Abstract
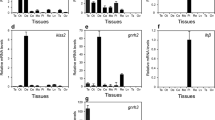
The kisspeptin–kisspeptin receptor (kissr)–gonadotropin-releasing hormone (GnRH) system plays a key role in regulating the onset of puberty in mammals. However, the role of this system in fish is still unclear. We examined the relative gene expression patterns for kiss1, kiss2, kissr2, sGnRH, and pjGnRH in all parts of the brains of Chinese sucker (Myxocyprinus asiaticus) females at the prepubertal and pubertal stages by using real-time PCR. We also analyzed the expression of kiss1 and GnRH1 via immunofluorescence. Two variants of kisspeptin; a variant of kissr (kissr2); and two variants of GnRH, pjGnRH (GnRH1), and sGnRH (GnRH3), were expressed in all parts of the brain. The mRNA expression of kiss1 was higher in the telencephalon, mesencephalon, and diencephalon at the pubertal stage than at the prepubertal stage, and the expression of kiss2 was higher in only the telencephalon. The expression of kissr2 was higher in all parts of the brain, except the medulla, at the pubertal stage than at the prepubertal stage. pjGnRH was highly expressed in all parts of the brain at the pubertal stage, whereas sGnRH expression showed no distinct changes, except in the epencephalon. Strong kiss1 and weak GnRH-1 immunoreactivity was observed in the pineal gland, lateral tuberal nucleus (NLT), and ventral part of the NLT in the diencephalon of the Chinese sucker females at the pubertal stage. Our results suggest that the kiss1–kissr2–pjGnRH system was expressed highly at the onset of pubertal female Chinese sucker.




Similar content being viewed by others
References
Akazome Y, Kanda S, Okubo K, Oka Y (2010) Functional and evolutionary insights into vertebrate kisspeptin systems from studies of fish brain. J Fish Biol 76:161–182. https://doi.org/10.1111/j.1095-8649.2009.02496.x
Alvarado MV, Carrillo M, Felip A (2013) Expression of kisspeptins and their receptors, gnrh-/gnrhr-II-1a and gonadotropin genes in the brain of adult male and female European sea bass during different gonadal stages. Gen Comp Endocrinol 187:104–116. https://doi.org/10.1016/j.ygcen.2013.03.030
Bernier NJ, Van Der Kraak G, Farrell AP, Brauner CJ (2009) Fish Neuroendocrinology. Amsterdam; London: Academic Press:8
Cortes-Campos C, Letelier J, Ceriani R, Whitlock KE (2015) Zebrafish adult-derived hypothalamic neurospheres generate gonadotropin-releasing hormone (GnRH) neurons. Biol Open 4:1077–1086. https://doi.org/10.1242/bio.010447
Escobar S, Servili A, Espigares F, Gueguen MM, Brocal I, Felip A, Gómez A, Carrillo M, Zanuy S, Kah O (2013) Expression of kisspeptins and kiss receptors suggests a large range of functions for kisspeptin systems in the brain of the European sea bass. PLoS One 8:e70177. https://doi.org/10.1371/journal.pone.0070177
Espigares F, Carrillo M, Gomez A, Zanuy S (2015) The forebrain-midbrain acts as functional endocrine signaling pathway of Kiss2/Gnrh1 system controlling the gonadotroph activity in the teleost fish European sea bass (Dicentrarchus labrax). Biol Reprod 92:70. https://doi.org/10.1095/biolreprod.114.125138
Faheem M, Jahan N, Khaliq S, Lone KP (2018) Modulation of brain kisspeptin expression after bisphenol-A exposure in a teleost fish, Catla catla. Fish Physiol Biochem 45:33–42. https://doi.org/10.1007/s10695-018-0532-y
Felip A, Espigares F, Zanuy S, Gomez A (2015) Differential activation of kiss receptors by Kiss1 and Kiss2 peptides in the sea bass. Reproduction 150:227–243. https://doi.org/10.1530/REP-15-0204
Fernald RD, White RB (1999) Gonadotropin-releasing hormone genes: phylogeny, structure, and functions. Front Neuroendocrinol 20:224–240. https://doi.org/10.1006/frne.1999.0181
Filby AL, van Aerle R, Duitman J, Tyler CR (2008) The kisspeptin/gonadotropin-releasing hormone pathway and molecular signaling of puberty in fish. Biol Reprod 78:278–289. https://doi.org/10.1095/biolreprod.107.063420
Gopurappilly R, Ogawa S, Parhar IS (2013) Functional significance of GnRH and kisspeptin, and their cognate receptors in teleost reproduction. Front Endocrinol (Lausanne) 4:24. https://doi.org/10.3389/fendo.2013.00024
Greives TJ, Humber SA, Goldstein AN, Scotti MA, Demas GE, Kriegsfeld LJ (2008) Photoperiod and testosterone interact to drive seasonal changes in kisspeptin expression in Siberian hamsters (Phodopus sungorus). J Neuroendocrinol 20:1339–1347. https://doi.org/10.1111/j.1365-2826.2008.01790.x
Harris PM, Mayden RL (2001) Phylogenetic relationships of major clades of Catostomidae (Teleostei : Cypriniformes) as inferred from mitochondrial SSU and LSU rDNA sequences. Mol Phylogenet Evol 20:225–237. https://doi.org/10.1006/mpev.2001.0980
Imamura S, Hur SP, Takeuchi Y, Bouchekioua S, Takemura A (2017) Molecular cloning of kisspeptin receptor genes (gpr54-1 and gpr54-2) and their expression profiles in the brain of a tropical damselfish during different gonadal stages. Comp Biochem Physiol A Mol Integr Physiol 203:9–16. https://doi.org/10.1016/j.cbpa.2016.07.015
Kanda S et al (2008) Identification of KiSS-1 product kisspeptin and steroid-sensitive sexually dimorphic kisspeptin neurons in medaka (oryzias latipes). Endocrinology 149:2467–2476. https://doi.org/10.1210/en.2007-1503
Lethimonier C, Madigou T, Munoz-Cueto JA, Lareyre JJ, Kah O (2004) Evolutionary aspects of GnRHs, GnRH neuronal systems and GnRH receptors in teleost fish. Gen Comp Endocrinol 135:1–16
Li S et al (2009) Structural and functional multiplicity of the kisspeptin/GPR54 system in goldfish (Carassius auratus). J Endocrinol 201:407–418. https://doi.org/10.1677/JOE-09-0016
Maruska KP, Tricas TC (2011) Gonadotropin-releasing hormone (GnRH) modulates auditory processing in the fish brain. Horm Behav 59:451–464. https://doi.org/10.1016/j.yhbeh.2011.01.003
Mechaly AS, Vinas J, Piferrer F (2011) Gene structure analysis of kisspeptin-2 (Kiss2) in the Senegalese sole (Solea senegalensis): characterization of two splice variants of Kiss2, and novel evidence for metabolic regulation of kisspeptin signaling in non-mammalian species. Mol Cell Endocrinol 339:14–24. https://doi.org/10.1016/j.mce.2011.03.004
Mechaly AS, Vinas J, Piferrer F (2013) The kisspeptin system genes in teleost fish, their structure and regulation, with particular attention to the situation in Pleuronectiformes. Gen Comp Endocrinol 188:258–268. https://doi.org/10.1016/j.ygcen.2013.04.010
Mohamed JS, Benninghoff AD, Holt GJ, Khan IA (2007) Developmental expression of the G protein-coupled receptor 54 and three GnRH mRNAs in the teleost fish cobia. J Mol Endocrinol 38:235–244. https://doi.org/10.1677/jme.1.02182
Nakajo M et al (2018) Evolutionally conserved function of kisspeptin neuronal system is nonreproductive regulation as revealed by nonmammalian study. Endocrinology 159:163–183. https://doi.org/10.1210/en.2017-00808
Nishiguchi R, Azuma M, Yokobori E, Uchiyama M, Matsuda K (2012) Gonadotropin-releasing hormone 2 suppresses food intake in the zebrafish, Danio rerio. Front Endocrinol (Lausanne) 3:122. https://doi.org/10.3389/fendo.2012.00122
Nocillado JN, Levavi-Sivan B, Carrick F, Elizur A (2007) Temporal expression of G-protein-coupled receptor 54 (GPR54), gonadotropin-releasing hormones (GnRH), and dopamine receptor D2 (drd2) in pubertal female grey mullet, Mugil cephalus. Gen Comp Endocrinol 150:278–287. https://doi.org/10.1016/j.ygcen.2006.09.008
Nocillado JN, Biran J, Lee YY, Levavi-Sivan B, Mechaly AS, Zohar Y, Elizur A (2012) The Kiss2 receptor (Kiss2r) gene in Southern Bluefin Tuna, Thunnus maccoyii and in Yellowtail Kingfish, Seriola lalandi - functional analysis and isolation of transcript variants. Mol Cell Endocrinol 362:211–220. https://doi.org/10.1016/j.mce.2012.06.024
Ogawa S, Parhar IS (2013) Anatomy of the kisspeptin systems in teleosts. Gen Comp Endocrinol 181:169–174. https://doi.org/10.1016/j.ygcen.2012.08.023
Ogawa S, Nathan FM, Parhar IS (2014) Habenular kisspeptin modulates fear in the zebrafish. Proc Natl Acad Sci U S A 111:3841–3846. https://doi.org/10.1073/pnas.1314184111
Ohga H et al (2015) mRNA levels of kisspeptins, kisspeptin receptors, and GnRH1 in the brain of chub mackerel during puberty. Comp Biochem Physiol A Mol Integr Physiol 179:104–112. https://doi.org/10.1016/j.cbpa.2014.09.012
Ohga H, Adachi H, Kitano H, Yamaguchi A, Matsuyama M (2017) Kiss1 hexadecapeptide directly regulates gonadotropin-releasing hormone 1 in the scombroid fish, chub mackerel. Biol Reprod 96:376–388. https://doi.org/10.1095/biolreprod.116.142083
Ohga H, Selvaraj S, Matsuyama M (2018) The roles of kisspeptin system in the reproductive physiology of fish with special reference to chub mackerel studies as main axis. Front Endocrinol (Lausanne) 9:147. https://doi.org/10.3389/fendo.2018.00147
Parhar IS, Ogawa S, Sakuma Y (2004) Laser-captured single digoxigenin-labeled neurons of gonadotropin-releasing hormone types reveal a novel G protein-coupled receptor (Gpr54) during maturation in cichlid fish. Endocrinology 145:3613–3618. https://doi.org/10.1210/en.2004-0395
Pasquier J, Lafont AG, Jeng SR, Morini M, Dirks R, van den Thillart G, Tomkiewicz J, Tostivint H, Chang CF, Rousseau K, Dufour S (2012a) Multiple kisspeptin receptors in early osteichthyans provide new insights into the evolution of this receptor family. PLoS One 7:e48931. https://doi.org/10.1371/journal.pone.0048931
Pasquier J, Lafont AG, Tostivint H, Vaudry H, Rousseau K, Dufour S (2012b) Comparative evolutionary histories of kisspeptins and kisspeptin receptors in vertebrates reveal both parallel and divergent features. Front Endocrinol (Lausanne) 3:173. https://doi.org/10.3389/fendo.2012.00173
Pasquier J, Kamech N, Lafont AG, Vaudry H, Rousseau K, Dufour S (2014) Molecular evolution of GPCRs: kisspeptin/kisspeptin receptors. J Mol Endocrinol 52:T101–T117. https://doi.org/10.1530/JME-13-0224
Pinilla L, Aguilar E, Dieguez C, Millar RP, Tena-Sempere M (2012) Kisspeptins and reproduction: physiological roles and regulatory mechanisms. Physiol Rev 92:1235–1316. https://doi.org/10.1152/physrev.00037.2010
Roch GJ, Busby ER, Sherwood NM (2014) GnRH receptors and peptides: skating backward. Gen Comp Endocrinol 209:118–134. https://doi.org/10.1016/j.ygcen.2014.07.025
Selvaraj S et al (2012) Increased expression of kisspeptin and GnRH forms in the brain of scombroid fish during final ovarian maturation and ovulation. Reprod Biol Endocrinol 10:64. https://doi.org/10.1186/1477-7827-10-64
Servili A et al (2011) Organization of two independent kisspeptin systems derived from evolutionary-ancient kiss genes in the brain of zebrafish. Endocrinology 152:1527–1540. https://doi.org/10.1210/en.2010-0948
Song Y, Duan X, Chen J, Huang W, Zhu Z, Hu W (2015) The distribution of kisspeptin (Kiss)1- and Kiss2-positive neurones and their connections with gonadotrophin-releasing hormone-3 neurones in the zebrafish brain. J Neuroendocrinol 27:198–211. https://doi.org/10.1111/jne.12251
Song H, Wang M, Wang Z, Liu J, Qi J, Zhang Q (2017) Characterization of kiss2 and kissr2 genes and the regulation of kisspeptin on the HPG axis in Cynoglossus semilaevis. Fish Physiol Biochem 43:731–753. https://doi.org/10.1007/s10695-016-0328-x
Su S, Fang F, Liu Y, Li Y, Ren C, Zhang Y, Zhang X (2013) The compensatory expression of reproductive hormone receptors in the thymus of the male rat following active immunization against GnRH. Gen Comp Endocrinol 185:57–66. https://doi.org/10.1016/j.ygcen.2013.01.013
Su S, Sun X, Zhou X, Fang F, Li Y (2015) Effects of GnRH immunization on the reproductive axis and thymulin. J Endocrinol 226:93–102. https://doi.org/10.1530/JOE-14-0720
Sugama N, Park JG, Park YJ, Takeuchi Y, Kim SJ, Takemura A (2008) Moonlight affects nocturnal Period2 transcript levels in the pineal gland of the reef fish Siganus guttatus. J Pineal Res 45:133–141. https://doi.org/10.1111/j.1600-079X.2008.00566.x
Tang H et al (2015) The kiss/kissr systems are dispensable for zebrafish reproduction: evidence from gene knockout studies. Endocrinology 156:589–599. https://doi.org/10.1210/en.2014-1204
Thakuria D, Shahi N, Singh AK, Khangembam VC, Kumar S (2017) Conformational analysis of a synthetic fish kisspeptin 1 peptide in membrane mimicking environments. PLoS One 12:e0185892. https://doi.org/10.1371/journal.pone.0185892
Volkoff H, Peter RE (1999) Actions of two forms of gonadotropin releasing hormone and a GnRH antagonist on spawning behavior of the goldfish Carassius auratus. Gen Comp Endocrinol 116:347–355. https://doi.org/10.1006/gcen.1999.7377
Wang S (1998) China red data book of endangered animals: pisces (in Chinese with English abstract). Science Press, Beijing, pp 1–247
Zmora N, Stubblefield J, Zulperi Z, Biran J, Levavi-Sivan B, Munoz-Cueto JA, Zohar Y (2012) Differential and gonad stage-dependent roles of kisspeptin1 and kisspeptin2 in reproduction in the modern teleosts, morone species. Biol Reprod 86:177. https://doi.org/10.1095/biolreprod.111.097667
Zmora N, Stubblefield J, Golan M, Servili A, Levavi-Sivan B, Zohar Y (2014) The medio-basal hypothalamus as a dynamic and plastic reproduction-related kisspeptin-gnrh-pituitary center in fish. Endocrinology 155:1874–1886. https://doi.org/10.1210/en.2013-1894
Zohar Y, Munoz-Cueto JA, Elizur A, Kah O (2010) Neuroendocrinology of reproduction in teleost fish. Gen Comp Endocrinol 165:438–455. https://doi.org/10.1016/j.ygcen.2009.04.017
Funding
The authors would like to thank Luoxin Li for assisting with animal care. This study was supported by the Natural Science Foundation of Anhui Province, China [grant number 1508085MC54], and Fishery Industrial System of Anhui Province, China [grant number [2016]84].
Author information
Authors and Affiliations
Contributions
Qingqing Li, Qiming Xie, Chaozhen Rong, and Tiantian Chen performed the sample assays and all the experiments. Shiping Su, Xilei Li, and Jun Zhang designed the experiments and analyzed the data. Shiping Su wrote the manuscript.
Corresponding author
Ethics declarations
Ethics statement
All experimental and fish maintenance procedures were conducted strictly in accordance with the guidelines set by the China Council on Animal Care.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Su, S., Li, Q., Li, X. et al. Expression of the kisspeptin/gonadotropin-releasing hormone (GnRH) system in the brain of female Chinese sucker ( Myxocyprinus asiaticus ) at the onset of puberty . Fish Physiol Biochem 46, 293–303 (2020). https://doi.org/10.1007/s10695-019-00717-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10695-019-00717-3