Abstract
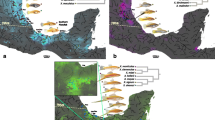
A fundamental goal of evolutionary ecology is understanding the processes responsible for contemporary patterns of morphological diversity and species richness. Transitions across the marine–freshwater interface are regarded as key triggers for adaptive radiation of many clades. Using the Australian terapontid fish family as a model system we employed phylogenetic analyses to compare the rates of ecological (dietary) and morphological evolution between marine and freshwater species of the family. Results suggested significantly higher rates of phenotypic evolution in key dietary and morphological characters in freshwater species compared to marine counterparts. Moreover, there was significant correlation between several of these dietary and morphological characters, suggesting an underlying ecomorphological aspect to these greater rates of phenotypic evolution in freshwater clades. Australia’s biogeographic history, which has precluded colonisation by many of the major ostariophysan fish families that make up much global freshwater fish diversity, appears to have provided the requisite ‘ecological opportunity’ to facilitate the radiation of invading marine-derived fish clades.





Similar content being viewed by others
References
Allen GR, Midgley SH, Allen M (2002) Field guide to the freshwater fishes of Australia. Western Australian Museum, Perth, WA
Barlow GW (2000) The cichlid fishes. Perseus Publishing, Cambridge, MA
Betancur-R R (2010) Molecular phylogenetics support multiple evolutionary transitions from marine to freshwater habitats in ariid catfishes. Mol Phylogenet Evol 55:249–258
Betancur-R R, Ortí G, Stein AM, Marceniuk AP, Pyron RA (2012) Apparent signal of competition limiting diversification after ecological transitions from marine to freshwater habitats. Ecol Lett 8:822–830
Bloom DD, Lovejoy NR (2012) Molecular phylogenetics reveals a pattern of biome conservatism in new world anchovies (family Engraulidae). J Evol Biol 25:701–715
Bloom DD, Weir JT, Piller KR, Lovejoy NR (2013) Do freshwater fishes diversify faster than marine fishes? A test using state-dependent diversification analyses and molecular phylogenetics of New World Silversides (Atherinopsidae). Evolution. doi:10.1111/evo.12074
Bollback JP (2006) SIMMAP: stochastic character mapping of discrete traits on phylogenies. BMC Bioinformatics 7:88
Burbrink FT, Ruane S, Pyron RA (2011) When are adaptive radiations replicated in areas? Ecological opportunity and unexceptional diversification in West Indian dipsadine snakes (Colubridae: Alsophiini). J Biogeogr 39:465–475
Burnham KP, Anderson DR (2002) Model selection and multimodel inference: a practical information theoretic approach, 2nd edn. Springer, New York
Burridge CP, Craw D, Waters JM (2006) River capture, range expansion, and cladogenesis: the genetic signature of freshwater vicariance. Evolution 60:1038–1049
Collar DC, O’Meara BC, Wainwright PC, Near TJ (2009) Piscivory limits diversification of feeding morphology in centrarchid fishes. Evolution 63:1557–1573
Davis AM, Pusey BJ, Thorburn DC, Dowe JL, Morgan DL, Burrows DW (2010) Riparian contributions to the diet of terapontid grunters (Pisces: Terapontidae) in wet-dry tropical rivers. J Fish Biol 76:862–879
Davis AM, Pearson RG, Pusey BJ, Perna C, Morgan DL, Burrows D (2011a) Trophic ecology of northern Australia’s terapontids: ontogenetic dietary shifts and feeding classification. J Fish Biol 78:265–286
Davis AM, Pusey BJ, Pearson RG (2011b) Trophic ecology of terapontid grunters: the role of morphology and ontogeny. Mar Freshw Res 63:128–141
Davis AM, Unmack PJ, Pusey BJ, Pearson RG (2012a) Marine-freshwater transitions are associated with the evolution of dietary diversification in terapontid grunters (Teleostei: Terapontidae). J Evol Biol 25:1163–1179
Davis AM, Blanchette ML, Pusey BJ, Pearson RG, Jardine TD (2012b) Gut-content and stable-isotope analyses provide complementary understanding of ontogenetic dietary shifts and trophic relationships among fishes in a tropical river. Freshw Biol 57:2156–2172
Davis AM, Unmack PJ, Pusey BJ, Pearson RG, Morgan DL (2013) Ontogenetic development of intestinal length and relationships to diet in an Australasian fish family (Terapontidae). BMC Evol Biol 13:53
Dray S, Dufour AB (2007) The ade4 package: implementing the duality diagram for ecologists. J Stat Softw 22:1–20
Drummond AJ, Rambaut A (2007) BEAST: Bayesian evolutionary analysis by sampling trees. BMC Evol Biol 7:214
Eschmeyer WN, Fong JD (2013) Species of Fishes by family/subfamily. Available from http://research.calacademy.org/research/ichthyology/catalog/SpeciesByFamily.asp. Accessed Jan 2013
Freckleton RP, Harvey PH, Pagel M (2002) Phylogenetic analysis and comparative data: a test and review of evidence. Am Nat 160:712–726
Freckleton RP, Harvey PH (2006) Detecting non-Brownian trait evolution in adaptive radiations. PLoS Biol 2006:2104–2111
Froese R, Pauly D (2000) FishBase 2000: concepts, design and data sources. ICLARM, Los Baños, Laguna
Froese R, Pauly D (eds) (2012) FishBase. World Wide Web electronic publication. www.fishbase.org, version (01/2013)
Garland T Jr, Dickerman AW, Janis CM, Jones JA (1993) Phylogenetic analysis of covariance by computer simulation. Syst Biol 42:265–292
Gavrilets S, Losos JB (2009) Adaptive radiation: contrasting theory with data. Science 323:732–737
Gillespie RG (2004) Community assembly through adaptive radiation in Hawaiian spiders. Science 303:356–359
Gonzalez I, Dejean S, Martin PGP, Baccini A (2008) CCA: an R package to extend canonical correlation analysis. J Stat Softw 23:1–14
Grafen A (1989) The phylogenetic regression. Philos Trans R Soc Lond B 326:119–157
Grant PR (1999) Ecology and evolution of Darwin’s finches. Princeton University Press, Princeton, NJ
Hansen TF, Martins EP (1996) Translating between microevolutionary process and macroevolutionary patterns: the correlation structure of interspecific data. Evolution 50:1404–1417
Harmon LJ, Weir JT, Brock CD, Glor RE, Challenger W (2008) GEIGER: investigating evolutionary radiations. Bioinformatics 24:129–131
Harmon LJ, Losos JB, Davies TJ, Gillespie RG, Gittleman JL, Jennings WB et al (2010) Early bursts of body size and shape evolution are rare in comparative data. Evolution 64:2385–2396
Hillis DM, Bull JJ (1993) An empirical test of bootstrapping as a method for assessing confidence in phylogenetic analysis. Syst Biol 42:182–192
Huelsenbeck JP, Nielsen R, Bollback JP (2003) Stochastic mapping of morphological characters. Syst Biol 52:131–158
Hugueny B, Pouilly M (1999) Morphological correlates of diet in an assemblage of West African freshwater fishes. J Fish Biol 54:1310–1325
Hurlbert SH (1978) The measurement of niche overlap and some relatives. Ecology 59:66–77
Hutcheon JM, Garland T (2004) Are megabats big? J Mamm Evol 22:257–277
Hyslop EJ (1980) Stomach content analysis—a review of methods and their application. J Fish Biol 17:411–429
Katoh K, Toh H (2010) Parallelization of the MAFFT multiple sequence alignment program. Bioinformatics 26:1899–1900
Kennard M, Pusey B, Arthington A (2001) Trophic ecology of freshwater fishes in Australia. Summary report CRC for freshwater ecology scoping study SCD6. CRC for Freshwater Ecology, Griffith University, Brisbane, Australia
Krebs CJ (1999) Ecological methodology, 2nd edn. Addison Wesley Longman, New York
Lee CE, Bell MA (1999) Causes and consequences of recent freshwater invasions by saltwater animals. Trends Ecol Evol 14:284–288
Lee CE, Posavi M, Charmantier G (2012) Rapid evolution of body fluid regulation following independent invasions into freshwater habitats. Evolution 25:625–633
Legendre P, Legendre L (1998) Numerical ecology. Elsevier Scientific, Amsterdam
Losos JB (2010) Adaptive radiation, ecological opportunity, and evolutionary determinism. Am Nat 175:623–639
Losos JB, Mahler DL (2010) The interaction of ecological opportunity, adaptation, and speciation. In: Bell M, Futuyma DJ, Eanes WF, Levinton JS (eds) Evolution since Darwin: the first 150 years, adaptive radiation. Sinauer, Sunderland, MA, pp 381–420
Lovejoy NR, Albert JS, Crampton WGR (2006) Miocene marine incursions and marine/freshwater transitions: evidence from Neotropical fishes. S Am Earth Sci 21:5–13
Lundberg JG, Kottelat M, Smith GR, Stiassny MLJ, Gill AC (2000) So many fishes, so little time: an overview of recent ichthyological discovery in continental waters. Ann Mo Bot Gard 87:26–62
Mahler DL, Revell LJ, Glor RE, Losos JB (2010) Ecological opportunity and the rate of morphological evolution in the diversification of Greater Antillean anoles. Evolution 64:2731–2745
May RM (1994) Biological diversity: differences between land and sea. Philos Trans R Soc Lond, Ser B 343:105–111
McDowall RM (1997) The evolution of diadromy in fishes (revisited) and its place in phylogenetic analysis. Rev Fish Biol Fish 7:443–462
Moore BR, Chan KMA, Donoghue MJ (2004) Detecting diversification rate variation in supertrees. In: Bininda-Emonds ORP (ed) Phylogenetic supertrees. Combining information to reveal the tree of life. Kluwer Academic Publishers, Dordrecht, pp 487–533
Nelson JS (2006) Fishes of the World. John Wiley and Sons, Hoboken
Nielsen R (2002) Mapping mutations on phylogenies. Syst Biol 51:729–739
Norton SF, Luczkovich JL, Motta PJ (1995) The role of ecomorphological studies in the comparative biology of fishes. Environ Biol Fishes 44:287–304
O’Meara B, Ane C, Sanderson M, Wainwright P (2006) Testing for different rates of continuous trait evolution using likelihood. Evolution 60:922–933
Orme D, Freckleton R, Thomas G, Petzoldt T, Fritz S, Isaac N, Pearse W (2012) Caper: comparative analyses of phylogenetics and evolution in R. R package version 0.5. Available at http://CRAN.R-project.org/package=caper. Accessed 29 June 2012
Paradis E, Claude J, Strimmer K (2004) APE: analysis of phylogenetics and evolution in R language. Bioinformatics 20:289–290
Peters RH (1986) The ecological implications of body size. Cambridge University Press, Cambridge
Posada D, Crandall KA (1998) Modeltest: testing the model of DNA substitution. Bioinformatics 14:817–818
Pouilly M, Lino F, Bretenoux JG, Rosales C (2003) Dietary-morphological relationships in a fish assemblage of the Bolivian Amazonian floodplain. J Fish Biol 62:1137–1158
Price SA, Holzman RA, Near T, Wainwright P (2011) Coral reefs promote the evolution of morphological diversity and ecological novelty in labrid fishes. Ecol Lett 14:462–469
R Core Team (2012) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. http://www.R-project.org/
Revell LJ (2012) Phytools: phylogenetic tools for comparative biology (and other things). Methods Ecol Evol. doi:10.1111/j.2041-210X.2011.00169.x
Revell LJ, Harrison AS (2008) PCCA: a program for phylogenetic canonical correlation analysis. Bioinformatics 24:1018–1020
Revell LJ, Mahler LD, Peres-Neto PR, Redelings BD (2011) A new phylogenetic method for identifying exceptional phenotypic diversification. Evolution 66:135–146
Rosenzweig ML (1995) Species diversity in space and time. Cambridge University Press, Cambridge
Santini F, Nguyen M, Sorenson L, Waltzek TB, Lynch Alfaro JW, Eastman JM, Alfaro ME (2013) Do habitat shifts drive the diversity in teleost fishes? An example from the pufferfishes (Tetraodontidae). J Evol Biol 26:1003–1018
Schaefer SA, Lauder GV (1986) Historical transformation of functional design: evolutionary morphology of feeding mechanisms in Loricarioid catfishes. Syst Zool 35:498–508
Schluter D (2000) The ecology of adaptive radiation. Oxford University Press, Oxford
Sidlauskas B (2008) Continuous and arrested morphological diversification in sister clades of characiform fishes: a phylomorphospace approach. Evolution 62:3135–3156
Simpson GG (1953) The major features of evolution. Columbia University Press, New York
Sokal RR, Rohlf FJ (1995) Biometry: the principles and practice of statistics in biological research, 3rd edn. W.H. Freeman and Co., New York
Swofford DL (2003) PAUP*. Phylogenetic analysis using parsimony (* and other methods), version 4.0b10. Sinauer, Sunderland, MA
ter Braak CJF (1986) Canonical correspondence analysis: a new eigenvector techniques for multivariate direct gradient analysis. Ecology 67:1167–1179
Turner S (1982) A catalogue of fossil fish in Queensland. Mem Qld Mus 20:599–611
Unmack PJ (2001) Biogeography of Australian freshwater fishes. J Biogeogr 28:1053–1089
Unmack PJ (2013) Biogeography. In: Humphries P, Walker KF (eds) The ecology of Australian freshwater fishes. CSIRO Publishing, Collingwood, VIC, pp 25–48
Vari RP (1978) The terapon perches (Percoidei: Teraponidae): a cladistic analysis and taxonomic revision. Bull Am Mus Nat Hist 159:175–340
Vega GC, Wiens JJ (2012) Why are there so few fish in the sea? Proc R Soc B 279:2323–2329
Wainwright PC, Smith WL, Price SA, Tang KL, Sparks JS, Ferry LA, Kuhn KL, Eytan RI, Near TJ (2012) The evolution of pharyngognathy: a phylogenetic and functional appraisal of the pharyngeal jaw key innovation in labroid fishes and beyond. Syst Biol 61:1001–1027
Williams WD, Allen GR (1987) Origins and adaptations of the fauna of inland waters. In: Hyne GR, Walton DW (eds) Fauna of Australia, volume 1A, general articles. Government Printing Service, Canberra, pp 184–201
Winemiller KO (1991) Ecomorphological diversification in lowland freshwater fish assemblages from five biotic regions. Ecol Monogr 61:343–365
Yagishita N, Miya M, Yamanoue Y, Shirai SM, Nakayama K, Suzuki N et al (2009) Mitogenomic evaluation of the unique facial nerve pattern as a phylogenetic marker within the perciform fishes (Teleostei: Percomorpha). Mol Phylogenet Evol 53:258–266
Yamanoue Y, Miya M, Doi H, Mabuchi K, Sakai H, Nishida M (2011) Multiple invasions into freshwater by pufferfishes (Teleostei: Tetraodontidae): a mitogenomic perspective. PLoS ONE 6:e17410
Yoder JB, Clancey E, Des Roches S, Eastman JM, Gentry L, Godsoe W, Hagey TJ et al (2010) Ecological opportunity and the origin of adaptive radiations. J Evol Biol 23:1581–1596
Zwickl DJ (2006) Genetic algorithm approaches for the phylogenetic analysis of large biological sequence datasets under the maximum likelihood criterion. Ph.D. Thesis. University of Texas, Austin
Acknowledgments
For their efforts in helping to collect and/or provide specimens, we thank Mark Adams, Gerald Allen, Jon Armbruster, Michael Baltzly, Cindy Bessey, Joshua Brown, Christopher Burridge, Stephen Caldwell, Adam Fletcher, David Galeotti, Chris Hallett, Michael Hammer, Jeff Johnson, Mark Kennard, Adam Kerezsy, Alfred Ko’ou, Andrew McDougall, Masaki Miya, Sue Morrison, Tim Page, Colton Perna, Ikising Petasi, Michael Pusey, Ross Smith and the Hydrobiology team, Dean Thorburn and the staff from ERISS and Northern Territory Fisheries. Additional samples for genetic work were provided by the Australian, Northern Territory, Queensland, South Australian, Western Australian, University of Kansas and the Smithsonian museums, and we thank their staff and donors. Field collection was partly funded by the Australian Government’s Natural Heritage Trust National Competitive Component and Land and Water Australia. PJU was supported by the W.M. Keck Foundation, R.M. Parsons Foundation, Natural History Museum of Los Angeles County and the National Evolutionary Synthesis Center (NESCent), NSF #EF-0905606. Two anonymous reviewers are thanked for input that greatly improved the final manuscript.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Davis, A.M., Unmack, P.J., Pusey, B.J. et al. Effects of an adaptive zone shift on morphological and ecological diversification in terapontid fishes. Evol Ecol 28, 205–227 (2014). https://doi.org/10.1007/s10682-013-9671-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10682-013-9671-x