Abstract
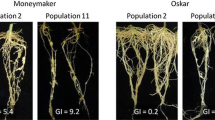
Soil and root samples were collected from major tomato growing areas of Ethiopia during the 2012/2013 growing season to identify root-knot nematode problems. DNA-based and isozyme techniques revealed that Meloidogyne incognita and M. javanica were the predominant Meloidogyne species across the sampled areas. The aggressiveness of different populations of these species was assessed on tomato cultivars Marmande and Moneymaker. The two most aggressive populations of each species were selected and further tested on 33 tomato genotypes. The resistance screening and mechanism of resistance was performed after inoculation with 100 freshly hatched (<24 h) second-stage juveniles (J2). Eight weeks after inoculation the number of egg masses produced on each cultivar was assessed. For the resistance mechanism study, J2 penetration and their subsequent development inside the tomato roots were examined at 1, 2, 4 and 6 weeks after inoculation. On both cultivars Marmande and Moneymaker all M. incognita and M. javanica populations formed a high number of egg masses indicating highly aggressive behaviour. Populations from ‘Jittu’ and ‘Babile’ for M. incognita and ‘Jittu’ and ‘Koka’ for M. javanica were selected as most aggressive. None of the 33 tomato genotypes were immune for these M. incognita and M. javanica populations. However, several tomato genotypes were found to have a significant effect on the number of egg masses produced indicating possible resistance. For M. javanica populations there were more plants from cultivars or breeding lines on which no egg masses were found compared to M. incognita populations. The lowest number of egg masses for both populations of M. incognita was produced on cultivars Bridget40, Galilea, and Irma while for M. javanica it was on Assila, Eden, Galilea, Tisey, CLN-2366A, CLN-2366B and CLN-2366C. Tomato genotypes, time (weeks after inoculation) and their interaction were significant sources of variation for J2 penetration and their subsequent development inside the tomato roots. Differential penetration was found in breeding lines such as CLN-2366A, CLN-2366B and CLN-2366C, but many of the selected tomato genotypes resistance for the tested M. incognita and M. javanica populations were expressed by delayed nematode development. Therefore, developing a simple screening technique to be used by local farmers or extension workers is crucial to facilitate selection of a suitable cultivar.






Similar content being viewed by others
References
Anwar SA, McKenry MV (2000) Penetration, development and reproduction of Meloidogyne arenaria on two new resistant Vitis spp. Nematropica 30:9–17
Bauchet G, Causse M (2012) Genetic diversity in Tomato (Solanum lycopersicum) and its wild relatives. In: Caliskan M. (Ed.). Environmental sciences-genetic diversity in plants, doi:10.5772/33073
Belan LL, Fonseca SO, Alves FR, Junior WCJ, Matta EP, Cabral PDS, Rabello LKC, Rodrigues AA (2009) Screening of Cherry tomato genotypes for resistance to Meloidogyne incognita and M. javanica. Nematol Bras 33(3):256–259
Bendezu IF, Starr J (2003) Mechanism of resistance to Meloidogyne arenaria in the peanut genotype COAN. J Nematol 35:115–118
Byrd DW, Kirkpatrick T, Barker KR (1983) An improved technique for clearing and staining plant tissues for detection of nematodes. J Nematol 15:142–143
Castagnone-Sereno P (2006) Genetic variability and adaptive evolution in parthenogenetic root-knot nematodes. Heredity 96:282–289
Cook R, Evans K (1987) Resistance and tolerance. In: Brown RH, Kerry BR (eds) Principles and practice of nematode control in crops. Academic Press, London, pp 179–231
Cortada L, Mantelin S, Verdejo-Lucas S, Kaloshian I (2012) Marker analysis for detection of the Mi-1.2 resistance gene in tomato hybrid rootstocks and cultivars. Nematology 14:631–642
Courtright EM, Wall DH, Virginia RA, Frisse LM, Vida JT, Thomas WK (2000) Nuclear and mitochondrial DNA sequence diversity in the antarctic nematode Scottnema lindsayae. J Nematol 32:143–153
Coyne DL, Fourie HH, Moens M (2009) Current and future management strategies in resource-poor farming. In: Perry RN, Moens M, Starr JL (eds) Root-knot nematodes. CABI, Wallingford, pp 444–475
Creech RG, Jenkins JN, Tang B, Lawrence GW, McCarty JC (1995) Cotton resistance to root-knot nematode. I: penetration and reproduction. Crop Sci 35:365–368
Daykin ME, Hussey RS (1985) Staining and histopathological techniques in nematology. In: Barker KR, Carter CC, Sasser JN (eds) An advanced Treatise on Meloidogyne, Volume II Methodology. North Carolina State University Graphics, Raleigh, pp 39–48
Devran Z, Sogut MA, Mutlu N (2010) Response of tomato rootstocks with the Mi resistance gene to Meloidogyne incognita race 2 at different soil temperatures. Phytopathol Mediterr 49:11–17
Dropkin VH (1969) The necrotic reaction of tomatoes and other hosts resistant to Meloidogyne: reversal by temperature. Phytopathology 59:1632–1637
Esbenshade P, Triantaphyllou A (1985) Use of enzyme phenotypes for identification of Meloidogyne species. J Nematol 17:6–20
Fassuliotis G (1979) Plant breeding for root-knot resistance. In: Lambert F, Taylor CE (eds) Root-knot Nematodes (Meloidogyne species) systematics, biology and control. Academic Press, New York, pp 425–453
Giovannucci E (1999) Tomatoes, tomato-based products, lycopene and cancer: review of the epidemiological literature. J Natl Cancer Inst 91:317–331
Hadisoeganda WW, Sasser JN (1982) Resistance of tomato, bean, Southern pea and garden pea genotypes to root-knot nematodes based on host suitability. Plant Dis 66:145–150
Herman M, Hussey RS, Boerma HR (1991) Penetration and development of Meloidogyne incognita on roots of resistant soybean genotypes. J Nematol 23:155–161
Holterman M, Van Der Wurff A, Van Den Elsen S, Van Megen H, Bongers T, Holovachov O, Bakker J, Helder J (2006) Phylum-wide analysis of SSU rDNA reveals deep phylogenetic relationships among nematodes and accelerated evolution toward crown clades. Mol Biol Evol 23:1792–1800
Janssen T, Karssen G, Verhaeven M, Coyne D, Bert W (2016) Mitochondrial coding genome analysis of tropical root-knot nematodes (Meloidogyne) supports haplotype based diagnostics and reveals evidence of recent reticulate evolution. Sci Rep 6:22591. doi:10.1038/srep22591
Jiregna TD (2013) Evaluation of agronomic performance and Lycopene variation in tomato (Lycopersicon esculentum Mill.) genotypes in Mizan, Southern Ethiopia. World Appl Sci J 27(11):1450–1454
Jones JT, Haegeman A, Danchin EGJ, Gaur HS, Helder J, Jones MGK, Kikuchi T, Rose ML, Palomares-Rius JE, Wesemale WML, Perry RN (2013) Review of top 10 plant-parasitic nematodes in molecular plant pathology. Mol Plant Pathol 1–16
Kesba HH, Hasnin NMM, Mahmoud AMA, Ali AHH (2015) Evaluation of Some Tomato Genotypes to Meloidogyne incognita Resistance. Am Eurasian J Agric Environ Sci 15(7):1402–1410
Khah EM, Rumbos CI, Sabir N (2011) Response of local and commercial tomato cultivars and rootstocks to Meloidogyne javanica infestation. Aust J Crop Sci 5(11):1388–1395
McSorley R (2011) Overview of organic amendments for management of plant-parasitic nematodes, with case studies from Florida. J Nematol 43(2):69–81
Mwesige R, Seid A, Wesemael WML (2016) Root-knot nematodes on tomatoes in Kyenjojo and Masaka districts in Uganda. Afr J Agric Res 11:3598–3606
Noling J (2000) Effects of continuous culture of a resistant tomato cultivar on Meloidogyne incognita soil population density and pathogenicity. J Nematol 32(4): 452 (abstract)
Ploeg AT, Maris PC (1999) Effect of temperature on suppression of Meloidogyne incognita by Tagetes Cultivars. Suppl J Nematol 31(4S):709–714
Reynolds ES, Carter WW, O’Bannon JH (1970) Symptomless resistance of alfalfa to Meloidogyne incognita acrita. J Nematol 2:131–134
Roberts PA (1992) Current status of the availability, development and use of host plant resistance to nematodes. J Nematol 24:213–227
Roberts PA, Matthews WC, Veremis, JC (1998) Genetic mechanisms of host-plant resistance to nematodes. In: K.R. Barker, G.A. Pederson, and G.L. Windham (eds.), Plant and nematode interactions, p 209–238. Amer. Soc. Agro., Crop Sci. Soc. Amer., and Soil Sci. Soc. Amer. Madison, Wisc
Roberts PA, Thomason IJ (1986) Variability in reproduction of isolates of M. incognita and M. javanica on resistant tomato genotypes. Plant Dis 24:547–551
Robertson L, Diez-Rojo MA, Lopez-Perez JA, Buena AP, Escuer M, Cepero JL, Martinez C, Bello A (2009) New host races of Meloidogyne arenaria, M. incognita and M. javanica from horticultural regions of Spain. Plant Dis 93:180–184
Sasser JN (1980) Root-knot nematodes: a Global Menace to Crop Production. Plant Dis 64:36–41
Schneider SM (1991) Penetration of susceptible and resistant tobacco cultivars by Meloidogyne juveniles. J Nematol 23:225–228
Seid A, Fininsa C, Mekete T, Decraemer W, Wesemael WML (2015) Tomato (Lycopersicon Esculentum L.) and root- knot nematodes (Meloidogyne spp.)-a century old battle. Nematology 17(9):995–1009
Silva RV, Oliveira RDL, Ferreira PS, Ferreira AO, Rodrigues FA (2013) Defense responses to Meloidogyne exigua in resistant coffee cultivar and non-host plant. Trop Plant Pathol 38:114–121
Singh SK, Khurma UR (2007) Susceptibility of six tomato cultivars to the root-knot nematode, Meloidogyne incognita. South Pac J Nat Sci 25:73–77
Starr JL, Mercer CF (2009) Development of resistant varieties. In: Perry RN, Moens M, Starr JL (eds) Root-knot nematodes. CABI, Wallingford, pp 326–337
Starr JL, McDonald AH, Claudius-Cole AO (2013) Nematode Resistance in Crops. In: Perry RN, Moens M (eds) Plant nematology, 2nd edn. CABI, Wallingford, pp 411–436
Sydenham GM, McSorley R, Dunn RA (1996) Effects of resistance in Phaseolus vulgaris on development of Meloidogyne species. J Nematol 28:485–491
Verdejo-Lucas S, Cortada L, Sorribas FJ, Ornat C (2009) Selection of Virulent populations of Meloidogyne javanica by repeated cultivation of Mi resistance tomato rootstocks in a plastic house. Plant Pathol 58:990–998
Walters SA, Wehner TC, Daykin ME, Barker KR (2006) Penetration rates of root-knot nem- atodes into Cucumis sativus and C. metuliferus roots and subsequent histological changes. Nematropica 36:231–242
Wesemael WML, Moens M (2012) Screening of common bean (Phaseolus vulgaris) for resistance against temperate root-knot nematodes (Meloidogyne spp.). Pest Manag Sci 68:702–708
Wesemael WML, Viaene N, Moens M (2011) Root-knot nematodes (Meloidogyne spp.) in Europe. Nematology 13:3–16
Windham GL, Williams WP (1994) Penetration and development of Meloidogyne incognita in roots of resistant and susceptible corn genotypes. J Nematol 26:80–85
Wishart J, Phillips MS, Blok VC (2002) Ribosomal intergenic spacer: a polymerase chain reaction diagnostic for Meloidogyne chitwoodi, M. fallax, and M. hapla. Phytopathology 92:884–892
Zijlstra C, Donkers-Venne DTHM, Fargette M (2000) Identification of Meloidogyne incognita, M. javanica and M. arenaria using sequence characterized amplified region (SCAR) based PCR assays. Nematology 2:847–853
Acknowledgements
This research was financially and technically supported by VLIR-UOS (ICP-PhD Scholarship) from Belgium.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Seid, A., Fininsa, C., Mekete, T.M. et al. Resistance screening of breeding lines and commercial tomato cultivars for Meloidogyne incognita and M. javanica populations (Nematoda) from Ethiopia. Euphytica 213, 97 (2017). https://doi.org/10.1007/s10681-017-1886-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10681-017-1886-4