Abstract
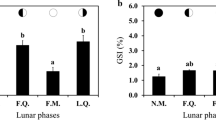
Many teleost fishes can withstand long feed deprivation periods, either due to an eventual lack of food or because of their behavior during reproduction and/or parental care. In this work, the effects of total food restriction on the oogenesis, spermatogenesis, and reproductive hormones of the neotropical cichlid fish Cichlasoma dimerus were studied. Specifically, different pairs were isolated after having a spawning event and were feed-deprived or daily fed for 3 weeks. After that period, gonadal histology, messenger levels of genes related to reproduction (gonadotropin-releasing hormone 1, gonadotropins, and insulin-like growth factor 1) and 11-ketotestosterone plasma levels were evaluated in both groups. Food restriction did not affect the reproductive axis in females since follicular maturation and gene expression showed no differences with respect to controls. However, in males, food restriction showed a stimulatory effect on the reproductive axis, reflected in a greater number of spermatozoa in their seminiferous lobes and spermatic ducts, and in an increase in follicle stimulating hormone messenger expression. Despite the negative effect reported for many fish species, C. dimerus seems to redirect their energy reserves towards gonadal development when faced with to a feed deprivation period.







Similar content being viewed by others
Data availability
The data that support the findings of this study are available from the corresponding author (PGV), upon reasonable request.
References
Benjamini Y, Hochberg Y (1995) Controlling the false discovery rate: a practical and powerful approach to multiple testing. J R Stat Soc Series B Stat Methodol 57:289–300. https://doi.org/10.2307/2346101
Berishivili G, D’Cotta H, Baroiller JF, Segner H, Reinecke M (2006) Differential expression of IGF-I mRNA and peptide in the male and female gonad during early development of bony fish, the tilapia Oreochromis niloticus. Gen Comp Endocrinol 146:204–210. https://doi.org/10.1016/j.ygcen.2005.11.008
Biran J, Levavi-Sivan B (2018) Endocrine control of reproduction, Fish. In: Skinner MK (ed.) Encyclopedia of Reproduction, vol. 6, pp. 362–368. https://doi.org/10.1016/B978-0-12-809633-8.20579-7
Bromley PJ, Ravier C, Witthames PR (2000) The influence of feeding regime on sexual maturation, fecundity and atresia in first-time spawning turbot. J Fish Biol 56:264–278. https://doi.org/10.1111/j.1095-8649.2000.tb02105.x
Caldwell LK, Pierce AL, Riley LG, Duncan CA, Nagler JJ (2014) Plasma nesfatin-1 is not affected by long-term food restriction and does not predict rematuration among iteroparous female rainbow trout (Oncorhynchus mykiss). PLoS One 9:e85700. https://doi.org/10.1371/journal.pone.0085700
Cerdá J, Carrillo M, Zanuy S, Ramos J (1994) Effect of food ration on estrogen and vitellogenin plasma levels, fecundity and larval survival in captive sea bass, Dicentrarchus labrax: preliminary observations. Aquat Living Resour 7:255–266. https://doi.org/10.1051/alr:1994028
Chang JP, Johnson JD, Sawisky GR, Grey CL, Mitchell G, Booth M, Volk MM, Parks SK, Thompson E, Goss GG, Klausen C, Habibi R (2009) Signal transduction in multifactorial neuroendocrine control of gonadotropin secretion and synthesis in teleosts—studies on the goldfish model. Gen Comp Endocrinol 161:42–52. https://doi.org/10.1016/j.ygcen.2008.09.005
Delgadin TH, Pérez Sirkin DI, Di Yorio MP, Arranz SE, Vissio PG (2015) GH, IGF-I and GH receptors mRNA expression in response to growth impairment following a food deprivation period in individually housed cichlid fish Cichlasoma dimerus. Fish Physiol Biochem 41:51–60. https://doi.org/10.1007/s10695-014-0005-x
Delgadin TH, Simó I, Pérez Sirkin DI, Di Yorio MP, Arranz SE, Vissio PG (2018) Cichlasoma dimerus responds to refeeding with a partial compensatory growth associated with an increment of the feed conversion efficiency and a rapid recovery of GH/IGFs axis. Aquac Nutr 24:1234–1243. https://doi.org/10.1111/anu.12661
Escobar S, Felip A, Zanuy S, Carrillo M (2016) Is the kisspeptin system involved in responses to food restriction in order to preserve reproduction in pubertal male sea bass (Dicentrarchus labrax)? Comp Biochem Physiol A Mol Integr Physiol 199:38–46. https://doi.org/10.1016/j.cbpa.2016.05.005
Frantzen M, Damsgård B, Tveiten H, Moriyama S, Iwata M, Johnsen HK (2004) Effects of fasting on temporal changes in plasma concentrations of sex steroids, growth hormone and insulin-like growth factor I, and reproductive investment in Arctic charr. J Fish Biol 65:1526–1542. https://doi.org/10.1111/j.0022-1112.2004.00564.x
García-Lopez A, Bogerd J, Granneman JC, van Dijk W, Trant JM, Taranger GL, Schulz RW (2008) Leydig cells express follicle-stimulating hormone receptors in African catfish. Endocrinology 150:357–365. https://doi.org/10.1210/en.2008-0447
Grier HJ, Uribe-Aranzábal MC, Patino R (2009) The ovary, folliculogenesis, and oogenesis in teleosts. In: Jamieson BGM (ed) Reproductive biology and phylogeny of fishes (agnathans and bony fishes). Science Publishers, Enfield, N.H., pp. 25–84
Grone BP, Carpenter RE, Lee M, Maruska KP, Fernald RD (2012) Food deprivation explains effects of mouthbrooding on ovaries and steroid hormones, but not brain neuropeptide and receptor mRNAs, in an African cichlid fish. Horm Behav 62:18–26. https://doi.org/10.1016/j.yhbeh.2012.04.012
Hunter JR, Macewicz J (1985) Rates of atresia in the ovary of captive and wild northern anchovy; Engraulis mordax. Fish Bull 83:119–136
Jobling M (2016) Fish nutrition research: past, present and future. Aquacult Int 24:767–786. https://doi.org/10.1007/s10499-014-9875-2
Kohler KE, Gill SM (2006) Coral Point Count with Excel extensions (CPCe): a visual basic program for the determination of coral and substrate coverage using random point count methodology. Comput Geosci 32:1259–1269. https://doi.org/10.1016/j.cageo.2005.11.009
Lubzens E, Young G, Bobe J, Cerdà J (2010) Oogenesis in teleosts: how fish eggs are formed. Gen Comp Endocrinol 165:367–389. https://doi.org/10.1016/j.ygcen.2009.05.022
MacKenzie DS, Vanputte CM, Leiner KA (1998) Nutrient regulation of endocrine function in fish. Aquaculture 161:3–25. https://doi.org/10.1016/S0044-8486(97)00253-6
Maruska KP, Cowie EG, Tricas TC (1996) Periodic gonadal activity and protracted mating in elasmobranch fishes. J Exp Zool 276:219–232. https://doi.org/10.1002/(SICI)1097-010X(19961015)276:3%3c219::AID-JEZ6%3e3.0.CO;2-Q
Maruska KP, Fernald RD (2011) Social regulation of gene expression in the hypothalamic-pituitary-gonadal axis. Physiology 26:412–423. https://doi.org/10.1152/physiol.00032.2011
Mechaly AS, Vinas J, Piferrer F (2011) Gene structure analysis of kisspeptin-2 (Kiss2) in the Senegalese sole (Solea senegalensis): characterization of two splice variants of Kiss2, and novel evidence for metabolic regulation of kisspeptin signaling in non-mammalian species. Mol Cell Endocrinol 339:14–24. https://doi.org/10.1016/j.mce.2011.03.004
Meijide FJ, Guerrero GA (2000) Embryonic and larval development of a substrate-brooding cichlid Cichlasoma dimerus (Heckel, 1840) under laboratory conditions. J Zool 252:481–493. https://doi.org/10.1111/j.1469-7998.2000.tb01231.x
Midwood JD, Larsen MH, Aarestrup K, Cooke SJ (2016) Stress and food deprivation: linking physiological state to migration success in a teleost fish. J Exp Biol 219:3712–3718. https://doi.org/10.1242/jeb.140665
Mills SC, Mourier J, Galzin R (2010) Plasma cortisol and 11-ketotestosterone enzyme immunoassay (EIA) kit validation for three fish species: the orange clownfish Amphiprion percula, the orangefin anemonefish Amphiprion chrysopterus and the blacktip reef shark Carcharhinus melanopterus. J Fish Biol 77:769–777. https://doi.org/10.1111/j.1095-8649.2010.02693.x
Montrezor LH, Urbinati EC (2017) Feed restriction and insulin-like growth factor-I (IGF-I) affect the oocyte maturation in matrinxã Brycon amazonicus. Zygote 25:56–64. https://doi.org/10.1017/S0967199416000344
Morandini L, Honji RM, Ramallo MR, Moreira RG, Pandolfi M (2014) The interrenal gland in males of the cichlid fish Cichlasoma dimerus: relationship with stress and the establishment of social hierarchies. Gen Comp Endocrinol 195:88–98. https://doi.org/10.1016/j.ygcen.2013.10.009
Nader MR, Miura T, Ando N, Miura C, Yamauchi K (1999) Recombinant human insulin-like growth factor I stimulates all stages of 11-ketotestosterone-induced spermatogenesis in the Japanese eel, Anguilla japonica, in vitro. Biol Reprod 61:944–947. https://doi.org/10.1095/biolreprod61.4.944
Navarro I, Gutiérrez J (1995) Fasting and starvation. In: Hochachka PW, Mommsen TP (eds) Biochemistry and molecular biology of fishes. Elseiver, Amsterdam 4, pp. 393–434
Ohta T, Miyake H, Miura C, Kamei H, Aida K, Miura T (2007) Follicle-stimulating hormone induces spermatogenesis mediated by androgen production in Japanese eel, Anguilla japonica. Biol Reprod 77:970–977. https://doi.org/10.1095/biolreprod.107.062299
Onuma TA, Makino K, Ban M, Ando H, Fukuwaka MA, Azumaya T, Swanson P, Urano A (2009) Elevation of the plasma level of insulin-like growth factor-I with reproductive maturation prior to initiation of spawning migration of chum salmon. Ann NY Acad Sci 1163:497–500. https://doi.org/10.1111/j.1749-6632.2008.03668.x
Pandolfi M, Cánepa MM, Meijide FJ, Alonso F, Rey Vázquez G, Maggese MC, Vissio PG (2009) Studies on the reproductive and developmental biology of Cichlasoma dimerus (Percifomes, Cichlidae). Biocell 33:1–18
Pike N (2011) Using false discovery rates for multiple comparisons in ecology and evolution. Methods Ecol Evol 2:278–282. https://doi.org/10.1111/j.2041-210X.2010.00061.x
Pikle RP, Jatiger RM, Ganesh CB (2017) Food-deprivation-induced suppression of pituitary–testicular-axis in the tilapia Oreochromis mossambicus. Int Aquat Res 9:203–213. https://doi.org/10.1007/s40071-017-0169-y
Ramakers C, Ruijter JM, Deprez RHL, Moorman AF (2003) Assumption-free analysis of quantitative real-time polymerase chain reaction (PCR) data. Neurosci Lett 339:62–66. https://doi.org/10.1016/S0304-3940(02)01423-4
Ramallo MR, Birba A, Honji RM, Morandini L, Somoza GM, Moreira RG, Pandolfi M (2015) A multidisciplinary study on social status and the relationship between inter-individual variation in hormone levels and agonistic behavior in a Neotropical cichlid fish. Horm Behav 69:139–151. https://doi.org/10.1016/j.yhbeh.2015.01.008
Ramallo MR, Honji RM, Birba A, Morandini L, Varela ML, Genovese G, Moreira RG, Somoza GM, Pandolfi M (2017) A game of two? Gene expression analysis of brain (cyp19a1b) and gonadal (cyp19a1a) aromatase in females of a Neotropical cichlid fish through the parental care period and removal of the offspring. Gen Comp Endocrinol 252:119–129. https://doi.org/10.1016/j.ygcen.2017.08.009
Reinecke M (2010) Insulin-like growth factors and fish reproduction. Biol Reprod 82:656–661. https://doi.org/10.1095/biolreprod.109.080093
Rey Vázquez G, Da Cuña RH, Meijide FJ, Guerrero GA (2012) Spermatogenesis and changes in testicular structure during the reproductive cycle in Cichlasoma dimerus (Teleostei, Perciformes). Acta Zool 93:338–350. https://doi.org/10.1111/j.1463-6395.2011.00508.x
Ruijter JM, Ramakers C, Hoogaars WMH, Karlen Y, Bakker O, Van den Hoff MJB, Moorman AFM (2009) Amplification efficiency: linking baseline and bias in the analysis of quantitative PCR data. Nucleic Acids Res 37:e45. https://doi.org/10.1093/nar/gkp045
Schulz RW, De França LR, Lareyre JJ, LeGac F, Chiarini-Garcia H, Nobrega RH, Miura T (2010) Spermatogenesis in fish. Gen Comp Endocrinol 165:390–411. https://doi.org/10.1016/j.ygcen.2009.02.013
Suchiang P, Gupta BBP (2011) Effects of partial and full feed restriction on the plasma levels of thyroid hormones and testicular activity in the male air-breathing catfish, Clarias gariepinus during different phases of the breeding cycle. Int J Biol 3:32. https://doi.org/10.5539/ijb.v3n2p32
Townshend TJ, Wootton RJ (1984) Effects of food supply on the reproduction of the convict cichlid, Cichlasoma nigrofasciatum. J Fish Biol 24:91–104. https://doi.org/10.1111/j.1095-8649.1984.tb04779.x
Tubert C, Lo Nostro F, Villafañe V, Pandolfi M (2012) Aggressive behavior and reproductive physiology in females of the social cichlid fish Cichlasoma dimerus. Physiol Behav 106:193–200. https://doi.org/10.1016/j.physbeh.2012.02.002
Varela ML, Ferreira MF, Da Cuña RH, Lo Nostro FL, Genovese G, Meijide FJ (2017) Dynamics of ovarian maturation throughout the reproductive cycle of the Neotropical cichlid fish Cichlasoma dimerus (Teleostei, Cichliformes). Can J Zool 95:485–498. https://doi.org/10.1139/cjz-2016-0198
Wood AW, Duan C, Bern HA (2005) Insulin-like growth factor signaling in fish. Int Rev Cytol 243:215–285. https://doi.org/10.1016/S0074-7696(05)43004-1
Yamamoto Y, Luckenbach JA, Goetz FW, Young G, Swanson P (2011) Disruption of the salmon reproductive endocrine axis through prolonged nutritional stress: changes in circulating hormone levels and transcripts for ovarian genes involved in steroidogenesis and apoptosis. Gen Comp Endocrinol 172:331–343. https://doi.org/10.1016/j.ygcen.2011.03.017
Yoneda M, Kitano H, Tanaka H, Kawamura K, Selvaraj S, Ohshimo S, Matsuyama M, Shimizu A (2014) Temperature- and income resource availability-mediated variation in reproductive investment in a multiple-batch-spawning Japanese anchovy. Mar Ecol Prog Ser 516:251–262. https://doi.org/10.3354/meps10969
Acknowledgements
We thank Dr. Ariadna Battista and Mr. Federico Obertello for their technical assistance and Lic. M. Luisa Varela for providing technical equipment.
Funding
This work has been supported by the Universidad de Buenos Aires (grant number: 20020160100110BA to PGV), Consejo Nacional de Investigaciones Científicas y Técnicas (CONICET) (PIP 2014–2016:11220130100501CO to PGV), and ANPCyT PICT 2015–2783 (GMS).
Author information
Authors and Affiliations
Contributions
DIPS, PGV, and GMS contributed to the design of the work, interpretation of the results, and writing of the manuscript; DIPS, MPDY, THD, PGV, and GMS contributed to the interpretation of the results; RMH and RGM contributed to the 11-KT plasma level measurement. All authors approved the final version of the manuscript.
Corresponding author
Ethics declarations
Ethics approval
Comisión Institucional para el Cuidado y Uso de Animales de Laboratorio, Facultad de Ciencias Exactas y Naturales, Buenos Aires, Argentina (Protocol number 26).
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Pérez-Sirkin, D.I., Di Yorio, M.P., Delgadin, T.H. et al. Post-spawning feed deprivation effects on testicular and ovarian maturation in the neotropical cichlid fish Cichlasoma dimerus. Fish Physiol Biochem 47, 1527–1540 (2021). https://doi.org/10.1007/s10695-021-00993-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10695-021-00993-y