Abstract
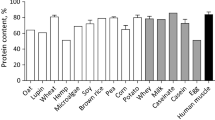
Muscle tissues contribute 34–48 % of the total body mass in fish. Proteomic analysis enables better understanding of the skeletal muscle physiology and metabolism. A proteome map reflects the general fingerprinting of the fish species and has the potential to identify novel proteins which could serve as biomarkers for many aspects of aquaculture including fish physiology and growth, flesh quality, food safety and aquatic environmental monitoring. The freshwater catfish Rita rita of the family Bagridae inhabiting the tropical rivers and estuaries is an important food fish with high nutritive value and is also considered a species of choice in riverine pollution monitoring. Omics information that could enhance utility of this species in molecular research is meager. Therefore, in the present study, proteomic analysis of Rita rita muscle has been carried out and functional genomics data have been generated. A reference muscle proteome has been developed, and 23 protein spots, representing 18 proteins, have been identified by MALDI-TOF/TOF-MS and LC-MS/MS. Besides, transcript information on a battery of heat shock proteins (Hsps) has been generated. The functional genomics information generated could act as the baseline data for further molecular research on this species.






Similar content being viewed by others
References
Addis MF, Cappuccinelli R, Tedde V, Pagnozzi D, Porcu MC, Bonaglini E, Roggio T, Uzzau S (2010) Proteomic analysis of muscle tissue from gilthead sea bream (Sparus aurata) farmed in offshore floating cages. Aquaculture 309:245–252
Al-Arabi SAM, Goksøyr A (2002) Cytochrome P4501A responses in two tropical fish species, riverine catfish (Rita rita) and marine mudfish (Apocryptes bato). Comp Biochem Physiol Part C Toxicol Pharmacol 131:61–71
Altschul SF, Gish W, Miller W, Myers EW, Lipman DJ (1990) Basic local alignment search tool. J Mol Biol 215:403–410
Barik SK, Banerjee S, Bhattacharjee S, Dasgupta S, Mohanty S, Mohanty BP (2013) Proteomic analysis of sarcoplasmic peptides of two related fish species for food authentication. Appl Biochem Biotechnol 171:1011–1021
Bhattacharjee S, Mohanty S, Sharma AP, Mohanty BP (2011) Effect of storage temperature as a preanalytical variable on the lens crystallins protein quality for proteomic studies. Proteomics Clin Appl 5:504–512
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Bringans S, Eriksen S, Kendrick T, Gopalakrishnakone P, Livk A, Lock R, Lipscombe R (2008) Proteomic analysis of the venom of Heterometrus longimanus (Asian black scorpion). Proteomics 8:1081–1096
Deane EE, Woo NYS (2005) Cloning and characterization of the hsp70 multigene family from silver Sea bream: modulation gene expression between warm and cold temperature acclimatization. Biochem Biophy Res Commun 330(3):776–783
Easton DP, Kaneko Y, Subjeck JR (2000) The Hsp110 and Grp170 stress proteins: newly “recognized relatives of the Hsp70s. Cell Stress Chaperones 5(4):276–290
Emami K, Askari V, Ullrich M, Mohinudeen K, Anil AC, Khandeparker L, Burgess JG, Mesbahi E (2012) Characterization of bacteria in ballast water using MALDI-TOF/TOF mass spectrometry. PLoS ONE 7:e38515
Godiksen H, Morzel M, Hyldig G, Jessen F (2009) Contribution of cathepsins B, L and D to muscle protein profiles correlated with texture in rainbow trout (Oncorhynchus mykiss). Food Chem 113:889–896
Hamilton AM, Heikkila JJ (2006) Examination of the stress-induced expression of the collagen binding heat shock protein, hsp47, in Xenopus laevis cultured cells and embryos. Comp Biochem Physiol 143(1):133–141
Laemmli UK (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227:680–685
Mann M, Hendrickson RC, Pandey A (2001) Analysis of proteins and proteomes by mass spectrometry. Annu Rev Biochem 70:437–473
Martin SAM, Mohanty BP, CashP Houlihan DF, Secombes CJ (2007) Proteome analysis of the Atlantic salmon (Salmosalar) cell line SHK-1 following recombinant IFN-γ stimulation. Proteomics 7:2275–2286
Mitra T, Ganguly S, Banerjee S, Mahanty A, Mohanty BP (2014) Nutrient profile of freshwater catfish Rita rita (Hamilton) from river Ganga. Book of Abstracts, The 10th Indian Fisheries and Aquaculture Forum (10ifaf) ICAR – National Bureau of Fish Genetic Resources, Lucknow, India 12–15 November 2014
Mohanty BP, Bhattacharjee S, Mondal K, Das MK (2010) HSP70 expression profiles in white muscles of riverine catfish Rita rita show promise as biomarker for pollution monitoring in tropical rivers. Natl Acad Sci Lett 33:177–182
Mohanty BP, Bhattacharjee S, Das MK (2011) Lens proteome map and α-crystallin profile of the catfish Rita rita. Ind J Biochem Biophys 48:35–41
Mohanty BP, Banerjee S, Bhattachrjee S, Mitra T, Purohit GK, Sharma AP, Karunakaran D, Mohanty S (2013a) Muscle proteomics of the Indian major carp catla (Catla catla, Hamilton). J Proteomics Bioinform 6:252–263
Mohanty BP, Bhattacharjee S, Paria P, Mahanty A, Sharma AP (2013b) Lipid biomarkers of lens ageing. Appl Biochem Biotechnol 169:192–200
Nguyen NH, Ponzoni RW, Hamzah A, Yee HY, Abu-Bakar KR, Khaw HL (2004) Genetics of flesh quality in fish. Fish Aqua 211:65–79
Ohlendieck K (2011) Skeletal muscle proteomics: current approaches, technical challenges and emerging techniques. Skelet Muscle 1:6
Picard B, Lefèvre F, Lebret B (2012) Meat and fish flesh quality improvement with proteomic applications. Animal Front 2:18–25
Porte C, Biosca X, Sole M, Albaiges J (2001) The integrated use of chemical analysis, cytochrome P450 and stress proteins in mussels to assess pollution along the Galician coast (NW Spain). Environ Pollut 112:261–268
Purohit GK, Mahanty A, Suar M, Sharma AP, Mohanty BP, Mohanty S (2014) Investigating hsp gene expression in liver of Channa striatus under heat stress for understanding the upper thermal acclimation. Biomed Res Int 2014:10
Roberts RJ, Agius C, Saliba C, Bossier P, Sung YY (2010) Heat shock proteins (chaperones) in fish and shellfish and their potential role in relation to fish health: a review. J Fish Dis 33:789–801
Terova G, Pisanu S, Roggio T, Preziosa E, Saroglia M, Addis MF (2014) Proteomic profiling of sea bass muscle by two-dimensional gel electrophoresis and tandem mass spectrometry. Fish Physiol Biochem 40(1):311–322
Tucker NR, Ustyugov A, Bryantsev AL, Konkel ME, Shelden EA (2009) Hsp27 is persistently expressed in zebrafish skeletaland cardiac muscle tissues but dispensable for their morphogenesis. Cell Stress Chaperones 14:521–533
Van Aggelen G, Ankley GT, Baldwin WS et al (2010) Integrating omic technologies into aquatic ecological risk assessment and environmental monitoring: hurdles, achievements, and future outlook. Environ Health Perspect 118:1–5
Veiseth-Kent E, Grove H, Faergestad EM, Fjaera SO (2010) Changes in muscle and blood plasma proteomes of Atlantic salmon (Salmo salar) induced by crowding. Aquaculture 309:272–279
Williams TL, Andrzejewski D, Lay JO, Musser SM (2003) Experimental factors affecting the quality and reproducibility of MALDI TOF mass spectra obtained from whole bacteria cells. J Am Soc Mass Spectrom 14:342. doi:10.1016/s1044-0305(03)00065-5
Wolfe RR (2006) The underappreciated role of muscle in health and disease. Am J Clin Nutr 84:475–482
Xu XY, Shen YB, Fu JJ, Liu F, GuoSZ Yang XM, Li JL (2011) Molecular cloning, characterization and expression patterns of HSP60 in the grass carp (Ctenopharyngodon idella). Fish Shellfish Imunol 31:864–870
Yarsan E, Yipel M (2013) The important terms of marine pollution “biomarkers and biomonitoring, Bioaccumulation, bioconcentration, biomagnification”. J Mol Biomark Diagn S1:003
Zhou Q, Zhang J, Fu J, Shi J, Jiang G (2008) Biomonitoring: an appealing tool for assessment of metal pollution in the aquatic ecosystem. Anal Chim Acta 606:135–150
Acknowledgments
This work was supported by the Indian Council of Agricultural Research, New Delhi, under CIFRI Core Project No. FHE/ER/07/06/005 to BPM. The authors are thankful to the Director, CIFRI, Barrackpore, for the facilities and encouragement. The authors wish to record their sincere appreciation to the unknown reviewers for meticulously reading the manuscript and offering suggestions that helped in substantially improving it.
Conflict of interest
The authors declare that they have no conflict of interests.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
10695_2015_46_MOESM1_ESM.pdf
ESM_1 The homology of mapped peptides with other species used for identification, the amino acid sequences of identified peptides and MALDI-TOF/TOF mass spectra of Rita rita (PDF 1378 kb)
10695_2015_46_MOESM2_ESM.xlsx
ESM_2 The homology of mapped peptides with other species used for identification, the amino acid sequences of identified peptides, i.e., Hsp90 sequence from LC-MS/MS (XLSX 117 kb)
10695_2015_46_MOESM3_ESM.xlsx
ESM_3 The homology of mapped peptides with other species used for identification, the amino acid sequences of identified peptides, i.e., α-actinin sequence from LC-MS/MS (XLSX 192 kb)
Rights and permissions
About this article
Cite this article
Mohanty, B.P., Mitra, T., Banerjee, S. et al. Proteomic profiling of white muscle from freshwater catfish Rita rita . Fish Physiol Biochem 41, 789–802 (2015). https://doi.org/10.1007/s10695-015-0046-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10695-015-0046-9