmle [online]. R Foundation for Statistical Computing, Vienna, Austria. https://cran.r-project.org/web/packages/bbmle/bbmle.pdf. Accessed 28 May 2020.
Burnham KP, Anderson DR (2004) Multimodel inference: understanding AIC and BIC in model selection. Sociolo Meth Res 33:261–304
Castro-Santos T, Letcher BH (2010) Modeling migratory energetics of Connecticut River American shad (Alosa sapidissima): implications for the conservation of an iteroparous anadromous fish. Can J Fish Aquat Sci 67:806–830. https://doi.org/10.1139/F10-026
CRASC 2016. Connecticut River Atlantic Salmon Commission (CRASC), Turners Falls, Massachusetts Meeting, June 28, 2016. https://www.fws.gov/r5crc/pdf/CRASC_Minutes_06_28_16.pdf. Accessed 28 May 2020.
Crecco VA, Savoy TF (1985) Effects of biotic and abiotic factors on growth and relative survival of young American shad, Alosa sapidissima, in the Connecticut River. Can J Fish Aquat Sci 42:1640–1648. https://doi.org/10.1139/f85-205
Dartnall HJG, Walkey M (1979) The distribution of glochidia of the swan mussel, Anodonta cygnea (Mollusca) on the three-spined stickleback Gasterosteus aculeatus (Pisces). J Zool 189:31–37. https://doi.org/10.1111/j.1469-7998.1979.tb03951.x
Davenport D, Warmuth M (1965) Notes on the relationship between the freshwater mussel Anodonta implicata Say and the Alewife Pomolobus pseudoharengus (Wilson). Lim Oceano 10:R74–R78. https://doi.org/10.4319/lo.1965.10.suppl2.r74
Glebe BD, Leggett WC (1981) Latitudinal differences in energy allocation and use during the freshwater migrations of American shad (Alosa sapidissima) and their life history consequences. Can J Fish Aquat Sci 38:806–820. https://doi.org/10.1139/f81-109
Glebe BD, Leggett WC (1981) Temporal, intra-population differences in energy allocation and use by American shad (Alosa sapidissima) during the spawning migration. Can J Fish Aquat Sci 38:795–805. https://doi.org/10.1139/f81-108
Greene KE, Zimmerman JL, Laney RW, Thomas-Blate JC (2009) Atlantic coast diadromous fish habitat: a review of utilization, threats, recommendations for conservation and research needs. Habitat Management Series No. 9. Atlantic States Marine Fisheries Commission, Washington, D.C. www.asmfc.org/files/Habitat/HMS9_Diadromous_Habitat_2009.pdf. Accessed 28 May 2020.
Hall CJ, Jordaan A, Frisk MG (2012) Centuries of anadromous forage fish loss: consequences for ecosystem connectivity and productivity. BioSci 62:723–731. https://doi.org/10.1525/bio.2012.62.8.5
Horký P, Douda K, Maciak M, Závorka L, Slavík O (2014) Parasite-induced alterations of host behaviour in a riverine fish: the effects of glochidia on host dispersal. Freshw Biol 59:1452–1461. https://doi.org/10.1111/fwb.12357
Horký P, Slavík O, Douda, K (2019) Altered thermoregulation as a driver of host behaviour in glochidia-parasitised fish. J Exp Biol 222:jeb184903. https://doi.org/10.1242/jeb.184903
Hothorn T, Bretz F, Westfall P, Heiberger RM, Schuetzenmeister A, Scheibe S, Hothorn MT (2016) Package ‘multcomp’. Simultaneous inference in general parametric models. R Foundation for Statistical Computing, Vienna, Austria. http://multcomp.r-forge.r-project.org/. Accessed 28 May 2020.
James G, Witten D, Hastie T, Tibshirani R (2013) An introduction to statistical learning: with applications in R. Springer-Verlag, New York. https://doi.org/10.1007/978-1-4614-7138-7
Johnston TA, Leggett WC (2002) Maternal and environmental gradients in the egg size of an iteroparous fish. Ecol 83:1777–1791. https://doi.org/10.1890/00129658(2002)083[1777:MAEGIT]2.0.CO;2
Kat PW (1984) Parasitism and the Unionacea (bivalvia). Biol Rev 59:189–207. https://doi.org/10.1111/j.1469-185X.1984.tb00407.x
Karna DW, Millemann RE (1978) Glochidiosis of salmonid fishes. III. Comparative susceptibility to natural infection with Margaritifera margaritifera (L.)(Pelecypoda: Margaritanidae) and associated histopathology. J Parasitol 64:528–537. https://doi.org/10.2307/3279799
Kassambara A, Kosinski M, Biecek P, Fabian S (2017) Package ‘survminer’. R Foundation for Statistical Computing, Vienna, Austria. https://mran.microsoft.com/snapshot/2017-04-16/web/packages/survminer/survminer.pdf. Accessed 28 May 2020.
Leach SD, Houde ED (1999) Effects of environmental factors on survival, growth, and production of American shad larvae. J Fish Biol 54:767–786. https://doi.org/10.1111/j.1095-8649.1999.tb02032.x
Leggett WC, Whitney RR (1972) Water temperature and migrations of American shad. Fish Bull 70:659–670
Leim AH (1924) The life history of the shad, Alosa sapidissima (Wilson), with special reference to the factors limiting its abundance. Contrib Can Biol Fish 2:161–284
Lenth RV (2017) Using lsmeans. J Stat Softw 69:1–33. R Foundation for Statistical Computing, Vienna, Austria. https://mran.microsoft.com/snapshot/2018-04-11/web/packages/lsmeans/vignettes/using-lsmeans.pdf. Accessed 28 May 2020.
Leonard JBK, McCormick SD (1999) Effects of migration distance on whole-body and tissue-specific energy use in American shad (Alosa sapidissima). Can J Fish Aquat Sci 56:1159–1171. https://doi.org/10.1139/f99-041
Leonard JBK, Norieka JF, Kynard B, McCormick SD (1999) Metabolic rates in an anadromous clupeid, the American shad (Alosa sapidissima). J Comp Physiol B 169:287–295. https://doi.org/10.1007/s003600050223
Limburg KE, Hattala KA, Kahnle A (2003) American shad in its native range. In: Limburg KE Waldman JR (eds) Biodiversity, status, and conservation of the world’s shads. American Fisheries Society Symposium, Bethesda, pp 125–140.
Moodie GEE, Loadman NL, Wiegand MD, Mathias JA (1989) Influence of egg characteristics on survival, growth and feeding in larval walleye (Stizostedion vitreum). Can J Fish Aquat Sci 46:516–521. https://doi.org/10.1139/f89-069
Nezlin LP, Cunjak RA, Zotin AA, Ziuganov VV (1994) Glochidium morphology of the freshwater pearl mussel (Margaritifera margaritifera) and glochidiosis of Atlantic salmon (Salmo salar): a study by scanning electron microscopy. Can J Zool 72:15–21
Olney JE, McBride RS (2003) Intraspecific variation in batch fecundity of American shad: revisiting the paradigm of reciprocal latitudinal trends in reproductive traits. In: Limburg KE Waldman JR (eds) Biodiversity, status, and conservation of the world’s shads. American Fisheries Society Symposium, Bethesda, pp. 185–192.
Pörtner HO, Knust R (2007) Climate change affects marine fishes through the oxygen limitation of thermal tolerance. Science 315:95–97. https://doi.org/10.1126/science.1135013
R Development Core Team. 2009. R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. http://www.R-project.org. Accessed 28 May 2020.
Raabe JK, Hightower JE (2014) American Shad migratory behavior, weight loss, survival, and abundance in a North Carolina river following dam removals. Trans Am Fish Soc 143:673–688. https://doi.org/10.1080/00028487.2014.882410
Ross RM, Bennett RM, Backman TWH (1993) Habitat use by spawning adult, egg, and larval American shad in the Delaware River. Rivers 4:227–238
Smith DG (1985) Recent range expansion of the freshwater mussel Anodonta implicata and its relationship to clupeid fish restoration in the Connecticut River system. Freshw Invert Biol 4:105–108
Therneau TM, Lumley T (2014) Package ‘survival’. R Foundation for Statistical Computing, Vienna, Austria. https://tbrieder.org/epidata/course_reading/e_therneau.pdf. Accessed 28 May 2020.
Trdan RJ (1981) Reproductive biology of Lampsilis radiata siliquoidea (Pelecypoda: Unionidae). Ameri Midl Natur 106:243–248. https://doi.org/10.2307/2425160
Wagenmakers EJ, Farrell S (2004) AIC model selection using Akaike weights. Psychonom Bull Rev 11:192–196
Walburg CH, Nichols PR (1967) Biology and management of the American shad and status of the fisheries, Atlantic coast of the United States, 1960. U.S. Fish and Wildlife Service Special Science Report for Fisheries 550.
Walter JF, Olney JE (2003) Feeding behavior of American Shad during spawning migration in the York River, Virginia. In: Limburg KE Waldman JR (eds) Biodiversity, status, and conservation of the world’s shads. American Fisheries Society Symposium, Bethesda, pp. 201–209.
Williams JD, Warren ML Jr, Cummings KS, Harris JL, Neves RJ (1993) Conservation status of freshwater mussels of the United States and Canada. Fisheries 18:6–22. https://doi.org/10.1577/1548-8446(1993)018%3C0006:CSOFMO%3E2.0.CO;2
Zeileis A, Cribari-Neto F, Gruen B, Kosmidis I, Simas AB, Rocha AV, Zeileis MA (2016) Package ‘betareg’. R package. R Foundation for Statistical Computing, Vienna, Austria. http://citeseerx.ist.psu.edu/viewdoc/download?doi=10.1.1.406.4779&rep=rep1&type=pdf. Accessed 28 May 2020.
Acknowledgements
We express our thanks and appreciation to the many individuals that assisted with this study, including K. Sprankle (U.S. Fish and Wildlife Service (USFWS); T. Castro-Santos (USGS); S. Leach, S. Humiston, and B. LaFlamme (Normandeau Associates); R. Murray (Holyoke Gas & Electric); M. Bailey, K. Cheung, and S. Hanlon (U.S. Fish and Wild. Ser.); J. Noreika, S. Walk, S. Parker, M. Garcia, D. Hall, A. Weinstock, L. Guo, C. Shaughnessy, J. Norstog, D. Ferreira Martins (USGS); and Meghan Donovan (Marine Institute). This work was funded by USGS/USFWS Science Support Partnership (SSP) Program (grant no. SSP-15-R5-02). All experiments were carried out under US Geological Survey Institutional Animal Care and Use Committee Guidelines under protocol no. C09076. Any use of trade, firm, or product names is for descriptive purposes only and does not imply endorsement by the US Government.
Funding
This work was funded by USGS/USFWS Science Support Partnership (SSP) Program (grant no. SSP-15-R5-02).
Author information
Authors and Affiliations
Contributions
SMB: conceptualization, methodology, validation, investigation, data curation, formal analysis, visualization, writing (original draft), writing (review and editing). AMR: conceptualization, methodology, validation, investigation, data curation, visualization, writing (review and editing). SDM: funding acquisition, conceptualization, methodology, validation, investigation, data curation, visualization, writing (review and editing).
Corresponding author
Ethics declarations
Ethics approval
All experiments were carried out under US Geological Survey Institutional Animal Care and Use Committee Guidelines under protocol no. C09076.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Bayse, S.M., Regish, A.M. & McCormick, S.D. Survival and spawning success of American shad (Alosa sapidissima) in varying temperatures and levels of glochidia infection. Fish Physiol Biochem 47, 1821–1836 (2021). https://doi.org/10.1007/s10695-021-01018-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10695-021-01018-4