Abstract
In order to study the variation of gonad lipidomics during reproductive cycle, black rockfish was employed as the research model in the present study. Using histology, lipidomics, and qPCR, the profile of gonad lipidomics and the expression levels of related genes during different developmental stages were detected and analyzed to show the potential regulatory network of lipid metabolism. Based on Ultra High-Performance Liquid Tandem Chromatography Quadrupole Time of Flight Mass Spectrometry (UHPLC-QTOFMS), four significant differential glycerophospholipid metabolic pathways including phosphatidylcholine (PC), phosphatidylethanolamine (PE), phosphatidylserine (PS), and phosphatidic acid (PA) were enriched by KEGG. Pathway-related enzyme-coding genes, including phosphatidylserine decarboxylase (pisd), phosphatidylserine synthase (ptdss1, ptdss2), and phospholipase D (pld1, pld2) were identified from the whole genome data and confirmed by cloning. The expression profiles of these genes were tested by qPCR in the tissues and gonads in developmental stages, and we found that pisd, pld, and ptdss genes were all downregulated through the developmental process in the brain of male, and the latter two genes were upregulated in the liver and testis at stage IV, which were the opposite trend observed in the female. Thus, our findings would be helpful in further understanding the substance metabolism and regulation during gonad development in ovoviviparity teleosts.









Similar content being viewed by others
Data Availability
The authors confirm that the data supporting the findings of this study are available within the article and its supplementary materials.
References
Aktas M, Wessel M, Hacker S, Klüsener S, Gleichenhagen J, Narberhaus F (2010) Phosphatidylcholine biosynthesis and its significance in bacteria interacting with eukaryotic cells. Eur J Cell Biol 89:888–894
Aloia RC (1983) Membrane fluidity in biology. Cellular Aspects:293–300
Bergo MO, Gavino BJ, Steenbergen R, Sturbois B, Parlow AF, Sanan DA, Skarnes WC, Vance JE, Young SG (2002) Defining the importance of phosphatidylserine synthase 2 in mice. J Biol Chem 277:47701–47708
Blankenberg FG, Katsikis PD, Tait JF, Davis RE, Naumovski L, Ohtsuki K, Kopiwoda S, Abrams MJ, Darkes M, Robbins RC, Maecker HT, SH W (1998) In vivo detection and imaging of phosphatidylserine expression during programmed cell death. Proc Natl Acad Sci U S A 95:6349–6354
Borkenhagen LF, Kennedy EP, Fielding L (1961) Enzymatic formation and decarboxylation of phosphatidylserine. J Biol Chem 236:28–PC30
Buckland AG, Wilton DC (2000) Anionic phospholipids, interfacial binding and the regulation of cell functions. Biochim Biophys Acta 1483:199–216
Calzada E, Onguka O, Claypool SM (2016) Phosphatidylethanolamine metabolism in health and disease. Int Rev Cell Mol Biol 321:29–88
Castellini C, Cardinali R, Bosco AD, Minelli A, Camici O (2006) Lipid composition of the main fractions of rabbit semen. Theriogenology 65:703–712
Dennis RV (2003) New perspectives on the regulation of intermembrane glycerophospholipid traffic. J Lipid Res 44:441–449
Drokin SI (1993) Phospholipids and fatty acids of phospholipids of sperm from several freshwater and marine species of fish. Comp Biochem Physiol B 104:423–428
English D, Cui Y, Siddiqui RA (1996) Messenger functions of phosphatidic acid. Chem Phys Lipids 80:117–132
Evans RW, Setchell BP (1978) Association of exogenous phospholipids with spermatozoa. J Reprod Fertil 53:357–362
Exton JH (1994) Phosphatidylcholine breakdown and signal transduction. BBA- Lipids and Lipid Metabolism 1212:26–42
Exton JH (2002) Regulation of phospholipase D. FEBS Lett 531:58–61
Gao TX, Ding K, Song N, Zhang X, Han Z (2017) Comparative analysis of multiple paternity in different populations of viviparous black rockfish, Sebastes schlegelii, a fish with long-term female sperm storage. Mar Biodivers 6:1–8
Geiger O, López-Lara IM, Sohlenkamp C (2013) Phosphatidylcholine biosynthesis and function in bacteria. BBA- Molecular and Cell Biology of Lipids 1831:503–513
Goldfine H (1982) Lipids of prokaryotes–structure and distribution. Curr Top Membr Trans 17:1–43
Hashemi HF, Goodman JM (2015) The life cycle of lipid droplets. Curr Opin Cell Biol 33:119–124
Hinkovska-Galcheva V, Petkova D, Koumanov K (1989) Changes in the phospholipid composition and phospholipid asymmetry of ram sperm plasma membranes after cryopreservation. Cryobiology 26:70–75
Hiratsuka S, Kitagawa T, Matsue Y, Hashidume M, Wada S (2004) Lipid class and fatty acid composition of phospholipids from the gonads of skipjack tuna. Fish Sci 70:903–909
Hu C, Heijden RVD, Wang M, Greef JVD, Hankemeier T, Xu G (2009) Analytical strategies in lipidomics and applications in disease biomarker discovery. J Chromatogr B Anal Technol Biomed Life Sci 877:2836–2846
Huang X, Yin Y, Shi Z, Li W, Zhou H, Lv W (2010) Lipid content and fatty acid composition in wild-caught silver pomfret (Pampus argenteus) broodstocks: effects on gonad development. Aquaculture 310:192–199
Kawaguchi M, Nakagawa M, Noda T, Yoshizaki N, Hiroi J, Nishida M, Iuchi I, Yasumasu S (2010) Hatching enzyme of the ovoviviparous black rockfish Sebastes schlegelii- environmental adaptation of the hatching enzyme and evolutionary aspects of formation of the pseudogene. FEBS J 275:2884–2898
Kevala JH, Kim HY (2001) Determination of substrate preference in phosphatidylserine decarboxylation by liquid chromatography–electrospray ionization mass spectrometry. Anal Biochem 292:130–138
Küllenberg D, Taylor LA, Schneider M, Massing U (2012) Health effects of dietary phospholipids. Lipids Health Dis 11:1–16
Lee ZW, Kweon SM, Kim BC, Leem SH, Shin I, Kim JH, Ha KS (1998) Phosphatidic acid-induced elevation of intracellular Ca2+ is mediated by RhoA and H2O2 in Rat-2 fibroblasts. J Biol Chem 273:12710–12715
Lin Q, London E, Holowka D (2014) Preparation of artificial plasma membrane mimicking vesicles with lipid asymmetry. PLoS One 9:e87903
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2−ΔΔCT Method. Methods 25:402–408
Ma LM, Wang WJ, Liu CH, Yu HY, Wang ZG, Wang XB, Qi J, Zhang QQ (2013) Selection of reference genes for reverse transcription quantitative real-time PCR normalization in black rockfish (Sebastes schlegeli). Mar Genomics 11:67–73
Marei WF, Wathes DC, Fouladi-Nashta AA (2012) Differential effects of linoleic and alpha-linolenic fatty acids on spatial and temporal mitochondrial distribution and activity in bovine oocytes. Reprod Fert Develop 24:679–690
McDaniel M, Maier SF, Einstein G (2002) “Brain-specific” nutrients: a memory cure? Psychol Sci Public Interest 3:12–38
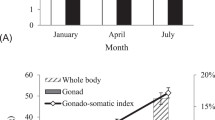
Mori H, Nakagawa M, Soyano K, Koya Y (2011) Annual reproductive cycle of black rockfish Sebastes schlegeli in captivity. Fish Sci 69:910–923
Mürke E, Stoll S, Lendeckel U, Reinhold D, Schild L (2016) The mitochondrial phospholipid cardiolipin is involved in the regulation of T-cell proliferation. BBA - Mol Cell Biol L 1861:748–754
Nicolson GL, Ash ME (2017) Membrane lipid replacement for chronic illnesses, aging and cancer using oral glycerolphospholipid formulations with fructooligosaccharides to restore phospholipid function in cellular membranes, organelles, cells and tissues. BBA - Biomembranes 1859:1704–1724
Paczkowski M, Schoolcraft WB, Krisher RL (2014) Fatty acid metabolism during maturation affects glucose uptake and is essential to oocyte competence. Reproduction 148:429–439
Paulini F, Silva RC, JLJdPRCM L (2014) Ultrastructural changes in oocytes during folliculogenesis in domestic mammals. J Ovarian Res 7:102
Roustaian P, Kamarudin MS, Omar H, Saad CR, Ahmad MH (1999) Changes in fatty acid profile during larval development of freshwater prawn Macrobrachium rosenbergii (de Man). Aquac Res 30:815–824
Rücker-Martin C, Hénaff M, Hatem SN, Delpy E, Mercadier J-J (1999) Early redistribution of plasma membrane phosphatidylserine during apoptosis of adult rat ventricular myocytes in vitro. Basic Res Cardiol 94:171–179
Salze G, Tocher DR, Roy WJ, Robertson DA (2010) Egg quality determinants in cod (Gadus morhua L.): egg performance and lipids in eggs from farmed and wild broodstock. Aquac Res 36:1488–1499
Schmelzle T, Hall MN (2000) TOR, a central controller of cell growth. Cell 103:253–262
Shiao Y, Lupo G, Vance JE (1995) Evidence that phosphatidylserine is imported into mitochondria via a mitochondria-associated membrane and that the majority of mitochondrial phosphatidylethanolamine is derived from decarboxylation of phosphatidylserine. J Biol Chem 270:11190–11198
Simpson AM, Swan MA, White IG (1986) Action of phosphatidylcholine in protecting ram sperm from cold shock. Mol Reprod Dev 15:43–56
Sohlenkamp C, IM L, Geiger O (2003) Biosynthesis of phosphatidylcholine in bacteria. Prog Lipid Res 42:115–162
Songlin L, Wen W, Xu G, Xuxiong H, Naisong C (2018) Variation of lipids and fatty acids composition in the tissues of wild devil stinger (Inimicus japonicas) during sexual maturation. Aquaculture & Fisheries 3:115–121
Steenbergen R, Nanowski TS, Beigneux A, Kulinski A, Young SG, Vance JE (2005) Disruption of the phosphatidylserine decarboxylase gene in mice causes embryonic lethality and mitochondrial defects. J Biol Chem 280:40032–40040
Stone SJ, Vance JE (1999) Cloning and expression of murine liver phosphatidylserine synthase (PSS)-2: differential regulation of phospholipid metabolism by PSS1 and PSS2. Biochem J 342:57–64
Sturbois-Balcerzak B, Stone SJ, Sreenivas A, Vance JE (2001) Structure and expression of the murine phosphatidylserine synthase-1 gene. J Biol Chem 276:8205–8212
Taneva SG, Lee J, Knowles DG, Tishyadhigama C, Cornell RB (2019) Interdomain communication in the phosphatidylcholine regulatory enzyme, CCTα, relies on a modular αE helix. Journal of Biological Chemistry 294:jbc.RA119.009849
Tocher DR, Bendiksen EÅ, Campbell PJ, Bell JG (2008) The role of phospholipids in nutrition and metabolism of teleost fish. Aquaculture 280:21–34
Tomohiro S, Kawaguti A, Kawabe Y, Kitada S, Kuge O (2009) Purification and characterization of human phosphatidylserine synthases 1 and 2. Biochem J 418:421–429
Toyomizu M, Hanaoka K, Satake K, Nakagawa H (1977) Effect of storage temperatures on accumulation of glycerylphosphorylcholine and decomposition of phosphatidylcholine in fish muscle during cold storage. Nippon Suisan Gakkaishi 43:1181–1187
Vance JE (2015) Phospholipid synthesis and transport in mammalian cells. Traffic 16:1–18
Vance JE, Tasseva G (2013) Formation and function of phosphatidylserine and phosphatidylethanolamine in mammalian cells. BBA - Molecular and Cell Biology of Lipids 1831:543–554
Warzych E, Pawlak P, Pszczola M, Cieslak A, Madeja ZE, Lechniak D (2017) Interactions of bovine oocytes with follicular elements with respect to lipid metabolism. Anim Sci J 88:1491–1497
Wiegand MD (1996) Composition, accumulation and utilization of yolk lipids in teleost fish. Rev Fish Biol Fish 6:259–286
Wouters R, Molina C, Lavens P, Calderón J (2001) Lipid composition and vitamin content of wild female Litopenaeus vannamei in different stages of sexual maturation. Aquaculture 198:307–323
Zwaal RFA, Schroit AJ (1997) Pathophysiologic implications of membrane phospholipid asymmetry in blood cells. Blood 89:1121–1132
Code availability
Not applicable.
Funding
This study was supported by the National Natural Science Foundation of China (41676126, 41976089) and the National Key R&D Program of China (2018YFD0901204). Our funding agencies did not play a role in the study design, data collection, analysis, interpretation of the data, or preparation of the manuscript.
Author information
Authors and Affiliations
Contributions
WHS, LY, and QX designed the study. SM performed the metabolome and qRT-PCR experiment. LLK, WXJ, ZY, SM, and LJS performed in samples collection. LJS wrote the manuscript, and QX provided manuscript editing and feedback. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethical approval
All procedures involved in dealing of fish in this study were approved by the Animal Research and Ethics Committees of the Ocean University of China (Permit Number: 20141201) prior to the initiation of the study. The studies did not involve endangered or protected species. And all experiments were performed in accordance with the Guidelines for the Care and Use of Laboratory Animals in China.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary figure 1:
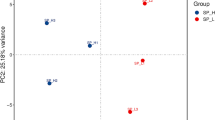
Lipidomics profile of male black rockfish gonads during different gonadal stages. a,b total ion current chromatograms (TIC) of QC samples in positive and negative ion mode, respectively. c,d Score scatter plot of total samples PCA model in positive and negative ion mode, respectively. (PDF 361 kb)
Supplementary figure 2:
The multivariate data analysis between stage IV and III of testis. a,b Score plot of PCA analysis in positive and negative ion mode, respectively. c,d Score plot of OPLS-DA model in positive and negative ion mode. e,f Permutation test of OPLS-DA model in positive and negative ion mode, respectively. (PDF 2525 kb)
Supplementary figure 3:
The multivariate data analysis between stage V and IV of testis. a,b Score plot of PCA analysis in positive and negative ion mode, respectively. c,d Score plot of OPLS-DA model in positive and negative ion mode, respectively. e,f Permutation test of OPLS-DA model in positive and negative ion mode, respectively. (PDF 2539 kb)
Supplementary figure 4:
Lipidomics profile of female black rockfish gonads during different gonadal stages. a,b TIC of QC samples in positive and negative ion mode, respectively. c,d Score scatter plot of total samples PCA model in positive and negative ion mode, respectively. (PDF 1865 kb)
Supplementary figure 5:
The multivariate data analysis between stage IV and II of ovaries. a,b Score plot of PCA analysis in positive and negative ion mode, respectively. c,d Score plot of OPLS-DA model in positive and negative ion mode. e,f Permutation test of OPLS-DA model in positive and negative ion mode, respectively. (PDF 2453 kb)
Supplementary figure 6:
The multivariate data analysis between stage V and IV of ovaries. a,b Score plot of PCA analysis in positive and negative ion mode, respectively. c,d Score plot of OPLS-DA model in positive and negative ion mode, respectively. e,f Permutation test of OPLS-DA model in positive and negative ion mode, respectively. (PDF 2470 kb)
Supplementary figure 7:
Phylogenetic tree of gene. a, pld. b, ptdss. c, pisd. (PDF 1338 kb)
Supplementary figure 8:
Gene expression levels in different tissue. a,b pld. c,d ptdss. e, pisd. (PDF 282 kb)
Rights and permissions
About this article
Cite this article
Li, J., Song, M., Wen, H. et al. Gonadal lipidomics profile of an ovoviviparity teleost, black rockfish, during gonadal development. Fish Physiol Biochem 47, 811–828 (2021). https://doi.org/10.1007/s10695-021-00936-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10695-021-00936-7