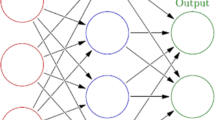
Neural networks are a universal tool used to investigate the dependences between the structure of organic compounds and a broad spectrum of their physicochemical properties. The potential of neural network modeling is not yet exhausted, as the increasing number of publications on their use indicates. Neural network models can solve both classification (for a discrete set of values of the modeled property) and regression problems (for continuous values of the modeled property). The reason for the popularity of neural network models in applied research is their clarity and the fact that no deep knowledge of mathematical statistics is required for their effective use.
Similar content being viewed by others
References
J. Zupan and J. Gasteiger, Neural Networks for Chemists, National Institute of Chemistry, Slovenia (1993).
J. Gasteiger, Computer-Chemie-Centrum and Institute for Organic Chemistry, University of Erlangen-N•rnberg, Germany.
R. Hecht-Nielsen, “Counterpropagation networks,” in: Proceedings of the IEEE First International Conference on Neural Networks, M. Caudill and C. Butler (eds.), Vol. 2, SOS Printing, San Diego, CA (1987), pp. 19–32.
R. Hecht-Nielsen, Appl. Optics, No. 26 (23), 4979–4984 (1987).
R. Hecht-Nielsen, Neural Networks, No. 1, 131–139 (1988).
R. Todeschini and V. Consonni, Handbook of Molecular Descriptors, Wiley-VCH, Weinheim (2000).
N. Majcen and J. Zupan, Anal. Chem. (Wash.) (Print ed.), 67, 2154–2161 (1995).
J. Gasteiger, “Neural networks with counterpropagation learning strategy used for modelling,” Chemometr. Intell. Lab. Syst. [Print ed.], 27, 175–187 (1995).
J. Zupan, “Counterpropagation learning strategy in neural networks and its application in chemistry. V,” in: Further Advances in Chemical Information, H. Collier (ed.), Special Publication No. 142, Royal Soc. Chem., Cambridge (1994), pp. 92–108.
M. Novic and M. Vracko, “Artificial neural networks in molecular structures — property studies. V, in: Nature-inspired Methods in Chemometrics: Genetic Algorithms and Artificial Neural Networks, (Data Handling in Science and Technology, 23), R. Leardi (ed.), Elsevier, Amsterdam (2003), pp. 231–256.
M. Vracko, Current Computer-Aided Drug Design [Print ed.], 90, No. 1, 84–91 (2008).
M. Vracko, SAR QSAR Environ. Res., 17, No. 3, 265–284 (2006).
I. Kuzmanovski and M. Novic, Chemometr. Intell. Lab. Syst. [Print ed.], 90, No. 1, 84–91 (2008).
M. Novic and J. Zupan, Vestn. Slov. Kem. Dru•. (Doc. Chem. Yugoslavica), 39, No. 2, 195–212 (1992) [COBISS.SI-ID 32664576].
J. Gasteiger, Li Xinzhi, V. Simon, et al., J. Mol. Struct. [Print ed.], 292, 141–160 (1993) [COBISS.SI-ID 842266].
M. Novic and J. Zupan, J. Chem. Inf. Comput. Sci., 35, 454–466 (1995).
D. Brodnjak-Voncina, D. Dobcnik, et al., Chemometr. Intell. Lab. Syst. [Print ed.], No. 47, 79–88 (1999) [COBISS.SI-ID 4318742].
Y. V. Heyden, P. Vankeerberghen, et al., Talanta (Oxford) [Print ed.], 51, 455–466 (2000) [COBISS.SI-ID 2130458].
J. Zupan, M. Novic, and I. Ruisanchez, Chemometr. Intell. Lab. Syst. [Print ed.], 38, 1–23 (1997).
M. Novic, “Kohonen and counterpropagation neural networks applied for mapping and interpretation of IR spectra, V,” in: Artificial Neural Networks: Methods and Applications, D. Livingstone (ed.), Humana Pres (2007).
J. Lozano, M. Novi_, et al., Chemometr. Intell. Lab. Syst. [Print ed.], 28, 61–72 (1995). Use of neural networks in gas chromatography presented in the work
M. Pompe, M. Razinger, et al., Anal. Chim. Acta [Print ed., 348, 215–221 (1997).
Author information
Authors and Affiliations
Additional information
Translated from Khimicheskie Volokna, No. 3, pp. 82–86, May–June, 2008.
Rights and permissions
About this article
Cite this article
Fedorova, N., Zupan, Y., Novic, M. et al. Neural network models for predicting the properties of chemical compounds. Fibre Chem 40, 281–286 (2008). https://doi.org/10.1007/s10692-008-9049-y
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10692-008-9049-y