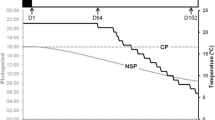
Abstract
Photoperiod and temperature are commonly accepted as the determinant factors for the control of the reproductive cycle in freshwater fishes. However, this determining effect is dependent on fish species. While applying a constant long photoperiod has an inhibitory effect in some species, the same photoperiodic manipulation has a stimulating effect in others. In cyprinids, a decrease in temperature or photoperiod can induce the gonad recrudescence. However, in roach Rutilus rutilus an early spring spawner cyprinid, there is little knowledge about the cueing role of each environmental factor. The aim of this work was to study the effect of a constant long photoperiod on the gametogenesis in roach. Fish were kept under either naturally simulated photoperiod or artificial constant long photoperiod and sampled at three times: at the beginning of photoperiod decrease, at the beginning of temperature decrease, and at the end of temperature decrease. Morphological parameters (gonado-somatic, hepato-somatic, and viscera-somatic indexes), plasma sexual steroids, and proportion of gametogenesis stages were estimated at each sampling time. The results showed that a constant, long photoperiod exerted inhibitory effects on gametogenesis advancement in both females and males that could stem from decrease of sex steroid production. Roach displayed a similar response to photoperiodic manipulations to other early spring spawners like percids, such as European perch, yellow perch and pikeperch. These results clearly showed the cueing role of the photoperiod in the induction of the reproductive cycle in roach.






Similar content being viewed by others
References
Amer MA, Miura T, Miura C, Yamauchi K (2001) Involvement of sex steroid hormones in the early stages of spermatogenesis in Japanese huchen (Hucho perryi ). Biol Reprod 65:1057–1066. https://doi.org/10.1095/biolreprod65.4.1057
Atkinson S, Yoshioka M (2007) Endocrinology of reproduction, in: reproductive biology and phylogeny of Cetacea: whales, porpoises and Dolphins2. Science publishers, pp 171–192. https://doi.org/10.1201/b10257-3
Babin PJ, Carnevali O, Lubzens E, Schnider WJ (2007) Molecular aspects of oocyte vitallogenesis in fish. In: Babin PJ, Cerdà J, Lubzens E (eds) The fish oocyte: from basic studies to biotechnological applications. Springer, Netherlands, pp 39–78
Baggerman B (1980) Photoperiod and endogenous control of the annual reproductive cycle in teleost fishes. In: Ali MA (ed) Environmental physiology of fishes. Springer US, Boston, MA, pp 533–567. https://doi.org/10.1007/978-1-4899-3659-2_21
Bates D, Mächler M, Bolker B, Walker S (2014) Fitting linear mixed-effects models using lme4. J Stat Softw 67. https://doi.org/10.18637/jss.v067.i01
Bayarri MJ, Rodríguez L, Zanuy S, Madrid JA, Sánchez-Vázquez FJ, Carrillo M (2004) Effect of photoperiod manipulation on daily rhythms of melatonin and reproductive hormones in caged European sea bass (Dicentrarchus labrax). Gen Comp Endocrinol 136:72–81. https://doi.org/10.1023/B:FISH.0000030468.98771.c0
Ben Ammar I, Teletchea F, Milla S, Ndiaye WN, Ledoré Y, Missaoui H, Fontaine P (2015) Continuous lighting inhibits the onset of reproductive cycle in pikeperch males and females. Fish Physiol Biochem 41:345–356. https://doi.org/10.1007/s10695-014-9987-7
Ben Ammar I, Milla S, Missaoui H, Ledoré Y, Teletchea F, Fontaine P (2017) Does constant photoperiod inhibit the onset of the reproductive cycle in northern pike (Esox lucius) males? Fish Physiol Biochem:1–10. https://doi.org/10.1007/s10695-017-0435-3
Billard R, Fostier A, Weil C, Breton B (1982) Endocrine control of spermatogenesis in teleost fish. Can J Fish Aquat Sci 39:65–79. https://doi.org/10.1139/f82-009
Borg B (1994) Androgens in teleost fishes. Comp Biochem Physiol Part C Comp 109:219–245. https://doi.org/10.1016/0742-8413(94)00063-G
Borg B, Ekström P (1981) Gonadal effects of melatonin in the three-spined stickleback, Gasterosteus aculeatus L., during different seasons and photoperiods. Reprod Nutr Dev 21:919–927
Bornestaf C, Antonopoulou E, Mayer I, Borg B (1997) Effects of aromatase inhibitors on reproduction in male three-spined sticklebacks, Gasterosteus aculeatus, exposed to long and short photoperiods. Fish Physiol Biochem 16:419–423
Bromage N, Porter M, Randall C (2001) The environmental regulation of maturation in fermed finfish with special reference to the role of photoperiod and melatonin. Aquaculture 197:63–68
Cakić M, Lenhardt P (2002) Seasonal reproductive cycle of pike, Esox lucius L., from the river Danube. J Appl Ichthyol 18:7–13. https://doi.org/10.1046/j.1439-0426.2002.00285.x
Dabrowski K (1996) Reproductive physiology of yellow perch (Perca flavescens): environmental and endocrinological cues. J Appl 12:139–148. https://doi.org/10.1111/j.1439-0426.1996.tb00079.x
Davies PR, Hanyu I (1986) Effect of temperature and photoperiod on sexual maturation and spawning of the common carp I. under conditions of high temperature*. Aquac. Elsevier Sci. Publ. B.V 51, pp 277–288
Davies PR, Hanyu I, Furukawa K, Nomura’ M (1986). Effect of Temperature and Photoperiod on Sexual Maturation and Spawning of the Common Carp Ii. Under Conditions of Low Temperature*. Aquac. Elsevier Sci. Publ. B.V 52, 51–58. https://doi.org/10.1016/0044-8486(86)90107-9
Dey R, Bhattacharya S, Maitra SK (2005) Importance of photoperiods in the regulation of ovarian activities in Indian major carp Catla catla in an annual cycle. J Biol Rhythm 20:145–158. https://doi.org/10.1177/0748730404272925
Divers SL, McQuillan HJ, Matsubara H, Todo T, Lokman PM (2010) Effects of reproductive stage and 11-ketotestosterone on LPL mRNA levels in the ovary of the shortfinned eel. J Lipid Res 51:3250–3258. https://doi.org/10.1194/jlr.M009027
Escaffre A, Billard R (1976) Le cycle spermatogénétique du Gardon Rutilus rutilus. Cah du Lab Montereau 3:43–46
Fostier A, Jalabert B, Billard R, Breton B, Zohar Y (1983) The gonadal steroids. In: Hoar SW, Randall DJ, Donaldson EM (eds) Fish physiology, pp 277–372. https://doi.org/10.1016/S1546-5098(08)60291-5
Fox J, Weisberg S, Adler D, Bates D, Baud- G, Ellison S, Firth D, Friendly M, Gor- G., Graves S, Heiberger R, Laboissiere R, Mon- G, Murdoch D, Nilsson H, Ogle D, Rip- B (2014) Package ‘car’ (version 2.1-3), second. Ed, URL https://r-forge.r-project.org/projects/car/, http://CRAN.R-project.org/package=car, http://socserv.socsci.mcmaster.ca/jfox/Books/Companion/index.html. SAGE publications, Inc
Gagné F, Blaise C (2000) Organic alkali-labile phosphates in biological materials: a generic assay to detect Vitellogenin in biological tissues. Environ Toxicol 15:243–247. https://doi.org/10.1002/1522-7278(2000)15:3<243::AID-TOX9>3.0.CO;2-D
Geraudie P, Gerbron M, Hill E, Minier C (2010a) Roach (Rutilus rutilus) reproductive cycle: a study of biochemical and histological parameters in a low contaminated site. Fish Physiol Biochem 36:767–777. https://doi.org/10.1007/s10695-009-9351-5
Geraudie P, Gerbron M, Minier C (2010b) Seasonal variations and alterations of sex steroid levels during the reproductive cycle of male roach (Rutilus rutilus). Mar Environ Res 69:S53–S55. https://doi.org/10.1016/j.marenvres.2009.11.008
Gillet C, Quétin P (2006) Effect of temperature changes on the reproductive cycle of roach in Lake Geneva from 1983 to 2001. J Fish Biol 69:518–534. https://doi.org/10.1111/j.1095-8649.2006.01123.x
Henderson BA, Wong JL, Nepszy SJ (1996) Reproduction of walleye in Lake Erie: allocation of energy. Can J Fish Aquat Sci 53:127–133. https://doi.org/10.1139/f95-162
Hervé M (2015) R package ‘RVAideMemoire’: diverse basic statistical and graphical functions. Version: 0.9-45-2
Higgins JJ, Tashtoush S (1994) An aligned rank transform test for interaction. Nonlinear World 1:201–211
Imsland AK, Folkvord A, Jónsdóttir ÓDB, Stefansson SO (1997) Effects of exposure to extended photoperiods during the first winter on long-term growth and age at first maturity in turbot (Scophthalmus maximus). Aquaculture 159:125–141. https://doi.org/10.1016/S0044-8486(97)00152-X
Kestemont P (1990) Dynamic aspects of ovogenesis in an asynchronous fish, the gudgeon Gobio gobio L.(Teleostei, Cyprinidae), under controlled temperature and photoperiod conditions. Aquat Living Resour 3:61–74
Knapp R, Carlisle SL (2011) Testicular function and hormonal regulation in fishes, in: hormones and reproduction of vertebrates. Elsevier, pp 43–63
Koskimies-Soininen K, Nyberg H (1991) Effects of temperature and light on the glycolipids of Sphagnum fimbriatum. Phytochemistry 30:2529–2536. https://doi.org/10.1016/0031-9422(91)85094-G
Langeron M (1913) Précis de microscopie: technique, expérimentation, diagnostic. Libraires de l’académie de médecine, Paris
Le Menn F, Cerdà J, Babin PJ (2007) Ultrastructural aspects of the ontogeny and differentiation of ray-finned fish ovarian follicles, in: the fish oocyte. Springer, pp 1–37
Lenth RV (2016) Least-squares means: the R package lsmeans. J Stat Softw 69:1–33. https://doi.org/10.18637/jss.v069.i01
Lofts B, Pickford GE, Atz JW (1968) The effects of low temperature, and cortisol, on testicular regression in the hypophysectomized cyprinodont fish, Fundulus heteroclitus. Biol Bull 134:74–86
Lokman PM, George KAN, Divers SL, Algie M, Young G (2007) 11-Ketotestosterone and IGF-I increse the size of previttelogenic oocytes from shortfinned eel, Anguilla australis, in vitro. Reproduction 133:955–967. https://doi.org/10.1530/REP-06-0229
Lubzens E, Young G, Bobe J, Cerdà J (2010) Oogenesis in teleosts: how fish eggs are formed. Gen Comp Endocrinol 165:367–389. https://doi.org/10.1016/j.ygcen.2009.05.022
Lukšienè D, Svedang H (1997) A rewiew on fish reproduction with special reference to temperature anomalies. Fisk. Kustlaboratoriet 35 p
Malison JA, Procarione LS, Barry TP, Kapuscinski AR, Kayes TB (1994) Endocrine and gonadal changes during the annual reproductive cycle of the freshwater teleost, Stizostedion vitreum. Fish Physiol Biochem 13:473–484. https://doi.org/10.1007/BF00004330
Migaud H, Fontaine P, Kestemont P, Wang N, Brun-Bellut J (2004a) Influence of photoperiod on the onset of gonadogenesis in Eurasian perch Perca fluviatilis. Aquaculture 241:561–574. https://doi.org/10.1016/j.aquaculture.2004.07.031
Migaud H, Gardeur JN, Kestemont P, Fontaine P (2004b) Off-season spawning of Eurasian perch Perca fluviatilis. Aquac Int 12:87–102. https://doi.org/10.1023/B:AQUI.0000017190.15074.6c
Migaud H, Wang N, Gardeur JN, Fontaine P (2006) Influence of photoperiod on reproductive performances in Eurasian perch Perca fluviatilis. Aquaculture 252:385–393. https://doi.org/10.1016/j.aquaculture.2005.07.029
Migaud H, Davie A, Taylor JF (2010) Current knowledge on the photoneuroendocrine regulation of reproduction in temperate fish species. J Fish Biol 76:27–68. https://doi.org/10.1111/j.1095-8649.2009.02500.x
Milla S, Mandiki SNM, Hubermont P, Rougeot C, Mélard C, Kestemont P (2009) Ovarian steroidogenesis inhibition by constant photothermal conditions is caused by a lack of gonadotropin stimulation in Eurasian perch. Gen Comp Endocrinol 163:242–250. https://doi.org/10.1016/j.ygcen.2009.04.010
Miura C, Miura T (2011) Analysis of spermatogenesis using an eel model. Aqua-BioSci Monogr 4:105–129. https://doi.org/10.5047/absm.2011.00404.0105
Miura T, Yamauchi K, Takahashi H, Nagahama Y (1991) Hormonal induction of all stages of spermatogenesis in vitro in the male Japanese eel (Anguilla japonica). Proc Natl Acad Sci U S A 88:5774–5778. https://doi.org/10.1073/pnas.88.13.5774
Miura C, Higashino T, Miura T (2007) A progestin and an estrogen regulate early stages of oogenesis in fish. Biol Reprod 77:822–828
Mylonas CC, Fostier A, Zanuy S (2010) Broodstock management and hormonal manipulations of fish reproduction. Gen Comp Endocrinol 165:516–534. https://doi.org/10.1016/j.ygcen.2009.03.007
Pohlert T (2016) Calculate Pairwise Multiple Comparisons of Mean Rank Sums Version 31
Poncin P, Melard C, Philippart JC (1987) Use of temperature and photoperiod in the control of the reproduction of three European cyprinids, Barbus Barbus (L), Leuciscus Cephalus (L.) and Tinca Tinca (L), reared in captivity. Preliminary results. Bull. Français la Pêche la Piscic 304:1–12
R Development Core Team (2005) R: a language and environment for statistical computing. In: R Foundation for Statistical Computing, R Foundation for statistical computing. Austria, Vienna
Rinchard J, Kestemont P (1996) Comparative study of reproductive biology in single- and multiple-spawner cyprinid fish. I. Morphological and histological features. J Fish Biol 49:883–894. https://doi.org/10.1006/jfbi.1996.0219
Rohr DH, Lokman PM, Davie PS, Young G (2001) 11-Ketotestosterone induces silvering-related changes in immature female short-finned eels, Anguilla australis. Comp Biochem Physiol - A Mol Integr Physiol 130:701–714. https://doi.org/10.1016/S1095-6433(01)00402-0
Rosa HJD, Bryant MJ (2003) Seasonality of reproduction in sheep. Small Rumin Res 48:155–171. https://doi.org/10.1016/S0921-4488(03)00038-5
Schulz RW, Nóbrega RH (2011) Anatomy and histology of fish testis. In: Farrell AP, Cech JJ, Richards JG, Stevens ED (eds) Encyclopedia of fish physiology: from genome to environment. Academic Press / Elsevier, London, pp 616–626. https://doi.org/10.1016/B978-0-1237-4553-8.00246-X
Schulz RW, de França LR, Lareyre JJ, LeGac F, Chiarini-Garcia H, Nobrega RH, Miura T (2010) Spermatogenesis in fish. Gen Comp Endocrinol 165:390–411. https://doi.org/10.1016/j.ygcen.2009.02.013
Scott DBC (1979) Environmental timing and the control of reproduction in teleost fish, in: symposium of the Zoological Society of London, pp 105–132
Shewmon LN, Godwin JR, Murashige RS, Daniels HV, Losordo TM (2007) Environmental manipulation of growth and sexual maturation in yellow perch, Perca flavescens. J World Aquac Soc 38:383–394
Stanton G (1968) Colorimetric determination of inorganic phosphate in the presence of biological material and adenosine triphosphate. Anal Biochem 22:27–34
Sulistyo I, Fontaine P, Rinchard J, Gardeur JN, Migaud H, Capdeville B, Kestemont P (2000) Reproductive cycle and plasma levels of steroids in male Eurasian perch Perca fluviatilis. Aquat Living Resour 13:99–106. https://doi.org/10.1016/S0990-7440(00)00146-7
Sumpter JP (1990) Chp. 2. General concepts of seasonal reproduction. Reprod. Seas. Teleosts environ. Influ. 13–31
Tarkan AS (2006) Reproductive ecology of two cyprinid fishes in an oligotrophic lake near the southern limits of their distributions range. Ecol Freshw Fish 15:131–138. https://doi.org/10.1111/j.1600-0633.2006.00133.x
Teletchea F, Fontaine P (2014) Levels of domestication in fish: implications for the sustainable future of aquaculture. Fish Fish 15:181–195. https://doi.org/10.1111/faf.12006
Teletchea F, Fostier A, Kamler E, Gardeur JN, Le Bail PY, Jalabert B, Fontaine P (2009) Comparative analysis of reproductive traits in 65 freshwater fish species: application to the domestication of new fish species. Rev Fish Biol Fish 19:403–430. https://doi.org/10.1007/s11160-008-9102-1
Turner CL (1919) The seasonal cycle in the spermary of the perch. J Morphol 32:681–711. https://doi.org/10.1002/jmor.1050320307
Tyler CR, Jobling S (2008) Roach, sex, and gender-bending chemicals: the feminization of wild fish in English rivers. Bioscience 58:1051–1059
Villacorta PJ (2015) ART: aligned rank transform for nonparametric Foctorial analysis, R package version 1.0 1–5. https://doi.org/10.1145/1978942.1978963>
Wang N, Teletchea F, Kestemont P, Milla S, Fontaine P (2010) Photothermal control of the reproductive cycle in temperate fishes. Rev Aquac 2:209–222. https://doi.org/10.1111/j.1753-5131.2010.01037.x
Zhu D, Yang K, Gul Y, Song W, Zhang X, Wang W (2014) Effect of photoperiod on growth and gonadal development of juvenile Topmouth gudgeon Pseudorasbora parva. Environ Biol Fish 97:147–156. https://doi.org/10.1007/s10641-013-0133-7
Acknowledgments
We would like to thank all members of the AFPA research unit for their active collaboration in this study. We would also like to thank Julien Ruelle and Maryline Harroué from UMR 1092 AgroParisTech INRA Laboratoy of study of forest and wood resources “LERFoB”, technical platform xylosciences for the provision of the necessary equipment to realize the histological analyses (Technical Platform of functional ecogenomics, INRA Champenoux, France). We are thankful to Daniel Van Vlaender (Facultés Universitaires Notre-Dame de la Paix FUNDP, Belgium) for his help with histological protocols, Jos Fey (Faculty of Kinesiology and Rehabilitation Science, University of Leuven), Marie Fernandez (University of Saint Etienne, France) for their advices about statistics with R software, and Mark Holmes (University of Namur, Belgium) for the proofreading.
Funding
This study was partly funded by the Tunisian Ministry of Higher Education and Research.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(DOCX 3537 kb).
Rights and permissions
About this article
Cite this article
Ben Ammar, I., Milla, S., Ledoré, Y. et al. Constant long photoperiod inhibits the onset of the reproductive cycle in roach females and males. Fish Physiol Biochem 46, 89–102 (2020). https://doi.org/10.1007/s10695-019-00698-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10695-019-00698-3