Abstract
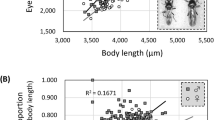
Handicap models of sexual selection propose that females use male sexual ornaments as a cue in mate choice because they reflect commodities that increase female fitness, either directly or indirectly. In contrast to studies on vertebrates, most investigations of ornaments in insects and other invertebrate taxa have been conducted under laboratory conditions. There is a pressing need to address questions relating to sexual signalling of quality in natural populations, as the arbitrary and uniform environments found in the laboratory fail to reflect the world under which animals have evolved. We investigated associations between male ornaments (exaggerated eyespan), attractiveness, and reproductive quality in a wild population of the sexually ornamented stalk-eyed fly, Teleopsis dalmanni. We also explored the relationship between eyespan and reproductive quality in females to evaluate the potential for sexually antagonistic selection on eyespan. We show that eyespan is a generic correlate of reproductive quality, acting as a reliable mirror of variation in reproductive fitness in both sexes. Our findings suggest that male ornaments signal commodities that are of interest to females in the natural environment in which they, and mate preferences for them, have evolved. In addition, the covariance between female eyespan and reproductive output suggests that the former may be a reliable cue of quality in its own right. Our data provide important insights into the evolutionary forces that shape the evolution of exaggerated eyespan in wild populations of this species.



Similar content being viewed by others
References
Al-khairulla H, Warburton D, Knell RJ (2003) Do the eyestalks of female Diopsid flies have a function in intrasexual aggressive encounters? J Insect Behav 16:679–686. doi:10.1023/B:JOIR.0000007703.84691.ad
Andersson M (1986) Evolution of condition-dependent sex ornaments and mating preferences; sexual selection based on viability differences. Evol Int J Org Evol 40:804–816. doi:10.2307/2408465
Andersson M (1994) Sexual selection. Princeton University Press, Princeton
Baker RH, Wilkinson GS (2001) Phylogenetic analysis of sexual dimorphism and eyespan allometry in stalk-eyed flies (Diopsidae). Evol Int J Org Evol 55:1373–1385
Baker RH, Ashwell RIS, Richards TA et al (2001) Effects of multiple mating and male eye span on female reproductive output in the stalk-eyed fly Cyrtodiopsis dalmanni. Behav Ecol 12:732–739. doi:10.1093/beheco/12.6.732
Baker RH, Denniff M, Futerman P et al (2003) Accessory gland size influences time to sexual maturity and mating frequency in the stalk-eyed fly, Cyrtodiopsis dalmanni. Behav Ecol 14:607–611. doi:10.1093/beheco/arg053
Bjorksten TA, Pomiankowski A, Fowler K (2001) Temperature shock during development fails to increase the fluctuating asymmetry of a sexual trait in stalk-eyed flies. Proc R Soc Lond B Biol Sci 268:1503–1510. doi:10.1098/rspb.2001.1575
Burkhardt D, de la Motte I (1985) Selective pressures, variability, and sexual dimorphism in stalk-eyed flies (Diopsidae). Naturwissenschaften 72:204–206. doi:10.1007/BF01195763
Charmantier A, Sheldon BC (2006) Testing genetic models of mate choice evolution in the wild. Trends Ecol Evol 21:417–419. doi:10.1016/j.tree.2006.06.001
Cornwallis CK, Birkhead TR (2008) Plasticity in reproductive phenotypes reveals status-specific correlations between behavioral, morphological, and physiological sexual traits. Evol Int J Org Evol 62:1149–1161. doi:10.1111/j.1558-5646.2008.00346.x
Cotton S, Fowler K, Pomiankowski A (2004a) Do sexual ornaments demonstrate heightened condition-dependent expression as predicted by the handicap hypothesis? Proc R Soc Lond B Biol Sci 271:771–783. doi:10.1098/rspb.2004.2688
Cotton S, Fowler K, Pomiankowski A (2004b) Condition dependence of sexual ornament size and variation in the stalk-eyed fly Cyrtodiopsis dalmanni (Diptera: Diopsidae). Evol Int J Org Evol 58:1038–1046
Cotton S, Fowler K, Pomiankowski A (2004c) Heightened condition dependence is not a general feature of male eyespan in stalk-eyed flies (Diptera: Diopsidae). J Evol Biol 17:1310–1316. doi:10.1111/j.1420-9101.2004.00754.x
David P, Hingle A, Greig D et al (1998) Male sexual ornament size but not asymmetry reflects condition in stalk-eyed flies. Proc R Soc Lond B Biol Sci 265:2211–2216. doi:10.1098/rspb.1998.0561
David P, Bjorksten T, Fowler K et al (2000) Condition-dependent signalling of genetic variation in stalk-eyed flies. Nature 406:186–188. doi:10.1038/35018079
Ellegren H, Sheldon BS (2008) Genetic basis of fitness differences in natural populations. Nature 452:169–175. doi:10.1038/nature06737
Fisher RA (1958) The genetical theory of natural selection. Clarendon Press, Oxford (reprinted 1999, Oxford Univ Press, Oxford)
Fry C (2006) Juvenile hormone mediates a trade-off between primary and secondary sexual traits in stalk-eyed flies. Evol Dev 8:191–201. doi:10.1111/j.1525-142X.2006.00089.x
Getty T (2006) Sexually selected signals are not similar to sports handicaps. Trends Ecol Evol 21:83–88. doi:10.1016/j.tree.2005.10.016
Grafen A (1990) Sexual selection unhandicapped by the fisher process. J Theor Biol 144:473–516. doi:10.1016/S0022-5193(05)80087-6
Greenfield MD (1997) Sexual selection in resource defense polygamy: lessons from territorial grasshoppers. In: Chloe J, Crespi B (eds) The evolution of mating systems in insects and arachnids. Cambridge University Press, Cambridge, pp 75–88
Hingle A, Fowler K, Pomiankowski A (2001a) Size-dependent mate preference in the stalk-eyed fly Cyrtodiopsis dalmanni. Anim Behav 61:589–595. doi:10.1006/anbe.2000.1613
Hingle A, Fowler K, Pomiankowski A (2001b) The effect of transient food stress on female mate preference in the stalk-eyed fly Cyrtodiopsis dalmanni. Proc R Soc Lond B Biol Sci 268:1239–1244. doi:10.1098/rspb.2001.1647
Iwasa Y, Pomiankowski A (1994) The evolution of mate preferences for multiple handicaps. Evol Int J Org Evol 48:853–867. doi:10.2307/2410492
Iwasa Y, Pomiankowski A (1999) Good parent and good genes models of handicap evolution. J Theor Biol 200:97–109. doi:10.1006/jtbi.1999.0979
Iwasa Y, Pomiankowski A, Nee S (1991) The evolution of costly mate preferences. II. The “handicap” principle. Evol Int J Org Evol 45:1431–1442. doi:10.2307/2409890
Johnstone RA (1995) Sexual selection, honest advertisement and the handicap principle: reviewing the evidence. Biol Rev Camb Philos Soc 70:1–65. doi:10.1111/j.1469-185X.1995.tb01439.x
King RC (1970) Ovarian development in Drosophila melanogaster. Academic Press, New York
Kokko H, Jennions MD, Brooks R (2006) Unifying and testing models of sexual selection. Annu Rev Ecol Syst 37:43–66. doi:10.1146/annurev.ecolsys.37.091305.110259
Lande R (1980) Sexual dimorphism, sexual selection, and adaptation in polygenic characters. Evol Int J Org Evol 34:292–305. doi:10.2307/2407393
Lande R, Arnold SJ (1985) Evolution of mating preference and sexual dimorphism. J Theor Biol 117:651–664. doi:10.1016/S0022-5193(85)80245-9
Lorch P, Wilkinson GS, Reillo RP (1993) Copulation duration and sperm precedence in the stalk-eyed fly Cyrtodiopsis whitei. Behav Ecol Sociobiol 32:303–311. doi:10.1007/BF00183785
Maynard Smith J, Harper D (2003) Animal signals. Oxford University Press, Oxford
Meier R, Baker RH (2002) A cladistic analysis of Diopsidae (Diptera) based on morphological and DNA sequence data. Insect Syst Evol 33:325–336
Panhuis TM, Wilkinson GS (1999) Exaggerated eyespan influences male contest outcome in stalk-eyed flies. Behav Ecol Sociobiol 46:221–227. doi:10.1007/s002650050613
Pomiankowski A (1987) Sexual selection: the handicap principle does work—sometimes. Proc R Soc Lond B Biol Sci 231:123–145
Pomiankowski A (1988) The evolution of female mate preferences for male genetic quality. Ox Surv Evol Biol 5:136–184
Rogers DW, Baker RH, Chapman T et al (2005a) Direct and correlated responses to artificial selection on male mating frequency in the stalk-eyed fly Cyrtodiopsis dalmanni. J Evol Biol 18:642–650. doi:10.1111/j.1420-9101.2004.00860.x
Rogers DW, Chapman T, Fowler K et al (2005b) Mating-induced reduction in reproductive organ size in the stalk-eyed fly Cyrtodiopsis dalmanni. BMC Evol Biol 5:37. doi:10.1186/1471-2148-5-37
Rogers DW, Grant CA, Chapman T et al (2006) The influence of male and female eyespan on fertility in the stalk-eyed fly Cyrtodiopsis dalmanni. Anim Behav 72:1363–1369. doi:10.1016/j.anbehav.2006.03.027
Rogers DW, Denniff M, Chapman T et al (2008) Male ornament size signals reproductive quality in the stalk-eyed fly Teleopsis dalmanni. BMC Evol Biol 8:236
Sgrò CM, Hoffmann AA (2004) Genetic correlations, trade-offs and environmental variation. Heredity 93:241–248. doi:10.1038/sj.hdy.6800532
Simmons LW, Emlen DJ (2006) Evolutionary trade-off between weapons and testes. Proc Natl Acad Sci USA 103:16346–16351. doi:10.1073/pnas.0603474103
Simmons LW, Zuk M, Rotenberry JT (2005) Immune function reflected in calling song characteristics in a natural population of the cricket Teleogryllus commodus. Anim Behav 69:1235–1241. doi:10.1016/j.anbehav.2004.09.011
Wilkinson GS (1993) Artificial selection alters allometry in the stalk-eyed fly Cyrtodiopsis dalmanni (Diptera: Diopsidae). Genet Res Camb 62:213–222
Wilkinson GS, Dodson GN (1997) Function and evolution of antlers and eye stalks in flies. In: Chloe J, Crespi B (eds) The evolution of mating systems in insects and arachnids. Cambridge University Press, Cambridge, pp 310–328
Wilkinson GS, Reillo PR (1994) Female choice response to artificial selection in an exaggerated male trait in a stalk-eyed fly. Proc R Soc Lond B Biol Sci 225:1–6. doi:10.1098/rspb.1994.0001
Wilkinson GS, Amitin EG, Johns PM (2005) Sex-linked correlated responses in female reproductive traits to selection on male eye span in stalk-eyed flies. Integr Comp Biol 45:500–510. doi:10.1093/icb/45.3.500
Zahavi A (1975) Mate selection—a selection for a handicap. J Theor Biol 53:205–214. doi:10.1016/0022-5193(75)90111-3
Zuk M (1988) Parasite load, body size, and age of wild-caught male field crickets (Orthoptera: Gryllidae): effects on sexual selection. Evol Int J Org Evol 42:969–976. doi:10.2307/2408912
Acknowledgments
This work was supported by awards from the Association for the Study of Animal Behaviour (A.P. and R.H.), the Royal Society (A.P. and R.H.), the BBSRC (grants to K. Fowler, A.P., T. Chapman and H. Smith; studentship to J.S.), the Department of Biology, University College London (J.S. and A.P.), and a NERC Fellowship (S.C.). We thank two anonymous reviewers for comments on a previous version of the manuscript, and staff at the Gombak Valley Field Research Centre, University of Malaya, Kuala Lumpur for assistance.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Cotton, S., Small, J., Hashim, R. et al. Eyespan reflects reproductive quality in wild stalk-eyed flies. Evol Ecol 24, 83–95 (2010). https://doi.org/10.1007/s10682-009-9292-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10682-009-9292-6