Abstract
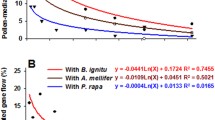
Three-year field assessment of gene flow from the genetically modified herbicide-resistant spring wheat variety ‘Andros’ to its non-transgenic counterpart has been conducted. A circular field trial design where the wheat plants containing the bar and gfp genes were planted in a central plot, while the recipient non-transgenic plants were grown in eight compass sectors at a distance of 1–5 m from the pollen source, has been developed. Gene flow was analyzed by testing the glufosinate-based herbicide resistance in seedling progeny. The phenotypic and molecular examination of more than 712,000 germinated seeds allowed us to draw a conclusion that the pollen-mediated transgene flow might occur at a low frequency (<0.8 %) even if the nontransgenic wheat was located in the proximity to its transgenic counterpart. The strong asymmetric distribution of gene flow and the maximum outcrossing rate were detected in compass sectors following the direction of the dominant wind. The gene flow rate averaged over all wind direction varied in different years from 0.134 to 0.416 %. The dramatic reduction in transgene flow frequencies was observed while increasing the distance between transgenic and non-transgenic plots. The rise of average gene flow due to significant increase of the transgene pollen area was not confirmed statistically. These results predict that the pollen-mediated transgene flow in spring wheat can be maintained at negligible levels while the existence of a short isolation distance in order to prevent contamination of adjacent non-GM wheat.



Similar content being viewed by others
References
Barro F, Barceló P, Lazzeri PA, Shewry PRA, Ballesteros JM (2002) Field evaluation and agronomic performance of transgenic wheat. Theor Appl Genet 105:980–984
Becker D, Brettschneider R, Lörz H (2004) Fertile transgenic wheat from microprojectile bombardment of scutellar tissue. Plant J 5(2):299–307
Beckie HJ, Warwick SI, Sauder CA, Hall LM, Harker KN, Lozinski C (2011) Pollen-mediated gene flow in commercial fields of spring wheat in Western Canada. Crop Sci 51:306–313
Canadian regulations and procedures for pedigreed seed crop production (2015) Canadian Seed Growers’ Association. Circular 6, Section 02. Cereals. Revision 1.10, 2015. http://seedgrowers.ca/seed-growers/regulations/. Accessed 1 Feb 2015
Cannell ME, Doherty A, Lazzeri PA, Barcelo P (1999) A population of wheat and tritordeum transformants showing a high degree of marker gene stability and heritability. Theor Appl Genet 99:772–784
Chandler S, Dunwell JM (2008) Gene flow, risk assessment and the environmental release of transgenic plants. Crit Rev Plant Sci 27:25–49
Code of Federal Regulation #201.76. U.S. Code: Title 7—AGRICULTURE. § 201.76 Minimum Land, Isolation, Field, and Seed Standards. https://www.law.cornell.edu/cfr/text/7/201.76 Accessed 10 Sept 2015
D’Souza VL (1970) Investigations concerning the suitability of wheat as pollen-donor for cross-pollination by wind as compared to rye, Triticale, and Secalotricum. Z Pflzücht 63:246–269
De Vries AP (1971) Flowering biology of wheat, particularly in view of hybrid seed production—a review. Euphytica 20:152–170
De Vries AP (1972) Some aspects of cross pollination in wheat (Triticum aestivum L.) 1. Pollen concentration in the field as influenced by variety, diurnal pattern, weather conditions, and level as compared to the height of the pollen donor. Euphytica 21:185–203
Devos Y, Reheul D, De Schrijver A (2005) The Co-existence between transgenic and non-transgenic maize in European Union: a focus on pollen flow and cross-fertilization. Environ Biosaf Res 4:71–87
Dunwell JM (2014) Transgenic cereals: current status and future prospects. J Cereal Sci 59:419–434
Ellstrand NC (2003) Current knowledge of gene flow in plants: implications for transgene flow. Philos T Roy Soc B 358:1163–1170
FAOSTAT (2014) http://faostat3.fao.org. Accessed 01 Aug 2015
Foetzki A, Quijano CD, Moullet O, Fammartino A, Kneubuehler Y, Mascher F, Sautter C, Bigler F (2012) Surveying of pollen-mediated crop-to-crop gene flow from a wheat field trial as a biosafety measure. GM Crops Food 3(2):115–122
Gaines TA, Preston C, Byrne P, Henry WB, Westra P (2007) Adventitious presence of herbicide resistant wheat in certified and farm-saved seed lots. Crop Sci 47:749–754
Gatford KT, Zainuddin B, Edlington J, Lloyd J, Qureshi JA, Brettell R, Fincher GB (2006) Gene flow from transgenic wheat and barley under field conditions. Euphytica 151:383–391
Gustafson DI, Horak MJ, Rempel CB, Metz SG, Gigax DR, Hucl P (2005) An empirical model for pollen-mediated gene flow in wheat. Crop Sci 45:1286–1294
Hanson BD, Mallory-Smith CA, Shafii B, Thill DC, Zemetra RS (2005) Pollen mediated gene flow from blue aleurone wheat to other wheat cultivars. Crop Sci 45:1610–1617
Hucl P, Matus-Cádiz M (2001) Isolation distances for minimizing out-crossing in spring wheat. Crop Sci 41:1348–1351
Indian Minimum Seed Certification Standards (2013) The Central Seed Certification Board Department of Agriculture & Cooperation Ministry of Agriculture Government of India New Delhi. http://agricoop.nic.in/imagedefault/seed/INDIAN_MINIMUM_SEED_CERTIFICATION_STANDARDS.pdf
James C (2014) Global status of commercialized biotech/GM crops: 2014. ISAAA Brief 49-2014. ISAAA, Ithaca
Jensen NF (1968) Results of a survey on isolation requirements for wheat. Ann Wheat Newsl 15:26–28
Jones HD, Sparks CA (2009) Selection of transformed plants, Chapter 2. In: Jones HD, Shewry PR (eds) Transgenic wheat, barley and oats; production and characterisation protocols. Methods in Biotechnology. Humana Press, Totowa, pp 23–37
Khan MN, Heyne EG, Arp AL (1973) Pollen distribution and seedset on Triticum aestivum L. Crop Sci 13:223–226
Lawrie RG, Matus-Ca´diz MA, Hucl P (2006) Estimating outcrossing rates in spring wheat cultivars using the contact method. Crop Sci 46:247–249
Loureiro I, Escorial C, Santin I, Chueca C (2011) Pollen mediated gene flow in GM Crops: The use of herbicides as markers for detection. The case of wheat. In: Larramendy M (ed) Herbicides, Theory and application. InTech Europe. Croatia. doi: 10.5772/13042
Loureiro I, Escorial MC, González Á, Chueca MC (2012) Pollen-mediated gene flow in wheat (Triticum aestivum L.) in a semiarid field environment in Spain. Transgenic Res 21:1329–1339
Lu BR, Snow AA (2005) Gene flow from genetically modified rice and its environmental consequences. BioSci 55:669–678
Mallory-Smith C, Hall LM, Burgos NR (2015) Experimental methods to study gene flow. Weed Sci 63:12–22
Matus-Cádiz MA, Hucl P, Horak MJ, Blomquist LK (2004) Gene flow in wheat at the field scale. Crop Sci 44:718–727
Matus-Cádiz MA, Hucl P, Dupuis B (2007) Pollen-mediated gene flow in wheat at the commercial scale. Crop Sci 47:571–579
Messeguer J, Marfà V, Català MM, Guiderdoni E, Melé E (2004) A field study of pollen-mediated gene flow from Mediterranean GM rice to conventional rice and the red rice weed. Mol Breed 13:103–112
Miroshnichenko D, Filippov M, Babakov A, Dolgov S (2007a) Genetic engineering of Russian wheat genotypes for abiotic stress resistance. In: Burk HT, Nisi JE, Salomon N (eds) Developments in plant breeding, vol 12., Wheat production in stressed environmentsSpringer, Dordrecht, pp 715–721
Miroshnichenko D, Filippov M, Dolgov S (2007b) Genetic transformation of Russian wheat cultivars. Biotechnol Biotechnol Equip 4:399–402
Miroshnichenko DN, Poroshin GN, Dolgov SV (2011) Genetic transformation of wheat using mature seed tissues. Appl Biochem Microbiol 47(8):767–775
National Standard of Russian Federation, GOST 52325-2005 (2005) Seeds of agricultural crops. Varietal and sowing characteristics. General specifications (in Russian)
Richards HA, Rudas VA, Sun H, McDaniel JK, Tomaszewski Z, Conger BV (2001) Construction of a GFP-BAR plasmid and its use for switchgrass transformation. Plant Cell Rep 20:48–54
Rieben S, Kalinina O, Schmid B, Zeller S (2011) Gene flow in genetically modified wheat. PLoS One 6(12):e29730. doi:10.1371/journal.pone.0029730
Ritala A, Nuutila AM, Aikasalo R, Kauppinen V, Tammisola J (2002) Measuring gene flow in the cultivation of transgenic barley. Crop Sci 42:278–285
Rogers SO, Bendich AJ (1994) Extraction of total cellular DNA from plants, algae and fungi. In: Gelvin SB, Schilperoort RA (eds) Plant molecular biology manual, 2nd edn. Kluwer Academic Publishers, Dordrecht, pp 183–190
Rong J, Lu BR, Song ZP, Su J, Snow AA, Zhang XS, Sun SG, Chen R, Wang F (2007) Dramatic reduction of crop-to-crop gene flow within a short distance from transgenic rice fields. New Phytol 173:346–353
Rong J, Song ZP, de Jong TJ, Zhang XS, Sun SG, Xu X, Xia H, Liu B, Lu BR (2010) Modelling pollen-mediated gene flow in rice: risk assessment and management of transgene escape. Plant Biotechnol J 8:452–464
Rong J, Wang F, Song Z, Su J, Chen R, Lu BR (2012) Scale effect on rice pollen-mediated gene flow: implications in assessing transgene flow from genetically engineered plants. Ann Appl Biol 161(1):3–11
Schmidt M, Bothma G (2006) Risk assessment for transgenic sorghum in Africa: crop-to-crop gene flow in Sorghum bicolor (L.) moench. Crop Sci 46:790–798
Seed Services Australia (2013) Seed Certification Manual. 8.2. Field Crop & Other Species. Published by a business unit of the Rural Solutions SA Division of Primary Industries & Resources SA (PIRSA). http://pir.sa.gov.au/__data/assets/pdf_file/0003/148134/Seed_Certification_Manual.pdf
Virmani SS, Edwards IB (1983) Current status and prospects for breeding hybrid rice and wheat. Adv Agron 36:145–214
Westgate ME, Passioura JB, Munns R (1996) Water status and ABA content of floral organs in drought-stressed wheat. Aust J Plant Physiol 23:763–772
Willenborg CJ, Brule-Babel AL, Van Acker RC (2009) Low crop plant population densities promote pollen-mediated gene flow in spring wheat (Triticum aestivum L.). Transgenic Res 18:841–854
Yuan QH, Shi L, Wang F, Cao B, Qian Q, Lei XM, Liao YL, Liu WG, Cheng L, Jia SR (2007) Investigation of rice transgene flow in compass sectors by using male sterile line as a pollen detector. Theor Appl Genet 115(4):549–560
Acknowledgments
This work was supported by the Ministry of Science and Education of Russian Federation, Grant No 14.M04.12.0015. Appreciation is expressed to the staff of the Institute of Horticultural Crops Breeding of Russian Federation for help with the field trials. We also thank Ekaterina Sidonskaya for her assistance with PCR analysis and Yuliya Chernykh, Grigory Poroshin and Mikhail Filippov for their technical assistance throughout the research. We appreciate Mariya Chernobrovkina (All Russian Research Institute of Agricultural Biotechnology, Russia) for English editing of the manuscript and helpful comments on the manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Miroshnichenko, D., Pushin, A. & Dolgov, S. Assessment of the pollen-mediated transgene flow from the plants of herbicide resistant wheat to conventional wheat (Triticum aestivum L.). Euphytica 209, 71–84 (2016). https://doi.org/10.1007/s10681-016-1637-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10681-016-1637-y