Abstract
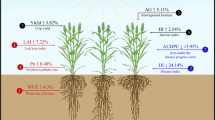
Maize (Zea mays L.) is an important crop worldwide, but in many regions is increasingly faced with the issue of phosphorus (P)-deficient soils. The aim of this study was to screen for low P-tolerance in maize and to dissect the underlying phenotypic response. To this end, we evaluated a panel of 380 diverse inbred lines in the field under low and normal P conditions for 17 morphological, biomass- and yield-related traits. All traits showed a significant genotypic variation and a moderate to high repeatability for each P condition. Under P deficiency, all traits were significantly lower compared to the control with normal P availability. The variance due to the interaction between genotype and P condition was significant for most traits, but generally small compared to the genotypic variance. Regarding the low P tolerance index, i.e. the phenotype under low P relative to the control, we observed a generally similar response within the morphological, biomass- or yield-related traits. Interestingly, genotypes showing little yield reduction under low P appear to achieve this tolerance through different strategies. Based on our results, low-P tolerant and low-P sensitive lines were identified, which can be used for further genetic research as well as to improve this globally important trait in maize breeding.




Similar content being viewed by others
References
Anwar J, Subhani GM, Hussain M et al (2011) Drought tolerance indices and their correlation with yield in exotic wheat genotypes. Pak J Bot 43:1527–1530. https://doi.org/10.3417/2009060
Bernal-Vasquez AM, Utz HF, Piepho HP (2016) Outlier detection methods for generalized lattices: a case study on the transition from ANOVA to REML. Theor Appl Genet 129:787–804. https://doi.org/10.1007/s00122-016-2666-6
Cai HG, Chu Q, Gu RL et al (2012) Identification of QTLs for plant height, ear height and grain yield in maize (Zea mays L.) in response to nitrogen and phosphorus supply. Plant Breed 131:502–510. https://doi.org/10.1111/j.1439-0523.2012.01963.x
Covarrubias-Pazaran G (2016) Genome-Assisted prediction of quantitative traits using the r package sommer. PLoS ONE 11:e0156744. https://doi.org/10.1371/journal.pone.0156744
Cullis BR, Smith AB, Coombes NE (2006) On the design of early generation variety trials with correlated data. J Agric Biol Environ Stat 11:381–393. https://doi.org/10.1198/108571106X154443
Epskamp S, Schmittmann VD, Borsboom D (2012) qgraph: network visualizations of relationships in psychometric data. 48:1–16. https://doi.org/10.18637/jss.v048.i04
Gemenet DC, Tom CT, Sy O et al (2014) Pearl millet inbred and testcross performance under low phosphorus in West Africa. Crop Sci 54:2574–2585. https://doi.org/10.2135/cropsci2014.04.0277
Gilmour AR, Gogel BJ, Cullis BR, Thompson R (2009) ASReml User Guide Release 3.0. VSN International Ltd, UK
Gu RL, Chen FJ, Long LZ et al (2016) Enhancing phosphorus uptake efficiency through QTL-based selection for root system architecture in maize. J Genet Genomics 43:663–672. https://doi.org/10.1016/j.jgg.2016.11.002
Kante M, Rattunde F, Nébié B et al (2019) Sorghum hybrids for low-Input farming systems in west Africa: quantitative genetic parameters to guide hybrid breeding. Crop Sci 59:2544–2561. https://doi.org/10.2135/cropsci2019.03.0172
Kochian LV (2012) Plant nutrition: rooting for more phosphorus. Nature 488:466–467. https://doi.org/10.1038/488466a
Liu ZG, Gao K, Shan SC et al (2017) Comparative analysis of root traits and the associated QTLs for maize seedlings grown in paper roll, hydroponics and vermiculite culture system. Front Plant Sci 8:1–13. https://doi.org/10.3389/fpls.2017.00436
Liu ZG, Liu XS, Craft EJ et al (2018) Physiological and genetic analysis for maize root characters and yield in response to low phosphorus stress. Breed Sci 68:268–277. https://doi.org/10.1270/jsbbs.17083
Luo XJ, Liu J, Luo Y et al (2009) FactoMineR: an R Package for multivariate analysis. Environ Int 35:253–258. https://doi.org/10.1016/j.envint.2008.06.007
Luo B, Ma P, Nie Z et al (2019) Metabolite profiling and genome-wide association studies reveal response mechanisms of phosphorus deficiency in maize seedling. Plant J 97:947–969. https://doi.org/10.1111/tpj.14160
Maharajan T, Ceasar SA, Ajeesh krishna TP et al (2018) Utilization of molecular markers for improving the phosphorus efficiency in crop plants. Plant Breed 137:10–26. https://doi.org/10.1111/pbr.12537
Mendiburu F, Simon R (2015) Agricolae—ten years of an Open source Statistical tool for experiments in Breeding, agriculture and biology. PeerJ Prepr 3:1–17. https://doi.org/10.7287/peerj.preprints.1404v1
Pommel B, Gallais A, Coque M et al (2006) Carbon and nitrogen allocation and grain filling in three maize hybrids differing in leaf senescence. Eur J Agron 24:203–211. https://doi.org/10.1016/j.eja.2005.10.001
Ravelombola W, Qin J, Shi AN et al (2017) Association mapping revealed SNP markers for adaptation to low phosphorus conditions and rock phosphate response in USDA cowpea (Vigna unguiculata (L.) Walp.) germplasm. Euphytica 213:183. https://doi.org/10.1007/s10681-017-1971-8
Ud-Din N, Carver BF, Clutter AC (1992) Genetic analysis and selection for wheat yield in drought-stressed and irrigated environments. Euphytica 62:89–96. https://doi.org/10.1007/BF00037933
Vejchasarn P, Lynch JP, Brown KM (2016) Genetic variability in phosphorus responses of rice root phenotypes. Rice 9:1–16. https://doi.org/10.1186/s12284-016-0102-9
Wang XL, Wang HW, Liu SX et al (2016) Genetic variation in ZmVPP1 contributes to drought tolerance in maize seedlings. Nat Genet 48:1233–1241. https://doi.org/10.1038/ng.3636
Wang W, Ding GD, White PJ et al (2018) Mapping and cloning of quantitative trait loci for phosphorus efficiency in crops: opportunities and challenges. Plant Soil 439:91–112. https://doi.org/10.1007/s11104-018-3706-6
Wissuwa M, Yano M, Ae N (1998) Mapping QTLs for phosphorus deficiency tolerance in rice (Oryza sativa L.). Theor Appl Genet 97:1361–1369. https://doi.org/10.1007/s001220051030
Xu C, Zhang HW, Sun JH et al (2018) Genome-wide association study dissects yield components associated with low-phosphorus stress tolerance in maize. Theor Appl Genet 131:1699–1714. https://doi.org/10.1007/s00122-018-3108-4
Yang XH, Gao SB, Xu ST et al (2011) Characterization of a global germplasm collection and its potential utilization for analysis of complex quantitative traits in maize. Mol Breed 28:511–526. https://doi.org/10.1007/s11032-010-9500-7
Yang N, Lu YL, Yang XH et al (2014) Genome wide association studies using a new nonparametric model reveal the genetic architecture of 17 agronomic traits in an enlarged maize association panel. PLoS Genet 10:e1004573. https://doi.org/10.1371/journal.pgen.1004573
Zhang LT, Li J, Rong TZ et al (2014) Large-scale screening maize germplasm for low-phosphorus tolerance using multiple selection criteria. Euphytica 197:435–446. https://doi.org/10.1007/s10681-014-1079-3
Zhang HW, Xu RN, Xi CX et al (2015) Large-scale evaluation of maize germplasm for low-phosphorus tolerance. PLoS ONE 10:e0124212. https://doi.org/10.1371/journal.pone.0124212
Acknowledgements
We thank Prof. Jianbing Yan at Huazhong Agricultural University and Prof. Xiaohong Yang at China Agricultural University for providing materials in our experiment.
Funding
This project was funded by the National Key Research and Development Program of China (2018YFD0100201 & 2016YFD0101201), and by the Deutsche Forschungsgemeinschaft (DFG, German Research Foundation) – 328017493/GRK 2366 (Sino-German International Research Training Group AMAIZE-P).
Author information
Authors and Affiliations
Contributions
WL, TW: designed the study. DL, ZC, MW, ZZ, SC collected the phenotypic data. DL performed data analysis. DL, WLL, TMW, SC, FC, LY, TW, WL wrote the manuscript. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
10681_2020_2727_MOESM1_ESM.png
Fig. S1 Trait distributions under low and normal P, and genotypic response to the two P conditions. Results are shown for (A) ear number per plant (ENPP), (B) kernel number per plant (KNPP), (C) kernel number per ear (KNPE), (D) cob length (CL), (E) cob diameter (CD), (F) hundred grain weight (HGW), (G) all dry weight per plant (ADWPP), (H) ear height (EH), (I) ear leaf length (ELL), (J) ear leaf width (ELW), (K) ear leaf order (ELO). (PNG 218 kb)
10681_2020_2727_MOESM2_ESM.png
Fig. S2 Boxplots showing LPTI for different subpopulations for the traits (A) ear number per plant (ENPP), (B) kernel number per plant (KNPP), (C) kernel number per ear (KNPE), (D) cob length (CL), (E) cob diameter (CD), (F) hundred grain weight (HGW), (G) all dry weight per plant (ADWPP), (H) ear height (EH), (I) ear leaf length (ELL), (J) ear leaf width (ELW), and (K) ear leaf order (ELO). Tem, temperate subpopulation; TST, tropical/subtropical subpopulation; G1, lines had no ear under low P conditions and had ears under normal P; G2, lines that had ears under both P conditions. Letters indicate significant differences between the groups at the 0.05 significance level. (PNG 110 kb)
10681_2020_2727_MOESM3_ESM.png
Fig. S3 Network analysis illustrating correlations among of all evaluated traits under low and normal P conditions. (PNG 215 kb)
10681_2020_2727_MOESM7_ESM.png
Fig. S7 Network analysis illustrating correlations among all evaluated traits in the G1 and G2 subgroups under low and normal P conditions. (PNG 247 kb)
10681_2020_2727_MOESM8_ESM.png
Fig. S8 PCA results of all traits under low and normal P conditions. (A) Heatmap of the quality of representation of all traits and biplot for the data under low P conditions. (B) Heatmap of the quality of representation of all traits and biplot for the data under normal P conditions. Dim, the abbreviation of dimension. (PNG 171 kb)
Rights and permissions
About this article
Cite this article
Li, D., Chen, Z., Wang, M. et al. Dissecting the phenotypic response of maize to low phosphorus soils by field screening of a large diversity panel. Euphytica 217, 12 (2021). https://doi.org/10.1007/s10681-020-02727-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10681-020-02727-2