Abstract
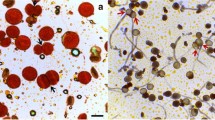
Intra- and interspecific hybridization in the genus Urochloa is complex due to apomixis and polyploidy. Such barriers can be overcome by crossing apomictic tetraploid plants as pollen donors with artificial tetraploid sexual plants as female parents. Another barrier is the incompatibility between the genomes of the different species used in hybridization. Since the meiotic behavior is a good indication of affinity between genomes in a hybrid, the present study analyzed the meiotic behavior of a sexual polyploid interspecific hybrid of Urochloa from the breeding program at Embrapa Beef Cattle. In addition to the segregational abnormalities associated with polyploidy, cytological studies revealed that, after diakinesis, chromosomes presented an abnormal arrangement in the metaphase plate. In 31.33% of meiocytes, chromosomes were organized into more than one plate, forming up to four metaphase plates. Another abnormality observed in 5.72% of the cells was the lack of convergence of chromosomes to the equatorial plate and consequent abnormal anaphase segregation. These abnormalities led to the formation of several nuclei within the same cell then separated by an irregular cytokinesis pattern, forming tetrads with micronuclei, tetrads with microcytes and polyads. Although the sexual hybrid S44 was selected due to its good agronomic characteristics, such as resistance to pasture spittlebugs, the main insect pest in Urochloa, the abnormal meiotic behavior seriously may compromise the formation of viable gametes and consequent formation of viable seeds.



Similar content being viewed by others
Data availability
The datasets generated during and analysed in the current study are available from the corresponding author.
References
Adamowski EV, Pagliarini MS, Valle CB (2008) Meiotic behavior in three interspecific three-way hybrids between Brachiaria ruziziensis and B. brizantha (Poaceae: Paniceae). J Genet 87(1):33–38. https://doi.org/10.1007/s12041-008-0005-7
Akiyama Y, Yamada-Akiyama H, Ebina M (2010) Morphological diversity of chromosomes bearing ribosomal DNA loci in Brachiaria species. Grassl Sci 56(4):217–223. https://doi.org/10.1111/j.1744-697X.2010.00197.x
Alonso L, Kimber G (1981) The analysis of meiosis in hybrids. II. Triploid hybrids. Can J Genet Cytol 23(2):221–234. https://doi.org/10.1139/g81-025
Baldissera JNC, Mendes ABD, Coan MMD et al (2020) Selection based on meiotic behavior in Urochloa decumbens hybrids from non-shattered seed. Trop Grassl Forrajes Trop 8(2):133–140. https://doi.org/10.17138/tgft(8)133-140
Bernini C, Marin-Morales M (2001) Karyotype analysis in Brachiaria (Poaceae) species. Cytobios 104(407):157–172
Boldrini KR, Pagliarini MS, Valle CB (2009) Meiotic behavior of a nonaploid accession endorses x = 6 for Brachiaria humidicola (Poaceae). Genet Mol Res 8(4):1444–1450. https://doi.org/10.4238/vol8-4gmr679
Calisto V, Fuzinatto VA, Message HJ et al (2008) Desynapsis and precocious cytokinesis in Brachiaria humidicola (Poaceae) compromise meiotic division. J Genet 87:27–31. https://doi.org/10.1007/s12041-008-0004-8
Chen C, Marcus A, Li W et al (2002) The Arabidopsis ATK1 gene is required for spindle morphogenesis in male meiosis. Development 129(10):2401–2409. https://doi.org/10.1242/dev.129.10.2401
Corrêa CTR, Bonetti NGZ, Barrios SCL et al (2020) GISH-based comparative genomic analysis in Urochloa P. Beauv. Mol Biol Rep 47(2):887–896. https://doi.org/10.1007/s11033-019-05179-7
Davies D (1974) Chromosome elimination in interspecific hybrids. Heredity 32(2):267–270. https://doi.org/10.1038/hdy.1974.30
Felismino MF, Pagliarini MS, Valle CB (2010) Meiotic behavior of interspecific hybrids between artificially tetraploidized sexual Brachiaria ruziziensis and tetraploid apomictic B. brizantha (Poaceae). Sci Agricola 67(2):191–197. https://doi.org/10.1590/S0103-90162010000200010
Ferreira RCU, Moraes ACL, Chiari L et al (2021) An overview of the genetics and genomics of the Urochloa species most commonly used in pastures. Front Plant Sci 12:770461. https://doi.org/10.3389/fpls.2021.770461
Fuzinato VA, Pagliarini MS, Valle CB (2008) Evaluation of microsporogenesis in an interspecific Brachiaria hybrid (Poaceae) collected in distinct years. Genet Mol Res 7(2):424–432. https://doi.org/10.4238/vol7-2gmr430
Fuzinatto VA, Pagliarini MS, Valle CB (2007) Microsporogenesis in sexual Brachiaria hybrids (Poaceae). Genet Mol Res 6(4):1107–1117
Fuzinatto VA, Pagliarini MS, Valle CB (2012) Meiotic behavior in apomictic Brachiaria ruziziensis × B. brizantha (Poaceae) progenies. Sci Agricola 69:380–385. https://doi.org/10.1590/S0103-90162012000600006
Gale M, Miller T (1987) The introduction of alien genetic variation in wheat. In: Lupton F (ed) Wheat breeding. Springer, Cham, pp 173–210. https://doi.org/10.1007/978-94-0093131-2
Gobbe J, Swenne A, Louant BP (1981) Diploïdes naturels et autotetraploïdes induits chez Brachiaria ruziziensis Germain et Evrard: critères d’identification. Agron Trop 36(4):339–346
Jank L, Barrios SC, Valle CB et al (2014) The value of improved pastures to Brazilian beef production. Crop Pasture Sci 65(11):1132–1137. https://doi.org/10.1071/CP13319
Koduru P, Rao M (1981) Cytogenetics of synaptic mutants in higher plants. Theor Appl Genet 59:197–214. https://doi.org/10.1007/BF00265494
Marfil CF, Masuelli RW, Davison J et al (2006) Genomic instability in Solanum tuberosum × Solanum kurtzianum interspecific hybrids. Genome 49(2):104–113. https://doi.org/10.1139/g05-088
Mendes-Bonato AB, Junqueira Filho RG, Pagliarini MS et al (2002) Unusual cytological patterns of microsporogenesis in Brachiaria decumbens: abnormalities in spindle and defective cytokinesis causing precocious cellularization. Cell Biol Int 26(7):641–646. https://doi.org/10.1006/cbir.2002.0929
Mendes-Bonato AB, Risso-Pascotto C, Pagliarini MS et al (2006) Cytogenetic evidence for genome elimination during microsporogenesis in interspecific hybrid between Brachiaria ruziziensis and B. brizantha (Poaceae). Genet Mol Biol 29(4):711–714. https://doi.org/10.1590/S1415-47572006000400021
Mendes-Bonato AB, Felismino MF, Souza-Kaneshima AM et al (2009) Abnormal meiosis in tetraploid genotypes of Brachiaria brizantha (Poaceae) induced by colchicine: its implications for breeding. J Appl Genet 50:83–87. https://doi.org/10.1007/BF03195658
Oleszczuk S, Grzechnik N, Mason AS et al (2019) Heritability of meiotic restitution in haploid triticale. Plant Cell Rep 38:1515–1525. https://doi.org/10.1007/s00299-019-02462-6
Pagliarini MS, Risso-Pascotto C, Souza-Kaneshima AM et al (2008) Analysis of meiotic behavior in selecting potential genitors among diploid and artificially induced tetraploid accessions of Brachiaria ruziziensis (Poaceae). Euphytica 164:181–187. https://doi.org/10.1007/s10681-008-9697-2
Paula CMP, Figueiredo KG, Sobrinho FS et al (2016) Microsporogenesis analysis validates the use of artificially tetraploidized Brachiaria ruziziensis in breeding programs. Genet Mol Res 15(3):gmr15038737. https://doi.org/10.4238/gmr.15038737
Pinheiro AA, Pozzobon MT, Valle CB et al (2000) Duplication of the chromosome number of diploid Brachiaria brizantha plants using colchicine. Plant Cell Rep 19:274–278. https://doi.org/10.1007/s002990050011
Qu L, Vorsa N (1999) Desynapsis and spindle abnormalities leading to 2n pollen formation in Vaccinium darrowi. Genome 42(1):35–40. https://doi.org/10.1139/g98-098
Ragalzi CM, Mendes ABD, Simeão RM et al (2021) Microsporogenesis associated with seed yield in Urochloa sexual polyploid hybrids. Crop Breed Appl Biotechnol. https://doi.org/10.1590/1984-70332021v21n4a57
Renvoize SA, Clayton WD, Kabuye CHS (1996) Morphology, taxonomy, and natural distribution of Brachiaria (Trin.) Griseb. In: Miles JW, Valle CB, Maass BL (eds) Brachiaria: biology, agronomy, and improvement, vol 259. CIAT, National City, pp 1–15
Ricci GCL, Souza-Kaneshima AM, Felismino MF et al (2011) Chromosome numbers and meiotic analysis in the pre-breeding of Brachiaria decumbens (Poaceae). J Genet 90:289–294. https://doi.org/10.1007/s12041-011-0087-5
Risso-Pascotto C, Pagliarini MS, Valle CB et al (2004) Asynchronous meiosis in an interspecific hybrid of Brachiaria ruziziensis and B. brizantha. Plant Cell Rep 23:304–310. https://doi.org/10.1007/s00299-004-0802-2
Risso-Pascotto C, Pagliarini MS, Valle CB (2005a) Meiotic behavior in interspecific hybrids between Brachiaria ruziziensis and Brachiaria brizantha (Poaceae). Euphytica 145:155–159. https://doi.org/10.1007/s10681-005-0893-z
Risso-Pascotto C, Pagliarini MS, Valle CB (2005b) Multiple spindles and cellularization during microsporogenesis in an artificially induced tetraploid accession of Brachiaria ruziziensis (Gramineae). Plant Cell Rep 23:522–527. https://doi.org/10.1007/s00299-004-0867-y
Sales GLM, Vidal IJA, Ragalzi CM et al (2021) Microsporogênese em híbridos intraespecíficos sexuais de U. humidicola (Rendle) Morrone & Zuloaga [syn. Brachiaria humidicola (Rendle) Schweick.]. Braz J Dev 7(4):37565–37575. https://doi.org/10.34117/bjdv7n4-293
Schwarzacher T, Heslop-Harrison JS, Anamthawat-Jonsson K et al (1992) Parental genome separation in reconstructions of somatic and premeiotic metaphases of Hordeum vulgare × H. bulbosum. J Cell Sci 101(1):13–24. https://doi.org/10.1242/jcs.101.1.13
Shamina N (2005) A catalogue of abnormalities in the division spindles of higher plants. Cell Biol Int 29(5):384–391. https://doi.org/10.1016/j.cellbi.2005.02.004
Silkova OG, Shchapova AI, Shumny VK (2011) Meiotic restitution in amphihaploids in the tribe Triticeae. Russ J Gent 47(4):383–393. https://doi.org/10.1134/S1022795411040120
Simioni C, Valle C (2009) Chromosome duplication in Brachiaria (A. Rich.) Stapf allows intraspecific crosses. Crop Breed Appl Biotechnol 9(4):328–334
Simioni C, Valle C (2011) Meiotic analysis in induced tetraploids of Brachiaria decumbens Stapf. Crop Breed Appl Biotechnol 11:43–49. https://doi.org/10.1590/S1984-70332011000100006
Souza VF, Pagliarini MS, Valle CB et al (2015) Meiotic behavior of Brachiaria decumbens hybrids. Genet Mol Res 14(4):12855–12865. https://doi.org/10.4238/2015.October.21.5
Souza-Kaneshima AM, Simioni C, Felismino MF et al (2010) Meiotic behaviour in the first interspecific hybrids between Brachiaria brizantha and Brachiaria decumbens. Plant Breed 129(2):186–191. https://doi.org/10.1111/j.1439-0523.2009.01674.x
Sundberg E, Lagercrantz U, Glimelius K (1991) Effects of cell type used for fusion on chromosome elimination and chloroplast segregation in Brassica oleracea (+) Brassica napus hybrids. Plant Sci 78(1):89–98. https://doi.org/10.1016/0168-9452(91)90165-5
Swenne A, Louant BP, Dujardin M (1981) Induction par la colchicine de formes autotetraploides chez Brachiaria ruzizienses Germain et Evrard (Gramineae). Agron Trop 36(2):134–141
Tohme J, Palacios N, Lenis S et al (1996) Applications of biotechnology to Brachiaria, vol 259. CIAT, National City, pp 196–204
Valle CB, Pagliarini MS (2009) Biology, cytogenetics, and breeding of Brachiaria, vol 5. CRC Press, Boca Raton, pp 103–152
Valle C, Savidan Y (1996) Genetics and reproductive biology of Brachiaria. In: Miles JW, Valle CB, Maass BL (eds) Brachiaria: biology, agronomy, and improvement, vol 259. CIAT, National City, pp 147–163
Walczak CE, Vernos I, Mitchison TJ et al (1998) A model for the proposed roles of different microtubule-based motor proteins in establishing spindle bipolarity. Curr Biol 8(16):903–913. https://doi.org/10.1016/S0960-9822(07)00370-3
Zamariola L, Tiang CL, Storme N et al (2014) Chromosome segregation in plant meiosis. Front Plant Sci 5:279. https://doi.org/10.3389/fpls.2014.00279
Acknowledgements
The authors are grateful to Embrapa Beef Cattle and the Association for The Promotion of Research in Forage Breeding—UNIPASTO.
Funding
The authors declare that no funds, grants, or other support were received during the preparation of this manuscript.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by GLdMS and ABDM. The first draft of the manuscript was written by ABDM and CBdV, all authors commented on previous versions of the manuscript. All authors have read and agreed to the published version of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no disclosed any conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
de Melo Sales, G.L., do Valle, C.B. & Mendes, A.B.D. Chaotic spindles causes abnormal chromosome behavior in an interspecific sexual hybrid of Urochloa. Euphytica 220, 21 (2024). https://doi.org/10.1007/s10681-023-03280-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10681-023-03280-4