Abstract
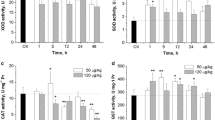
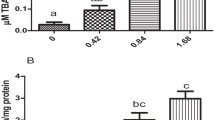
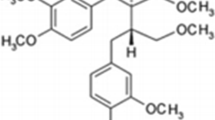
Microcystin-LR (MC-LR) is a well-known hepatotoxin; however, increasing evidence suggests that it might induce kidney injury. Grass carp (Ctenopharyngodon idella) is one of the most important farmed species and may be affected by MC-LR releasing into waterbody during cyanobacterial bloom. Here, this present study aimed to explore the nephrotoxicity of grass carp by MC-LR. The grass carp received a single intraperitoneal injection of different doses of MC-LR (0, 25, 75, and 100 μg/kg body weight (BW)), and the kidneys were isolated at 24 and 96 h post-injection (hpi). Histopathological examination revealed kidney lesions, with severe hemorrhage, necrosis of the interstitium, and dilation of Bowman’s capsule in the 75 and 100 μg MC-LR/kg BW groups. Under transmission electron microscopy, a larger number of swelling and vacuolated degeneration of mitochondria were observed; moreover, apoptotic features, such as condensed chromatin and shrinkage of cells, were observed in the 75 and 100 μg MC-LR/kg BW groups at 96 hpi. MC-LR significantly upregulated the number of apoptotic cells in the 75 and 100 μg/kg BW groups at 96 hpi as indicated by terminal deoxynucleotidyl transferase (TdT) dUTP nick-end labeling (TUNEL) assay (P < 0. 05). The results of quantitative assays showed that the mRNA expression of Bax, caspase-9, and caspase-3 in grass carp kidney were significantly increased at 96 hpi in the 75 and 100 μg MC-LR/kg BW groups compared with that in the control group, but Bcl-2 mRNA expression was significantly decreased in all the treatment groups at 24 and 96 hpi. Taken together, these results indicated that MC-LR damaged the kidney structure and resulted in renal apoptosis which may occur via the mitochondrial pathway.




Similar content being viewed by others
References
Atencio L, Moreno I, Jos A, Pichardo S, Moyano R, Blanco A, Camean AM (2008) Dose-dependent antioxidant responses and pathological changes in tenca (Tinca tinca) after acute oral exposure to Microcystis under laboratory conditions. Toxicon 52:1–12
Cadet J, Douki T, Ravanat JL (2015) Oxidatively generated damage to cellular DNA by UVB and UVA radiation. Photochem Photobiol 91:140–155
Chawla-Sarkar M, Lindner DJ, Liu YF, Williams BR, Sen GC, Silverman RH, Borden EC (2003) Apoptosis and interferons: role of interferon-stimulated genes as mediators of apoptosis. Apoptosis 8:237–249
Chipuk JE, Green DR (2008) How do BCL-2 proteins induce mitochondrial outer membrane permeabilization? Trends Cell Biol 18:157–164
Danial NN, Korsmeyer SJ (2004) Cell death: critical control points. Cell 116:205–219
Dietrich DR (2000) Microcystin-LR toxicodynamics, induced pathology, and immunohistochemical localization in livers of blue-green algae exposed rainbow trout (Oncorhynchus mykiss). Toxicol Sci 54(2):365–373
Drobac D, Tokodi N, Lujic J, Marinovic Z, Subakov-Simic G, Dulic T, Vazic T, Nybom S, Meriluoto J, Codd GA, Svircev Z (2016) Cyanobacteria and cyanotoxins in fishponds and their effects on fish tissue. Harmful Algae 55:66–76
Fladmark KE, Brustugun OT, Hovland R, Boe R, Gjertsen BT, Zhivotovsky B, Doskeland SO (1999) Ultrarapid caspase-3 dependent apoptosis induction by serine/threonine phosphatase inhibitors. Cell Death Differ 6:1099–1108
Gao X, Zheng Y, Ruan X, Ji H, Peng L, Guo D, Jiang S (2018) Salinomycin induces primary chicken cardiomyocytes death via mitochondria mediated apoptosis. Chem Biol Interact 282:45–54
Hensley P, Mishra M, Kyprianou N (2013) Targeting caspases in cancer therapeutics. Biol Chem 394:831–843
Hoeger SJ, Hitzfeld BC, Dietrich DR (2005) Occurrence and elimination of cyanobacterial toxins in drinking water treatment plants. Toxicol Appl Pharm 203:231–242
Honkanen RE, Zwiller J, Moore RE, Daily SL, Khatra BS, Dukelow M, Boynton AL (1990) Characterization of microcystin- LR, a potent inhibitor of type 1 and type 2A protein phosphatases. J Biol Chem 265(32):19401–19404
Huang H, Liu C, Fu X, Zhang S, Xin Y, Li Y, Xue L, Cheng X, Zhang H (2016) Microcystin-LR induced apoptosis in rat sertoli cells via the mitochondrial caspase-dependent pathway: role of reactive oxygen species. Front Physiol 7:397
Hughes MA, Powley IR, Jukes-Jones R, Horn S, Feoktistova M, Fairall L, Schwabe JW, Leverkus M, Cain K, MacFarlane M (2016) Co-operative and hierarchical binding of c-FLIP and caspase-8: a unified model defines how c-FLIP isoforms differentially control cell fate. Mol Cell 61:834–849
Jia J, Luo W, Lu Y, Giesy JP (2014) Bioaccumulation of microcystins (MCs) in four fish species from Lake Taihu, China: assessment of risks to humans. Sci Total Environ 487:224–232
Kotak BG, Semalulu S, Fritz DL, Prepas EE, Hrudey SE, Coppock RW (1996) Hepatic and renal pathology of intraperitoneally administered microcystin-LR in rainbow trout (Oncorhynchus mykiss). Toxicon 34:517–525
Kumar S (2007) Caspase function in programmed cell death. Cell Death Differ 14:32–43
La-Salete R, Oliveira MM, Palmeira CA, Almeida J, Peixoto FP (2008) Mitochondria a key role in microcystin-LR kidney intoxication. J Appl Toxicol 28:55–62
Li L, Xie P, Lei H, Zhang X (2013) Renal accumulation and effects of intraperitoneal injection of extracted microcystins in omnivorous crucian carp (Carassius auratus). Toxicon 70:62–69
Li Y, Li J, Huang H, Yang M, Zhuang D, Cheng X, Zhang H, Fu X (2016) Microcystin-LR induces mitochondria-mediated apoptosis in human bronchial epithelial cells. Exp Ther Med 12:633–640
Lin W, Li L, Chen J, Li D, Hou J, Guo H, Shen J (2018) Long-term crowding stress causes compromised nonspecific immunity and increases apoptosis of spleen in grass carp (Ctenopharyngodon idella). Fish Shellfish Immunol 80:540–545
MacKintosh C, Beattie KA, Klumpp S, Cohen P, Codd GA (1990) Cyanobacterial microcystin-LR is a potent and specific inhibitor of protein phosphatases 1 and 2A from both mammals and higher plants. FEBS Lett 264:187–192
Menke AL, Spitsbergen JM, Wolterbeek AP, Woutersen RA (2011) Normal anatomy and histology of the adult zebrafish. Toxicol Pathol 39:759–775
Nobre AC, Martins AM, Havt A, Benevides C, Lima AA, Fonteles MC, Monteiro HS (2003) Renal effects of supernatant from rat peritoneal macrophages activated by microcystin-LR: role protein mediators. Toxicon 41:377–381
Ola MS, Nawaz M, Ahsan H (2011) Role of Bcl-2 family proteins and caspases in the regulation of apoptosis. Mol Cell Biochem 351:41–58
Oyadomari S, Araki E, Mori M (2002) Endoplasmic reticulum stress-mediated apoptosis in pancreatic beta-cells. Apoptosis 7:335–345
Puddick J, Prinsep MR, Wood SA, Kaufononga SA, Cary SC, Hamilton DP (2014) High levels of structural diversity observed in microcystins from Microcystis CAWBG11 and characterization of six new microcystin congeners. Mar Drugs 12:5372–5395
Qi M, Dang Y, Xu Q, Yu L, Liu C, Yuan Y, Wang J (2016) Microcystin-LR induced developmental toxicity and apoptosis in zebrafish (Danio rerio) larvae by activation of ER stress response. Chemosphere 157:166–173
Qiao Q, Liu W, Wu K, Song T, Hu J, Huang X, Wen J, Chen L, Zhang X (2013) Female zebrafish (Danio rerio) are more vulnerable than males to microcystin-LR exposure, without exhibiting estrogenic effects. Aquat Toxicol 142-143:272–282
Qin W, Xu L, Zhang X, Wang Y, Meng X, Miao A, Yang L (2010) Endoplasmic reticulum stress in murine liver and kidney exposed to microcystin-LR. Toxicon 56:1334–1341
Sarkar S, Mukherjee S, Chattopadhyay A, Bhattacharya S (2017) Differential modulation of cellular antioxidant status in zebrafish liver and kidney exposed to low dose arsenic trioxide. Ecotoxicol Environ Saf 135:173–182
Singh NP, Ogburn CE, Wolf NS, van Belle G, Martin GM (2001) DNA double-strand breaks in mouse kidney cells with age. Biogerontology 2:261–270
Svircev Z, Lujic J, Marinovic Z, Drobac D, Tokodi N, Stojiljkovic B, Meriluoto J (2015) Toxicopathology induced by microcystins and nodularin: a histopathological review. J Environ Sci Health C Environ Carcinog Ecotoxicol Rev 33:125–167
Wang Z, Li G, Wu Q, Liu C, Shen J, Yan W (2019) Microcystin-LR exposure induced nephrotoxicity by triggering apoptosis in female zebrafish. Chemosphere 214:598–605
Wei L, Liu Y, Zhong S, Wu H, Ruan J, Liu M, Zhou Q, Zhong Q (2018) Transcriptome analysis of grass carp provides insights into the immune-related genes and pathways in response to MC-LR induction. Aquaculture 488:207–216
Wei L, He L, Fu J, Liu Y, Ruan J, Liu L, Zhong Q (2019) Molecular characterization of caspase-8-like and its expression induced by microcystin-LR in grass carp (Ctenopharygodon idella). Fish Shellfish Immunol 89:727–735
Zhang J, Song W, Sun Y, Shan A (2017) Effects of phoxim-induced hepatotoxicity on SD rats and the protection of vitamin E. Environ Sci Pollut Res Int 24:24916–24927
Funding
This research was funded by the National Natural Science Foundation of China (project nos. 31760764 and 31460146).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Fish care and experimental protocols were performed in accordance with the guidelines for the Care and Use of Research Animals by the Institutional Animal Care and Use Committee of China.
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
He, L., Liu, L., Lin, C. et al. Effects of MC-LR on histological structure and cell apoptosis in the kidney of grass carp (Ctenopharyngodon idella). Fish Physiol Biochem 46, 2005–2014 (2020). https://doi.org/10.1007/s10695-020-00833-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10695-020-00833-5