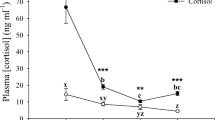
Abstract
This study aims to examine ionoregulatory parameters during exercise and cortisol elevation in common carp fed different food rations. Fish subjected to two different feeding regimes (0.5 or 3.0% body mass (BM) daily) received no implant or an intraperitoneal cortisol implant (250 mg/kg BM) or sham, and were monitored over a 168-h post-implant (PI) period under resting, low aerobic swimming or exhaustive swimming conditions. Plasma osmolality was maintained at relatively stable levels without much influence of feeding, swimming or cortisol, especially in low feeding groups. Nevertheless, a transient hyponatremia was observed in all low feeding fish implanted with cortisol. The hyponatremia was more pronounced in fish swum to exhaustion but even in this group, Na+ levels returned to control levels as cortisol levels recovered (168 h-PI). Cortisol-implanted fish also had lower plasma Cl− levels, and this loss of plasma Cl− was more prominent in fish fed a high ration during exhaustive swimming (recovered at 168 h-PI). Cortisol stimulated branchial NKA and H+ ATPase activities, especially in high ration fish. In contrast, low ration fish upregulated kidney NKA and H+ ATPase activities when experiencing elevated levels of cortisol. In conclusion, low feeding fish experience an ionoregulatory disturbance in response to cortisol implantation especially when swum to exhaustion in contrast to high feeding fish.




Similar content being viewed by others
References
Babitha GS, Peter MCS (2010) Cortisol promotes and integrates the osmotic competence of the organs in North African catfish (Clarias gariepinus Burchell): evidence from in vivo and in situ approaches. Gen Comp Endocrinol 168(1):14–21. https://doi.org/10.1016/j.ygcen.2010.03.021
Beaumont MW, Taylor EW, Butler PJ (2000) Exposure of brown trout, Salmo trutta, to a sublethal concentration of copper in soft acidic water: effects upon muscle metabolism and membrane potential. Aquat Toxicol 51(2):259–272. https://doi.org/10.1016/S0166-445X(00)00109-0
Bradford MM (1976) A rapid and sensitive method for the quantization of microgram quantities utilizing the principle of protein dye binding. Anal Biochem 72(1–2):248–254
Bucking C, Wood CM (2006a) Gastrointestinal processing of Na+, Cl-, and K+ during digestion: implications for homeostatic balance in freshwater rainbow trout. Am J Physiol Regul Integr Comp Physiol 291(6):1764–1772. https://doi.org/10.1152/ajpregu.00224.2006
Bucking C, Wood CM (2006b) Water dynamics in the digestive tract of the freshwater rainbow trout during the processing of a single meal. J Exp Biol 209:1883–1893. https://doi.org/10.1242/jeb.02205
Bucking C, Wood CM (2008) The alkaline tide and ammonia excretion after voluntary feeding in freshwater rainbow trout. J Exp Biol 211:2533–2541. https://doi.org/10.1242/jeb.015610
Bucking C, Wood CM (2012) Digestion of a single meal affects gene expression of ion and ammonia transporters and glutamine synthetase activity in the gastrointestinal tract of freshwater rainbow trout. J Comp Physiol B 182(3):341–350. https://doi.org/10.1007/s00360-011-0622-y
Bucking C, Landman MJ, Wood CM (2010) The role of the kidney in compensating the alkaline tide, electrolyte load, and fluid balance disturbance associated with feeding in the freshwater rainbow trout, Oncorhynchus mykiss. Comp Biochem and Physiol A: Mol Integr Physiol 156(1):74–83. https://doi.org/10.1016/j.cbpa.2009.12.021
Bui P, Bagherie-Lachidan M, Kelly SP (2010) Cortisol differentially alters claudin isoforms in cultured puffer fish gill epithelia. Mol Cell Endocrionol 317(1–2):120–126. https://doi.org/10.1016/j.mce.2009.12.002
Busk M, Jensen FB, Wang T (2000) Effects of feeding on metabolism, gas transport and acid-base balance in the bullfrog Rana catesbeiana. Am J Physiol Regul Integr Comp Physiol 278:R185–R195. https://doi.org/10.1152/ajpregu.2000.278.1.R185
Chasiotis H, Kelly SP (2012) Effects of elevated circulating cortisol levels on hydromineral status and gill tight junction protein abundance in the stenohaline goldfish. Gen Comp Endocrinol 175(2):277–283. https://doi.org/10.1016/j.ygcen.2011.11.024
Cooper CA, Wilson RW (2008) Post-prandial alkaline tide in freshwater rainbow trout: effects of meal anticipation on recovery from acid–base and ion regulatory disturbances. J Exp Biol 211(15):2542–2550. https://doi.org/10.1242/jeb.015586
Dang ZC, Lock RAC, Flik G, Wendelaar Bonga SE (2000) Na+, K+-ATPase immunoreactivity in branchial chloride cells of Oreochromis mossambicus exposed to copper. J Exp Biol 203(2):379–387
De Boeck G, Alsop D, Wood CM (2001a) Cortisol effects on aerobic and anaerobic metabolism, nitrogen excretion, and whole-body composition in juvenile rainbow trout. Physiol Biochem Zool 74(6):858–868. https://doi.org/10.1086/323796
De Boeck G, Vleaminck A, Balm PHM, Lock RAC, Wachter BD, Blust R (2001b) Morphological and metabolic changes in common carp, Cyprinus carpio, during short-term copper exposure: interactions between Cu2+ and plasma cortisol elevation. Environ Toxicol Chem 20(2):378–381. https://doi.org/10.1002/etc.5620200219
De Boeck G, van der Ven K, Hattink J, Blust R (2006) Swimming performance and energy metabolism of rainbow trout, common carp and gibel carp respond differently to sublethal copper exposure. Aquat Toxicol 80(1):92–100. https://doi.org/10.1016/j.aquatox.2006.07.017
Eckert SM, Yada T, Shepherd BS, Stetson MH, Hirano T, Grau EG (2001) Hormonal control of osmoregulation in the channel catfish Ictalurus punctatus. Gen Comp Endocrinol 122(2):270–286. https://doi.org/10.1006/gcen.2001.7633
Evans DH, Piermarini PM, Choe KP (2005) The multifunctional fish gill: dominant site of gas exchange, osmoregulation, acid-base regulation, and excretion of nitrogenous waste. Physiol Rev 85(1):97–177. https://doi.org/10.1152/physrev.00050.2003
Gallaugher PE, Thorarensen H, Kiessling A, Farrell AP (2001) Effects of high intensity exercise training on cardiovascular function, oxygen uptake, internal oxygen transport and osmotic balance in Chinook salmon (Oncorhynchus tshawytscha) during critical speed swimming. J Exp Biol 204(16):2861–2872
Gonzalez RJ, McDonald DG (1992) The relationship between oxygen consumption and ion loss in a freshwater fish. J Exp Biol 163(1):317–332
Gonzalez RJ, McDonald DG (1994) The relationship between oxygen uptake and ion loss in fish from diverse habitat. J Exp Biol 190(1):95–108
Goss GG, Wood CM (1990) Na+ and Cl− uptake kinetics, diffusive effluxes and acidic equivalent fluxes across the gills of rainbow trout: I. responses to environmental hyperoxia. J Exp Biol 152(1):521–547
Gregory TR, Wood CM (1999) The effects of chronic plasma cortisol elevation on the feeding behavior, growth, competition ability, and swimming performance of juvenile rainbow trout. Physiol Biochem Zool 72(3):286–295. https://doi.org/10.1086/316673
Heuer RM, Grosell M (2014) Physiological impacts of elevated carbon dioxide and ocean acidification on fish. Am J Physiol Regul Integr Comp Physiol 307:R1061–R1084. https://doi.org/10.1152/ajpregu.00064.2014
Holk K, Lykkeboe G (1998) The impact of endurance training on arterial plasma K+ levels and swimming performance of rainbow trout. J Exp Biol 210:1373–1380
Jones DR, Randall DJ (1978) The respiratory and circulatory and system during exercise. In: Hoar WS, Randall DJ (eds) Fish physiology, vol 7. Academic Press, New York, pp 425–501
Kiela PR, Ghishan FK (2016) Physiology of intestinal absorption and secretion. Best Pract Res Clin Gastroenterol 30(2):145–159. https://doi.org/10.1016/j.bpg.2016.02.007
Lall SP (2002) The minerals. In: Halver JE, Hardy RW (eds) Fish nutrition. Academic Press, New York, pp 261–307. https://doi.org/10.1016/B978-012319652-1/50006-9
Liew HJ, Sinha AK, Mauro N, Diricx M, Blust R, De Boeck G (2012) Fasting goldfish, Carassius auratus, and common carp, Cyprinus carpio, use different metabolic strategies when swimming. Comp BiochemPhysiol A: Mol Integr Physiol 163(3):327–335. https://doi.org/10.1016/j.cbpa.2012.07.012
Liew HJ, Sinha AK, Mauro N, Diricx M, Darras VM, Blust R, De Boeck G (2013a) Feeding and swimming modulate iono-and-hormonal regulation differently in goldfish Carassius auratus and common carp, Cyprinus carpio. Comp Biochem Physiol A: Mol Integr Physiol 165(1):13–21. https://doi.org/10.1016/j.cbpa.2013.02.009
Liew HJ, Chiarella D, Pelle A, Faggio C, Blust R, De Boeck G (2013b) Cortisol emphasizes the metabolic strategies employed by common carp, Cyprinus carpio at different feeding and swimming regimes. Comp Biochem Physiol A Mol Integr Physiol 166(3):449–464. https://doi.org/10.1016/j.cbpa.2013.07.029
Liew HJ, Fazio A, Faggio C, Blust R, De Boeck G (2015) Cortisol affects metabolic and ionoregulatory responses to a different extent depending on feeding ration in common carp, Cyprinus carpio. Comp Biochem Physiol A Mol Integr Physiol 189:45–57. https://doi.org/10.1016/j.cbpa.2015.07.011
Lin H, Randall DJ (1993) H+-ATPase activity in crude homogenates of fish gill tissue: inhibitor sensitivity and environmental and hormonal regulation. J Exp Biol 180(1):163–174
Lin H, Randall DJ (1995) Proton pumps in fish gills. In: Wood CW, Shuttleworth TJ (eds) Cellular and molecular approaches to fish ionic regulation. Academic Press, New York, pp 229–255
Lovell T (2013) Digestion and metabolism. In: Nutrition and Feeding of Fish. No. 2, XI, Springer US. pp 73–79. https://doi.org/10.1007/978-1-4615-4909-3
Marshall WS, Grosell M (2005) Ion transport and osmoregulation in fish. In: The Physiology of Fishes. D. Evans, (ed.) CRC Press, Boca Raton, Florida, 177–230
McCormick SD (1993) Methods for nonlethal gill biopsy and measurements of Na+, K+ -ATPase activity. Can J Fish Aquat Sci 50(3):656–658
McCormick SD, Regish A, O’Dea MF, Shrimpton JM (2008) Are we missing a mineralocorticoid in teleost fish? Effects of cortisol, deoxycorticosterone and aldosterone on osmoregulation, gill Na+, K+-ATPase activity and isoform mRNA levels in Atlantic salmon. Gen Comp Endocrinol 157(1):35–40. https://doi.org/10.1016/j.ygcen.2008.03.024
McDonald MD, Wood CM (2004) The effect of chronic cortisol elevation on urea metabolism and excretion in the rainbow trout (Oncorhynchus mykiss). J Comp Physiol B 174(1):71–81. https://doi.org/10.1007/s00360-003-0390-4
McGuire A, Aluru N, Takemura A, Weil R, Wilson JM, Vijayan MM (2010) Hyperosmotic shock adaptation by cortisol involves upregulation of branchial osmotic stress transcription factor 1 gene expression in Mozambique Tilapia. Gen Comp Endocrinol 165(2):321–329. https://doi.org/10.1016/j.ygcen.2009.07.016
McKenzie DJ, Shingles A, Taylor EW (2003) Sub-lethal plasma ammonia accumulation and the exercise performance of salmonids. Comp Biochem Physiol A 135(4):515–526. https://doi.org/10.1016/S1095-6433(03)00116-8
Metz JR, van den Burg EH, Wendelaar Bonga SE, Flik G (2003) Regulation of branchial Na+/K+-ATPase in common carp Cyprinus carpio L. acclimated to different temperatures. J Exp Biol 206(12):2273–2280. https://doi.org/10.1242/jeb.00421
Mommosen TP, Vijayan MM, Moon TW (1999) Cortisol in teleosts: dynamics, mechanisms of action and metabolic regulation. Rev Fish Biol Fish 9(3):211–268. https://doi.org/10.1023/A:1008924418720
Nawata CM, Hung CCY, Tsui TKN, Wilson JM, Wright PA, Wood CM (2007) Ammonia excretion in rainbow trout (Oncorhynchus mykiss): evidence for Rh glycoprotein and H+-ATPase involvement. Physiol Genomics 31(3):463–474. https://doi.org/10.1152/physiolgenomics.00061.2007
Nawata CM, Wood CM, O’Donnell MJ (2010) Functional characterization of Rhesus glycoproteins from an ammoniotelic teleost, the rainbow trout, using oocyte expression and SIET analysis. J Exp Biol 213(7):1049–1050. https://doi.org/10.1242/jeb.038752
Nilsson GE (2007) Gill remodeling in fish: a new fashion or an ancient secret? J Exp Biol 201(14):2403–2409. https://doi.org/10.1242/jeb.000281
Overli O, Kotzian S, Winberg S (2002) Effects of cortisol on aggression and locomotor activity in rainbow trout. Horm Behav 42(1):53–61. https://doi.org/10.1006/hbeh.2002.1796
Parks SK, Tresguerres M, Galvez F, Goss GG (2010) Intracellular pH regulation in isolated trout gill mitochondrion-rich cell (MR) subtypes: evidence for Na+/K+ activity. Comp Biochem Physiol A 155(2):139–145. https://doi.org/10.1016/j.cbpa.2009.10.025
Perry SF, Fryer JN (1997) Proton pumps in the fish gill and kidney. Fish Physiol Biochem 17(1-6):363–369. https://doi.org/10.1023/A:1007746217349
Perry SF, Goss GG (1994) The effects of experimentally altered gill chloride cell surface on acid-base regulation in rainbow trout during metabolic alkalosis. J Comp Physiol B (4):164, 327–336. https://doi.org/10.1007/BF00346451
Perry SF, Rivero-Lopez L, McNeill B, Wilson J (2006) Fooling a freshwater fish: how dietary salt transforms the rainbow trout gill into a seawater gill phenotype. J Exp Biol 209(23):4591–4596. https://doi.org/10.1242/jeb.02558
Postlethwaite EK, McDonald DG (1995) Mechanisms of Na+ and Cl- regulation in freshwater-adapted rainbow trout (Oncorhynchus mykiss) during exercise and stress. J Exp Biol 198(2):295–304
Potts WTW, McWilliams PG (1989) The effects of hydrogen and aluminium ions on fish gills. Acid Toxicity Aquat Anim:201–220. https://doi.org/10.1017/CBO9780511983344.013
Qin Z, Lewis JF, Perry SF (2010) Zebrafish (Danio rerio) gill neuroepithelial cells are sensitive chemoreceptors for environmental CO2. J Physiol 588:861–872. https://doi.org/10.1113/jphysiol.2009.184739
Salama A, Morgan IJ, Wood CM (1999) The linkage between Na+ uptake and ammonia excretion in rainbow trout: kinetic analysis, the effects of (NH4)2SO4 and NH4HCO3 infusion and the influence of gill boundary layer pH. J Exp Biol 202(6):697–709
Salman NA, Eddy FB (1987) Response of chloride cell numbers and gill Na+/K+ ATPase activity of freshwater rainbow trout (Salmo gairdneri Richardson) to salt feeding. Aquaculture 61(1):41–48. https://doi.org/10.1016/0044-8486(87)90336-X
Salman NA, Eddy FB (1988) Kidney function in response to salt feeding in rainbow trout (Salmo gairdneri Richardson). Comp Biochem Physiol A: Comp Physiol 89(4):535–539. https://doi.org/10.1016/0300-9629(88)90829-8
Salman NA, Eddy FB (1990) Increased sea-water adaptability of non-smolting rainbow trout by salt feeding. Aquaculture 86(2-3):259–270. https://doi.org/10.1016/0044-8486(90)90118-7
Sherwani FA, Parwez I (2008) Plasma thyroxine and cortisol profiles and gill and kidney Na+/K+-ATPase and SDH activities during acclimation of the catfish Heteropneustes fossilis (bloch) to higher salinity, with special reference to the effects of exogenous cortisol on hypo-osmoregulatory ability of the catfish. Zool Sci 25(2):164–171. https://doi.org/10.2108/zsj.25.164
Sinha AK, Liew HJ, Diricx M, Blust R, De Boeck G (2012) The interactive effects of ammonia exposure, nutritional status and exercise on metabolism and physiological responses in gold fish (Carrasisus auratus L.). Aquat Toxicol 109:33–46. https://doi.org/10.1016/j.aquatox.2011.11.002
Sinha AK, Liew HJ, Nawata CM, Blust R, Wood CM, De Boeck G (2013) Modulation of Rh glycoproteins, ammonia excretion and Na+ fluxes in three freshwater teleosts when exposed chronically to high environmental ammonia. J Exp Biol 216:2917–2930. https://doi.org/10.1242/jeb.084574
Sloman KA, Desforges PR, Gilmour KM (2001) Evidence for a mineralocorticoid-like receptor linked to branchial chloride cell proliferation in freshwater rainbow trout. J Exp Biol 204(22):3953–3961
Takahashi H, Sakamota T, Hyodo S, Shepherd BS, Kaneko T, Grau EG (2006) Expression of glucocorticoid receptor in the intestine of euryhaline teleost, the Mozambique tilapia (Oreochromis mossambicus): effect of seawater exposure and cortisol treatment. Life Sci 78(20):2329–2335. https://doi.org/10.1016/j.lfs.2005.09.050
Tudorache C, Viaenen P, Blust R, De Boeck G (2007) Longer flumes increase critical swimming speeds by increasing burst-glide swimming duration in carp Cyprinus carpio, L. J Fish Biol 71(6):1630–1638. https://doi.org/10.1111/j.1095-8649.2007.01620.x
Twitchen ID (1990) The physiological bases of resistance to low pH among aquatic insect larvae. The surface waters acidification Programme.pp 413-419
Uchida K, Kaneko T, Tagawa M, Hirano T (1998) Localisation of cortisol receptor in branchial chloride cells in chum salmon fry. Gen Comp Endocrinol 109(2):175–185. https://doi.org/10.1006/gcen.1997.7019
Umminger BL, Gist DH (1973) Effects of thermal acclimation on physiological responses to handling stress, cortisol and aldosterone injections in the goldfish, Carassius auratus. Comp Biochem Physiol A Physiol 44(3):967–977. https://doi.org/10.1016/0300-9629(73)90161-8
Van Dijk PLM, Van den Thillart GEEJM, Wendelaar Bonga SE (1993) Is there a synergistic effect between steady-state exercise and water acidification in carp? J Fish Biol 42(5):673–681. https://doi.org/10.1111/j.1095-8649.1993.tb00376.x
Veillette PA, Young G (2005) Tissue culture of sockeye salmon intestine: functional response of Na+-K+-ATPase to cortisol. Am J Physiol Regul Integr Comp Physiol 288(6):R1598–R1605. https://doi.org/10.1152/ajpregu.00741.2004
Walsh PJ (1998) Nitrogen excretion and metabolism. In: Evan DH (ed) The physiology of fishes, 2nd edn. CRC Press, Boca Raton, pp 199–214
Wicks BJ, Joensen R, Tang Q, Randall DJ (2002) Swimming and ammonia toxicity in salmonids: the effect of sub lethal ammonia exposure on the swimming performance of coho salmon and the acute toxicity of ammonia in swimming and resting rainbow trout. Aquat Toxicol 59(1-2):55–69. https://doi.org/10.1016/S0166-445X(01)00236-3
Wood CM (1988) Acid-base and ionic exchanges at gills and kidney after exhaustive exercise in the rainbow trout. J Exp Biol 136(1):461–481
Wood CM (1991) Acid-base and ion balance, metabolism, and their interactions, after exhaustive exercise in fish. J Exp Biol 160:285–308
Wood CM, Bucking C (2011) The role of feeding in salt and water balance. In: Grosell M, Farrel AP, Brauner CJ (eds.) Fish physiology the multifunctional gut of fish, vol 30. Academic Press, pp 165–212
Wood CM, Randall DJ (1973a) The influence of swimming activity on sodium balance in the rainbow trout (Salmo gairdneri). J Comp Physiol 82(3):207–234. https://doi.org/10.1007/BF00694237
Wood CM, Randall DJ (1973b) Sodium balance in the rainbow trout (Salmo gairdneri) during extended exercise. J Comp Physiol 82(3):235–256. https://doi.org/10.1007/BF00694238
Wood CM, Randall DJ (1973c) The influence of swimming activity on water balance in the rainbow trout (Salmo gairdneri). J Comp Physiol A Neuroethol Sens Neural Behav Physiol 82(3):257–276. https://doi.org/10.1007/BF00694239
Wood CM, Iftkar FI, Scott GR, De Boeck G, Sloman KA, Matey V, Valdez Domingos FX, Duarte RM, Almeida-Val VM, Val AL (2009) Regulation of gill transcellular permeability and renal function during acute hypoxia in the Amazonian Oscar (Astronotus ocellatus): new angles to osmorespiratory compromise. J Exp Biol 212(12):1949–1964. https://doi.org/10.1242/jeb.028464
Wu SC, Horng JL, Liu ST, Hwang PP, Wen ZH, Lin CS, Lin LY (2010) Ammonium-dependent sodium uptake in mitochondrion-rich cells of medaka (Oryzias latipes) larvae. Am J Physiol Cell Physiol 298(2):237–250. https://doi.org/10.1152/ajpcell.00373.2009
Zhang W, Cao ZD, Peng JL, Chen BJ, Fu SJ (2010) The effects of dissolved oxygen level on the metabolic interaction between digestion and locomotion in juvenile southern catfish (Silurus meridionalis Chen). Comp Biochem Physiol A 157:212–219. https://doi.org/10.1016/j.cbpa.2010.06.184
Zhou B, Kelly SP, Ianowski JP, Wood CM (2003) Effects of cortisol and prolactin on Na+ and Cl- transport in cultured branchial epithelia from FW rainbow trout. Am J Physiol Regul Integr Comp Physiol 285(6):R1305–R1316. https://doi.org/10.1152/ajpregu.00704.2002
Zimmer AM, Nawata CM, Wood CM (2010) Physiological and molecular analysis of the interactive effects of feeding and high environmental ammonia on branchial ammonia excretion and Na+ uptake in freshwater rainbow trout. J Comp Physiol B 180(8):1191–1204. https://doi.org/10.1007/s00360-010-0488-4
Acknowledgements
The authors would like to thank Joosen S, Meas N and Van Den Bergh K for their technical assistance. The authors also would like to thank Wilson JM and Alsop D for their valuable comments and time in editing the final version of the manuscript.
Funding
This study was financially supported by Bijzonder Onderzoeksfonds (BOF-IWS) research grant to De Boeck G. Liew HJ’s scholarship was funded by the Malaysian Ministry of Higher Education and Universiti Malaysia Terengganu (UMT). Chiarella D was an Erasmus scholar funded by the Erasmus Placement Program.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
All experimental procedures were approved by the University of Antwerp Animal Care Committee according to the guidelines of the Federation of European Laboratory Animal Science Associations.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Liew, H.J., Pelle, A., Chiarella, D. et al. Common carp, Cyprinus carpio, prefer branchial ionoregulation at high feeding rates and kidney ionoregulation when food supply is limited: additional effects of cortisol and exercise. Fish Physiol Biochem 46, 451–469 (2020). https://doi.org/10.1007/s10695-019-00736-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10695-019-00736-0